Ventricular Septal Defect with Nursing Management
- 2. Excluding biscuspid aortic valves, ventricular septal defects (VSDs) are the most common congenital cardiac lesion, occurring in isolation or in association with other congenital cardiac defects. The ventricular septum is divided into four components, the trabecular or muscular septum extending to the apex, the inlet (posterior) septum between the atrioventricular valves, the outlet septum which subtends the great arteries and the membranous septum which lies under the aortic root. Defects can arise in any position but the membraneous septum is most commonly affected.
- 3. The ventricular septum forms the medial wall and normally bulges in to the right ventricle. It is almost muscular except for the superiorly located membraneous septum just below the right and posterior (noncoronary) cusps of te aortic valve. On the left ventricular side the demarcation between the membraneous septum and the muscular septum is called limbus marginalis. The membranous septum is both interventricular and atrioventricular. The former portion lies between the left ventricle and the right ventricle. The latter, superior, part of the membraneous septum lies between the left atrium and the right atrium.
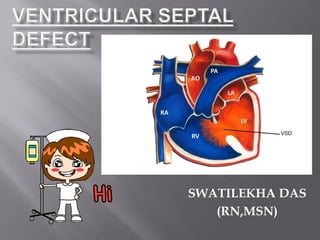
- 4. Anatomy of ventricular septum
- 5. A ventricular septal defect is an opening in the interventricular septum that permits communication between the two ventricles. or A VSD is an opening or hole (defect) in the wall (septum) between the heart’s two pumping chambers (ventricles).
- 7. Before a baby is born, the right and left ventricles of its heart are not separate. As the fetus grows, a wall forms to separate these two ventricles. If the wall does not completely form, a hole remains. This hole is known as a ventricular septal defect, or a VSD. Ventricular septal defect is one of the most common congenital heart defects. The baby may have no symptoms, and the hole can eventually close as the wall continues to grow after birth. If the hole is large, too much blood will be pumped to the lungs, leading to heart failure. The cause of VSD is not yet known. This defect often occurs along with other congenital heart defects. In adults, ventricular septal defects are a rare but serious complication of heart attacks. These holes do not result from a birth defect.
- 9. Defects of the ventricular septum, which may be large in relation to the size of the heart at birth, tend to become smaller or to close in early childhood. If closure is insufficient to prevent a large shunt, the small pulmonary vessels may be damaged by being exposed to the ejectile force and pressure of left ventricular contraction. Irreversible pulmonary hypertension may be produced. The effect of a ventricular septal defect (VSD) depends upon its size and upon the impedance to blood flow imposed by the pulmonary arterial vessels. If the defect is small, the jet of blood from the high pressure left ventricle to the low-pressure right ventricle has little hemodynamic effect. If the defect is large and the impedance of the pulmonary vessels low, a large shunt develops and the pulmonary blood flow becomes substantially more than systemic. If , on the other hand, there is a high pulmonary vascular resistance, the pulmonary blood flow is little or no more than the systemic and the pressure in both circuits is similar. If the pulmonary vascular resistance is very high, the shunt reverses (the so called Eisenmenger Complex).
- 11. Symptoms In VSD the patient with a small defect, there are no symptoms, but there is a loud ‘tearing’ pansystolic murmur often accompanied by a thrill, maximal to the left side of the lower sternum. Newborn infants have a high pulmonary vascular resistance which falls to normal levels in the first 3 months of life. At this point, a large defect at ventricular level is liable to produced cardiac failure and require treatment.
- 12. Signs Cardiac failure is usually obvious in the infant or small child with a significant left to right shunt through a large VSD. In the presence of a large left to right shunt the pulse is usually small and the venous pressure normal, unless there is right heart failure. Both left and right ventricles may be hyperdynamic and with a large shunt, a soft pansystolic murmur is heard at the lower sternal edge, usually accompanied by a mid-diastolic murmur at the apex due to high flow through the mitral valve,
- 13. Although there are some patients in whom large defects get smaller and become restrictive to flow, most, if untreated will ultimately progress to irreversible pulmonary hypertension as a result of pulmonary vascular disease. The development of pulmonary vascular disease is often heralded by an improvement in the symptoms of cardiac failure as the resistance to follow through the lungs decreases flow. Right ventricular hypertrophy is evident, the pulmonary second sound may be accentuated and followed by the early diastolic murmur of pulmonary regurgitation.
- 14. HISTORY- Infants with larger defects develop heart failure, usually by 3 months of age. Parents may note tachypnea, fatigue with feedings, poor weight gain and excessive sweating. Infants and children with small ventricular defects are asymptomatic; their defects are ordinarily detected early by routine examination because of the loudness of the murmur. PHYSICAL EXAMINATION- Typical loud, harsh holosystolic murmur Middiastole rumble Pulmonary rales and peripheral edema The second heart sound is loud and single cyanosis CHEST X-RAY It is normal with a small defect, but with a large left to right shunt there is some enlargement of the heart and, more specifically , prominence of the pulmonary vessels, left atrium and both ventricles.
- 15. ECG The ECG in small defects is normal. When the left to right shunt is large, there is usually evidence of biventricular enlargement, manifested by abnormally deep but narrow Q waves and tall R waves in the left chest leads. In cases with pulmonary vascular disease, the ECG pattern of isolated right ventricular hypertrophy develops. Large defects may involve the conducting tissue and left axis deviation occurs. ECHOCARDIOGRAPHY Two-dimensional echocardiography, along with Doppler echocardiography and colour flow imaging can assess the size and location of virtually all ventricular septal defects (VSDs). Doppler echocardiography also provides physiological information including right ventricular pressure, pulmonary artery pressure and the difference in pressure between the ventricles. Measurement of left atrial and left ventricular diameter provides semi- quantitative information about shunt volume. CARDIAC CATHETERIZATION There is a step-up in oxygen saturation at the right ventricular level. The degree of pulmonary hypertension can be measured, and pulmonary vascular resistance should be calculated. Selective biplane cineangiography in the left ventricle will outline the defect. 1. Establish diagnosis 2. Determine type and severity 3. Confirm and exclude associated lesions
- 16. Medications With a small lesion the prognosis is excellent and no intervention is required beyond antibiotic prophylaxis where indicated. In moderate size lesions a trial of furosemide, angiotensin- converting enzyme (ACE) inhibitor and digoxin may work. Large lesions with a big shunt usually require medical management for heart failure with diuretics ± ACE inhibitors. Otherwise surgical closure is required before vascular disease compromises the surgical outcome – usually within the first 6-12 months of life Surgical repair is required if: There is uncontrolled heart failure, including poor growth. Even very small babies may be considered. If the pulmonary artery pressure is raised then repair may be considered before the first birthday. Even with normal pulmonary artery pressure, if the ratio of aortic flow: pulmonary flow exceeds 2:1, then repair is required.
- 17. Palliative: Pulmonary artery banding (placing a band around the main pulmonary artery to decease pulmonary blood flow) in infants in severe CHF. It is unusual because of the improvement of the surgical techniques. Complete repair(procedure of choice): Small defects are repaired with a purse-string approach. Large defects usually require a knitted Dacron patch sewn over the opening. Both procedures are performed via cardiopulmonary bypass. The repair is generally approached through the right atrium and the tricuspid valve.
- 18. Except for VSD that is caused by a heart attack, this condition is always present at birth. Drinking alcohol and using the antiseizure medicines depakote and dilantin during pregnancy may increase the risk of VSDs. Other than avoiding these things during pregnancy, there is no known way to prevent a VSD.
- 19. Aortic insufficiency (leaking of the valve that separates the left ventricle from the aorta) Damage to the electrical conduction system of the heart during surgery (causing an irregular heart rhythm) Delayed growth and development (failure to thrive in infancy) Heart failure Infective endocarditis (bacterial infection of the heart) Pulmonary hypertension (high blood pressure in the lungs) leading to failure of the right side of the heart Postoperative problems can occur: A small residual ventricular septal defect (VSD) (not uncommon). Depending upon heart size, symptoms and degree of shunting it may be necessary to re-operate. Right bundle branch block may be caused by operative trauma, and occasionally complete heart block can occur. This has a late mortality. Ventricular dysfunction and ventricular arrhythmia can be a problem.
- 20. Nursing diagnosis Cough related to lung congestion secondary to the effect of general anesthesia Goal Reduce the coughing of the patient Interventions Assess the patient for coughing Auscultate chest of the patient with pediatric stethoscope Give chest physiotherapy. Give nebulisation with Levolin as dose of ¼ resp. Auscultate . after giving nebulization Expected outcomes Coughing will be reduced.
- 21. Nursing diagnosis High risk for fall related to the age and weakness of the child Goal Reduce the chances of fall Interventions Assess the chances of getting fall. Provide side rails to prevent fall. Provide calling bell for the patient. Allow the mother to stay with the patient. Do close observation of the patient. Expected outcomes No fall of the patient will be there.
- 22. Nursing diagnosis Pain related to the surgical wound as evidenced by facial pain scale Goal Reduce pain Interventions Assess the location & source of pain. Assess the severity of pain. Assess the patient from facial expression as per pediatric pain scale. Provide comfortable position. Administer oral analgesics (Syrup Ibugesic plus) 2.5ml as ordered by physician. Expected outcomes Pain will be reduced.
- 23. Nursing diagnosis High risk for infection related to the presence of surgical wound Goal Reduce risk of getting infection. Interventions Assess patient for signs of infection. Maintain aseptic technique during any procedure. Do hand washing before and after each procedure, touching patient. Check temperature every 4 hourly and SOS. Administer prescribed antibiotics (Syrup Augmentin Duo) 2.5ml as prescribed by physician. Check vital signs and record it. Expected outcomes No infection will be present.
- 24. Nursing diagnosis Knowledge deficit of parents of the child related to the disease process as evidenced by frequent questioning Goal Increase knowledge level of the parents. Interventions Assess the knowledge level of the parents of the patient. Provide information about disease process of the patient. Clear doubts of the parents. Explain about medicines, diet, comfort and sleep, wound care and follow up care. Take feedback from the parents. Expected outcomes Knowledge level of the parents will be increased.
- 25. Nursing diagnosis Anxiety of parents related to the disease process, treatment regimen, hospital stay as evidenced by facial expression and verbalization Goal Reduce anxiety of the parents. Interventions Assess the anxiety level of the parents. Provide information about disease process, treatment regimen, and hospital stay. Give psychological support to the parents. Counsel the parents if needed. Expected outcomes Anxiety of the parents will be reduced.
- 26. Fortunately most ventricular septal defects are small. Many become smaller close spontaneously during childhood. Among patients with large defects congestive heart failure is likely in infancy and causes death unless managed appropriately.
