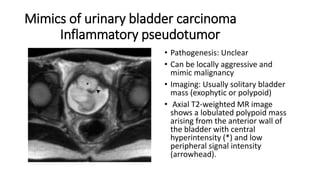
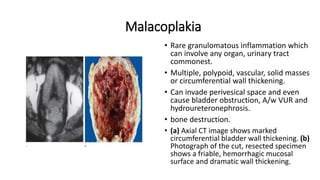
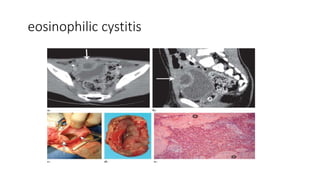
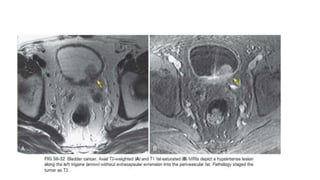
Urinary bladder cancer is the fourth most common cancer in men and tenth most common in women. About 90% are urothelial in origin. Risk factors include smoking, occupational exposure to chemicals, and genetic predisposition. Tumors are classified as non-invasive papillary or invasive into the muscle. Diagnosis is usually via cystoscopy following hematuria detection. Staging involves CT, MRI, or PET to assess tumor depth, lymph node involvement, and distant metastasis. Treatment depends on stage, with superficial tumors addressed via surgery and chemotherapy or immunotherapy, while muscle-invasive tumors require radical cystectomy.






















![• Positron Emission Tomography
Positron emission tomography (PET) with 2-[fluorine-18]fluoro-2-
deoxy-d-glucose is considered to be of lesser value in the local staging
of bladder cancer due to urinary excretion of the radiotracer](https://image.slidesharecdn.com/urinarybladderpathologyradiology-200213182924/85/Urinary-bladder-pathology-radiology-26-320.jpg)


![Treatment
• Non-Muscle-invasive Disease
Non-muscle-invasive tumors are usually treated with TURBT.
in patients with bulky Ta lesions or T1 tumors, the current trend is to perform
repeat TURBT 2–6 weeks after the first TURBT.
adjuvant intravesical chemotherapy (eg, with mitomycin C or gemcitabine) or
immunotherapy (with bacille Calmette-Guérin [BCG]).
Muscle-invasive Disease
For muscle-invasive tumors, radical cystectomy is the established treatment
with meticulous extended bilateral lymphadenectomy, since extent of
lymphadenectomy is associated with survival.](https://image.slidesharecdn.com/urinarybladderpathologyradiology-200213182924/85/Urinary-bladder-pathology-radiology-30-320.jpg)