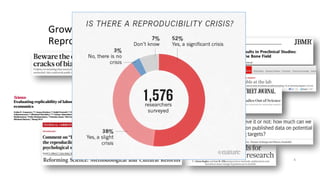
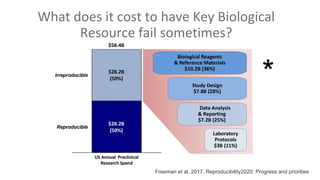
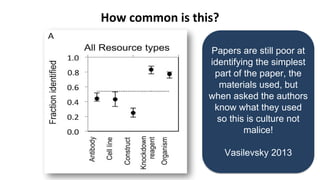
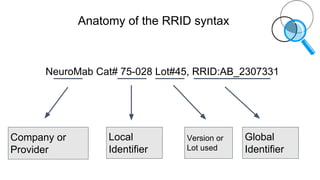
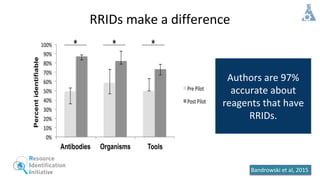
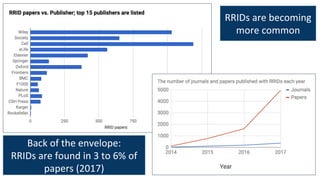
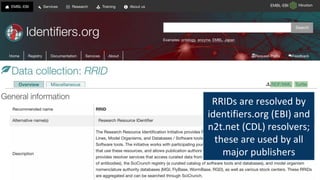
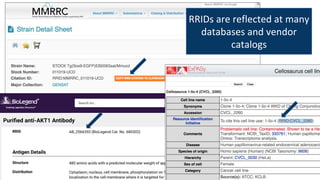
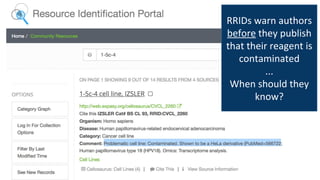
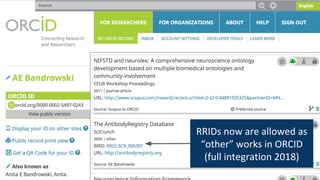
RRIDs were developed to enhance rigor and transparency in research, supporting NIH guidelines for the better identification of resources like antibodies and cell lines. They help institutions track resources, aid compliance with guidelines, and serve as a warning system for resource quality. The document emphasizes the collaborative effort in promoting RRIDs and provides resources for authentication and further information.