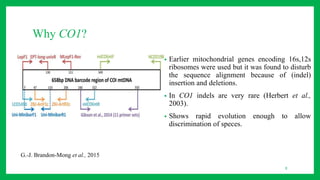
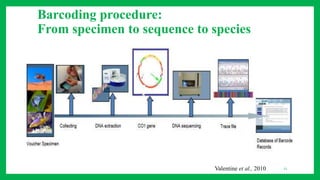
The document discusses the application of DNA barcoding in conserving insect biodiversity, highlighting its strengths and weaknesses. It details the methodology of DNA barcoding, the significance of the CO1 gene, and the process of developing DNA barcode libraries for species identification. Additionally, it emphasizes the importance of integrating DNA barcoding with other conservation strategies for effective outcomes.