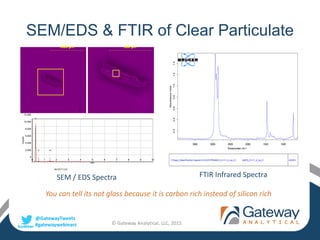
The document outlines a webinar by Gateway Analytical focusing on foreign particulate analysis in pharmaceutical manufacturing. It covers the characteristics, sources, and analysis methods of foreign particulate matter, while also providing case studies on contamination issues, such as hair and embedded materials in vials. Attendees will receive a replay link, presentation slides, and a digital certificate upon completion of the webinar.































![@GatewayTweets
#gatewaywebinars
Particle Size and Identification Using SPE™
Automated Raman System
© Gateway Analytical, LLC, 2015
Spectra # Material
Size Distribution [µm]
2 - 10 10 - 25 25 - 50 50 - 100 100-1000 Totals
1 Amorphous Carbon 7 0 0 0 0 7
2 beta-Carotene 1 0 0 0 0 1
3 Blue Pigment 28 3 2 4 2 39
4 Boot cover 0 0 0 1 0 1
5 Boron Carbide 1 1 1 6 3 12
6 Calcium Chloride 1 0 0 0 0 1
7 Cellulose 1 0 0 1 0 2
10 storage bag 0 0 1 1 0 2
11 Filling suite, Ext 12 2 0 3 0 17
12 Filter from air line 0 0 0 0 1 1
13 Fluorescence 175 24 26 74 28 327
17 Gowning material 1 0 0 3 0 4
18 Plunger 0 1 6 26 10 43
20 Laurylsulfate Sodium, SDS 0 0 0 1 0 1
21 Nitrate Sodium 0 0 0 2 0 2
25 Plastic bags 0 0 0 1 0 1
26 Poly(acrylonitrile) 4 2 0 1 1 8
28 Polycarbonate 2 0 0 0 0 2
30 Polysulfone 1 0 0 0 0 1
31 Porous Particle Production Air 7 0 0 0 0 7
32 Sample 1 1 0 0 0 0 1
33 Sample 5 0 0 0 1 0 1
37 Smalt 1 0 0 0 0 1
38 Starch 1 0 0 1 0 2
40 Talcum 8 3 0 1 0 12
41 Titanium(IV)oxide, Anatase 2 0 0 1 0 3
42 gasket 0 0 1 0 0 1
All Particles 10,260 1548 460 128 45 12,441](https://image.slidesharecdn.com/webinar-casestudiesinforeignparticulateanalysis-150727164115-lva1-app6891/85/Webinar-Case-Studies-in-Foreign-Particulate-Analysis-33-320.jpg)