- Therapix Biosciences is a clinical-stage pharmaceutical company focusing on proprietary synthetic cannabinoid technologies for central nervous system indications.
- They are developing novel sublingual and nasal formulations of FDA-approved synthetic THC (dronabinol) for indications like Tourette Syndrome and Mild Cognitive Impairment.
- For Tourette Syndrome, they are combining THC with PEA in a proprietary combination therapy called THX-TS01 that has begun Phase II clinical trials based on a 505(b)(2) regulatory pathway.
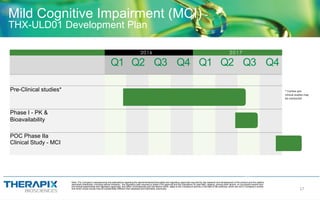
- For Mild Cognitive Impairment, they are developing an "Ultra-Low Dose THC" therapy called THX-ULD