Week 1 Introduction to Atoms.pptx
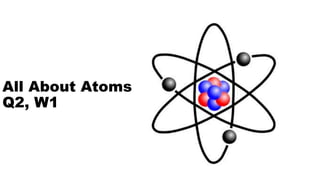
- 1. All About Atoms Q2, W1
- 2. Chemistry The study of the composition of matter and the changes that matter undergoes.
- 3. Created by G.Baker www.thesciencequeen.net •Matter is anything that takes up space and has mass. •All matter is made of atoms •Atoms are the building blocks of matter, sort of how bricks are the building blocks of houses.
- 5. Atom Atom – smallest particle of an element that can exist alone
- 6. Structure of the Atom Two regions of an atom •Nucleus Center of atom Protons and neutrons •Electron “cloud” Area surrounding nucleus containing electrons
- 8. No charge or 0 + -
- 9. Subatomic Particles Particle Symbol Charge Relative Mass e- -1 1/1840 p+ +1 1 1 0 n Electron Proton Neutron
- 10. Counting Atoms Atomic Number •Number of protons in nucleus •The number of protons determines identity of the element!!
- 13. •Mass Number (Atomic Mass) •Number of protons + neutrons •Units are g/mol
- 14. Isotopes •Atoms of the same element with varying number of neutrons •Different isotopes have different mass numbers because the number of neutrons is different
- 15. Ions- is a charged atom or molecule. It is charged because the number of electrons do not equal the number of protons in the atom or molecule. Cation = a positive ion Anion = a negative ion
- 17. •Mass Number = # protons + # neutron •# of electron = # protons = atomic number •Neutron= mass number- # of proton
- 18. Let’s Practice Aluminum (Al) (no periodic table) •Protons = 13 •Electrons = •Neutrons = 14 •Atomic Number = •Atomic Mass =
- 19. Let’s Practice w/ nuclear symbol notation •Nuclear Symbol notation (no periodic table) •Protons = •Electrons = •Neutrons = •Atomic Number = •Atomic Mass = 108 47
- 20. Let’s Practice w/ isotopes •use the periodic table •Protons = •Electrons = •Neutrons = •Atomic Number = •Atomic Mass = Uranium-235
- 21. Let’s Practice with Ions Use the periodic table •Charge = •Protons = •Electrons = •Neutrons = •Atomic Number = •Atomic Mass = K 39 1+