The history of the Balmer series
•
0 likes•114 views
From the history of the Balmer series, we can understand that physicists' experiments on the atomic emission spectrum started in the 1550s. But they lacked an empirical formula to imagine the wavelengths of spectral lines of an element. In 1885, the Balmer equation was the first empirical formula discovered to estimate the wavelengths of spectral emissions in the hydrogen spectrum. The Balmer equation was given by the mathematician Johann Jakob Balmer.
Report
Share
Report
Share
Download to read offline
Recommended
Supernova- Part 1

The document discusses different types of supernovae and their potential effects on Earth. It describes the Crab Nebula, formed from the remnants of a 1054 supernova, and supernova remnant N 63A in the Large Magellanic Cloud. Type Ia supernovae, which originate from white dwarfs, are considered most dangerous if close enough to affect Earth by depleting the ozone layer through gamma rays. A nearby supernova within 100 light years could impact the biosphere and may have caused a past extinction event.
Explanation Text About Aurora

Aurora are bright glows caused by collisions between charged particles from the solar wind and atoms in the Earth's upper atmosphere. The solar wind carries the Sun's magnetic field and particles through space at around 400 km/s, shaping the Earth's magnetosphere into an elongated bubble. Under certain conditions, the solar wind's magnetic field can merge with Earth's, creating electrical currents that drive particles into the polar atmosphere, causing the visible auroral displays usually seen in night skies at high latitudes, known as the northern lights in the northern hemisphere and southern lights in the southern hemisphere.
Explanation Text - Aurora

The document discusses auroras, or the northern and southern lights. It explains that auroras are natural light displays in the sky, predominantly seen in high latitude regions near the Arctic and Antarctic. Auroras are caused by charged particles from space interacting with gases in the atmosphere, which causes the gases to glow in colorful lights. Auroras typically occur in a band near the magnetic poles of the Earth and can expand during geomagnetic storms, bringing the lights to lower latitudes where they may be visible as a greenish or red glow along the horizon. Similar auroras also occur on other planets near their magnetic poles.
Aurora

Auroras are natural light displays in the sky, typically observed at night in polar regions. Charged particles from the solar wind are captured by Earth's magnetosphere and drawn to the magnetic poles, where excited electrons emit energy in the form of light creating the aurora effect. Auroras can take different forms like poleward arcs, theta aurora, and dayside arcs and occur in various colors from flickering to black or subvisual red arcs.
The Aurora Borealis

The Northern Lights, or Aurora Borealis, are caused by interactions between the Earth's magnetic field and particles from the Sun. When solar storms eject electrons and protons from the Sun's surface, they are guided by the magnetic field toward the magnetic poles, where they collide with gases in the atmosphere and release energy in the form of colorful light displays. The auroral oval, which is the main region where the Northern Lights occur, is centered around the magnetic poles and can vary in size and location depending on the level of solar activity and strength of the solar wind. The solar atmosphere reaches temperatures of millions of degrees, causing hydrogen to break into electrons and protons that are then carried away in the solar wind toward Earth and other planets
Polar exploration by o wen and scott

Polar exploration involved expeditions to both the Arctic and Antarctic regions led by notable explorers like Scott, Shackleton, and Amundsen. Roald Amundsen became the first person to reach the South Pole, beating Robert Falcon Scott who was the second explorer to arrive. Amundsen brought husky dogs on his expedition which were better suited to the extreme cold compared to the ponies used by Scott.
Polar exploration by o wen and scott2

Polar exploration involved expeditions led by famous explorers like Scott, Shackleton, and Amundsen to the Arctic and Antarctic regions. Roald Amundsen became the first person to reach the South Pole in 1911, beating Robert Falcon Scott who arrived second after encountering difficulties with his pony transport in the extreme cold weather conditions.
The emergence of aurora borealis

The Aurora Borealis Tours in Iceland is not only interesting, but, it also gives an amazing display which no one can forget.
Recommended
Supernova- Part 1

The document discusses different types of supernovae and their potential effects on Earth. It describes the Crab Nebula, formed from the remnants of a 1054 supernova, and supernova remnant N 63A in the Large Magellanic Cloud. Type Ia supernovae, which originate from white dwarfs, are considered most dangerous if close enough to affect Earth by depleting the ozone layer through gamma rays. A nearby supernova within 100 light years could impact the biosphere and may have caused a past extinction event.
Explanation Text About Aurora

Aurora are bright glows caused by collisions between charged particles from the solar wind and atoms in the Earth's upper atmosphere. The solar wind carries the Sun's magnetic field and particles through space at around 400 km/s, shaping the Earth's magnetosphere into an elongated bubble. Under certain conditions, the solar wind's magnetic field can merge with Earth's, creating electrical currents that drive particles into the polar atmosphere, causing the visible auroral displays usually seen in night skies at high latitudes, known as the northern lights in the northern hemisphere and southern lights in the southern hemisphere.
Explanation Text - Aurora

The document discusses auroras, or the northern and southern lights. It explains that auroras are natural light displays in the sky, predominantly seen in high latitude regions near the Arctic and Antarctic. Auroras are caused by charged particles from space interacting with gases in the atmosphere, which causes the gases to glow in colorful lights. Auroras typically occur in a band near the magnetic poles of the Earth and can expand during geomagnetic storms, bringing the lights to lower latitudes where they may be visible as a greenish or red glow along the horizon. Similar auroras also occur on other planets near their magnetic poles.
Aurora

Auroras are natural light displays in the sky, typically observed at night in polar regions. Charged particles from the solar wind are captured by Earth's magnetosphere and drawn to the magnetic poles, where excited electrons emit energy in the form of light creating the aurora effect. Auroras can take different forms like poleward arcs, theta aurora, and dayside arcs and occur in various colors from flickering to black or subvisual red arcs.
The Aurora Borealis

The Northern Lights, or Aurora Borealis, are caused by interactions between the Earth's magnetic field and particles from the Sun. When solar storms eject electrons and protons from the Sun's surface, they are guided by the magnetic field toward the magnetic poles, where they collide with gases in the atmosphere and release energy in the form of colorful light displays. The auroral oval, which is the main region where the Northern Lights occur, is centered around the magnetic poles and can vary in size and location depending on the level of solar activity and strength of the solar wind. The solar atmosphere reaches temperatures of millions of degrees, causing hydrogen to break into electrons and protons that are then carried away in the solar wind toward Earth and other planets
Polar exploration by o wen and scott

Polar exploration involved expeditions to both the Arctic and Antarctic regions led by notable explorers like Scott, Shackleton, and Amundsen. Roald Amundsen became the first person to reach the South Pole, beating Robert Falcon Scott who was the second explorer to arrive. Amundsen brought husky dogs on his expedition which were better suited to the extreme cold compared to the ponies used by Scott.
Polar exploration by o wen and scott2

Polar exploration involved expeditions led by famous explorers like Scott, Shackleton, and Amundsen to the Arctic and Antarctic regions. Roald Amundsen became the first person to reach the South Pole in 1911, beating Robert Falcon Scott who arrived second after encountering difficulties with his pony transport in the extreme cold weather conditions.
The emergence of aurora borealis

The Aurora Borealis Tours in Iceland is not only interesting, but, it also gives an amazing display which no one can forget.
de-Broglie equation.pdf

If everyone is thinking alike, we cannot solve the hurdles of a problem. It seems true if we observe these hypotheses' hierarchy once. Italian physicist Francesco Maria Grimaldi discovered the wave phenomenon of light in 1665. But the uncertainty about light's nature was finally solved by Einstein's explanation of the photoelectric effect. Similarly, Neil Bohr succeeded in describing the structure of an atom with quantized electron orbits. But his stipulation of allowed stationary orbits was only a supposition until the discovery of the de-Broglie equation.
Louis de-Broglie, a French physicist, presumed that moving microscopic and macroscopic objects are waves. He introduced a word called 'matter wave' to describe the waves of material objects in motion. As a result, matter exhibits a dual character of both particle and wave. Moreover, he derived an empirical formula to measure the wavelength of matter waves in 1923 called the de-Broglie equation.
Wien displacement law graph.pdf

It is a graphical explanation of a blackbody's emissive power and radiation wavelength at various temperatures. We know blackbody emissions are temperature specific. And a blackbody graph is an overall variation of a blackbody's emissive power as a function of wavelength measured at different temperatures. But the Wien displacement law graph focuses on spectral intensities of blackbody emissions as a function of wavelength at a peak wavelength state λmax at a specific temperature T.
MCQs of blackbody & its radiation .pdf

It is an e-book of MCQs & answers on blackbody. It is in PowerPoint format with 40 questions and detailed answer explanation that makes you clear every single point of blackbody and its radiation. The e-book has 54 beautifully designed slides. The contents of the e-book are below.
Multiple choice questions and answers - 20 no.s
True or false questions-10 no.s
Reasoning questions - 10 no.s
To disclose all the contents of the e-book, visit this link.
https://kameswariservices201831.myinstamojo.com/product/3556812/mcqs-answers-of-blackbody-and-its-radiation
Fatafat check list of blackbody.pdf

A blackbody is an imaginary object that absorbs all incident light without reflecting any, invented by German physicist Gustav Kirchhoff in 1860 to help understand thermal radiation. Examples of materials that approximate a blackbody include lamp black, graphite, and platinum black. The energy distribution of electromagnetic radiation emitted by a blackbody, known as blackbody radiation, depends only on the body's temperature according to Planck's law.
Wien displacement law .pdf

It discusses Wien displacement law. To go through the numerical problems of the Wien displacement law topic, visit our e-book at;
https://kameswariservices201831.myinstamojo.com/product/3493453/numerical-problems-on-wien-displacement-law-
Blackbody radiations infographic.pdf

The blackbody is a hollow enclosure with a pinhole to emit its radiations. The secure covering of the blackbody prevents the absorbed light from escaping.
A blackbody is a solid closed unreal body that is inexistent. But lamp black, platinum black, and graphite-coated surfaces are non-ideal black bodies for laboratory purposes. An object with above 0.95 emissivities is an approximate blackbody. Besides, the hotter bodies emitting electromagnetic radiation under thermal equilibrium conditions are also considered partial black bodies.
Blackbody mind map.pdf

An ideal black body is an imaginary concept developed by Gustav Kirchhoff that perfectly absorbs all electromagnetic radiation that falls on it and reflects none. It plays a significant role in quantum mechanics by being a perfect emitter and absorber of thermal radiation, releasing electromagnetic radiation when heated that follows specific laws like Planck's quantum law, Wien's displacement law, and Stefan-Boltzmann law. Real blackbodies have opaque, closed surfaces that absorb radiation based on factors like temperature, emissivity, and surface area, and their radiation is used in applications like thermal imaging, optical sensors, and burglar alarms.
Blackbody Vs. graybody.pdf

It explains the difference between a blackbody and a gray body following Kirchhoff's law of thermal radiation.
MCQs of blackbody & Kirchhoff's law.pdf

It is a PowerPoint presentation on multiple choice questions and answers on Blackbody & Kirchhoff's law topics. It discusses all practical examples and numerical problems of the said concepts.
Planck quantum theory.pdf

This PowerPoint presentation explains Planck's quantum theory and Planck's constant topics briefly. It is now available on Instamojo for just Rs.10/-. Save a personalized copy of this presentation by visiting Instamojo. The link is below.
https://kameswariservices201831.myinstamojo.com/product/3439450/planck-quantum-theory-postulates
Kirchhoff's law (1).pdf

It is a PowerPoint presentation of Kirchhoff's law of thermal radiation. And it gives a brief idea of Kirchhoff's law, its applications, and examples. Moreover, it explains the terms absorptivity, emissive power, thermal equilibrium, and emissivity.
Kirchhoff's law.pdf

It discusses Kirchhoff's law and its examples. It explains blackbody emissions in thermal equilibrium conditions.
We are sorry to inform you about a small error in Kirchhoff's formula section of the above infographic. Kirchhoff's formula states that the absorptivity and emissivity of the body are equal in thermal equilibrium conditions. 'a' is the absorbing power of the body at a wavelength λ. And 'e' is the emissivity of the body at the same wavelength. So, we will write aλ=e but not aλ=eλ. We request you read it correctly while going through the PDF.
Kirchhoff's law formula and its derivation. Numerical problems.pdf

It discusses Kirchhoff's formula and its derivation. And it includes numerical problems of Kirchhoff's law. For a colorful e-book for only 10/- rupees on Kirchhoff's law, visit our store at;
https://kameswariservices201831.myinstamojo.com/product/3467221/kirchhoffs-law-of-thermal-radiations
Planck's constant.pdf

It describes the definition of Planck's constant. Planck constant helps compute the discrete energy changes of a body by relating to the frequency of the photon. Planck constant explains the proportionality relationship between the photon's energy and the frequency.
For more information on this topic, kindly visit our blog article at;
https://jayamchemistrylearners.blogspot.com/2022/08/plancks-constant-chemistry-learners.html
Numerical problems of Planck's quantum theory.pdf

It is a PowerPoint presentation on numerical problems of Planck quantum theory topic.
It includes 12 numerical problems with solved answers. Besides, this PowerPoint presentation has a mind map to remember all formulas of Planck quantum law.
MCQ on Planck constant.pdf

This document contains 10 multiple choice questions about Planck's constant. It provides explanations for why Planck's constant was introduced, its value in different units, and that it explains both the quantum and particle nature of light. The questions cover topics like the relationship between photon energy and frequency, experimental methods used to determine Planck's constant, and why LED lights are used in these calculations. Additional resources on Planck's constant and quantum theory are provided at the end.
Detailed light theories.pdf

It explains both wave & particle theories of light. It also includes Maxwell's mathematical equations and postulates of Planck quantum theory.
Theories of light.pdf

It explains the theories of light, including electromagnetic wave theory and Planck's quantum theory.
MCQ's on hydrogen spectrum.pdf

The document contains 10 multiple choice questions about the hydrogen spectrum. It discusses that the essential condition to observe the hydrogen spectrum is high temperature and low pressure. The hydrogen spectrum appears as distinct lines, with the prominent line being red with a wavelength of 656 nm. It is an example of a line spectrum and can be recorded using a spectroscope. Robert Bunsen and Gustav Kirchhoff discovered the hydrogen spectrum and spectroscope, while Isaac Newton first discovered the word "spectrum".
Rydberg equation.pdf

The Rydberg formula helps to determine the wavenumber or wavelengths of hydrogen spectral lines obtained in the hydrogen spectrum. Previously, Johann Jakob Balmer discovered an empirical formula to determine the wavelengths of hydrogen spectral lines obtained in the visible region of the hydrogen spectrum. As we all know, the hydrogen spectrum is not limited to the visible zone only. It occupies the ultraviolet and infrared parts of the electromagnetic spectrum also. Hence, the scientists' quests to determine the spectral positions of various spectral lines of the hydrogen spectrum finally came to an end with the Rydberg formula.
More Related Content
More from SaiKalyani11
de-Broglie equation.pdf

If everyone is thinking alike, we cannot solve the hurdles of a problem. It seems true if we observe these hypotheses' hierarchy once. Italian physicist Francesco Maria Grimaldi discovered the wave phenomenon of light in 1665. But the uncertainty about light's nature was finally solved by Einstein's explanation of the photoelectric effect. Similarly, Neil Bohr succeeded in describing the structure of an atom with quantized electron orbits. But his stipulation of allowed stationary orbits was only a supposition until the discovery of the de-Broglie equation.
Louis de-Broglie, a French physicist, presumed that moving microscopic and macroscopic objects are waves. He introduced a word called 'matter wave' to describe the waves of material objects in motion. As a result, matter exhibits a dual character of both particle and wave. Moreover, he derived an empirical formula to measure the wavelength of matter waves in 1923 called the de-Broglie equation.
Wien displacement law graph.pdf

It is a graphical explanation of a blackbody's emissive power and radiation wavelength at various temperatures. We know blackbody emissions are temperature specific. And a blackbody graph is an overall variation of a blackbody's emissive power as a function of wavelength measured at different temperatures. But the Wien displacement law graph focuses on spectral intensities of blackbody emissions as a function of wavelength at a peak wavelength state λmax at a specific temperature T.
MCQs of blackbody & its radiation .pdf

It is an e-book of MCQs & answers on blackbody. It is in PowerPoint format with 40 questions and detailed answer explanation that makes you clear every single point of blackbody and its radiation. The e-book has 54 beautifully designed slides. The contents of the e-book are below.
Multiple choice questions and answers - 20 no.s
True or false questions-10 no.s
Reasoning questions - 10 no.s
To disclose all the contents of the e-book, visit this link.
https://kameswariservices201831.myinstamojo.com/product/3556812/mcqs-answers-of-blackbody-and-its-radiation
Fatafat check list of blackbody.pdf

A blackbody is an imaginary object that absorbs all incident light without reflecting any, invented by German physicist Gustav Kirchhoff in 1860 to help understand thermal radiation. Examples of materials that approximate a blackbody include lamp black, graphite, and platinum black. The energy distribution of electromagnetic radiation emitted by a blackbody, known as blackbody radiation, depends only on the body's temperature according to Planck's law.
Wien displacement law .pdf

It discusses Wien displacement law. To go through the numerical problems of the Wien displacement law topic, visit our e-book at;
https://kameswariservices201831.myinstamojo.com/product/3493453/numerical-problems-on-wien-displacement-law-
Blackbody radiations infographic.pdf

The blackbody is a hollow enclosure with a pinhole to emit its radiations. The secure covering of the blackbody prevents the absorbed light from escaping.
A blackbody is a solid closed unreal body that is inexistent. But lamp black, platinum black, and graphite-coated surfaces are non-ideal black bodies for laboratory purposes. An object with above 0.95 emissivities is an approximate blackbody. Besides, the hotter bodies emitting electromagnetic radiation under thermal equilibrium conditions are also considered partial black bodies.
Blackbody mind map.pdf

An ideal black body is an imaginary concept developed by Gustav Kirchhoff that perfectly absorbs all electromagnetic radiation that falls on it and reflects none. It plays a significant role in quantum mechanics by being a perfect emitter and absorber of thermal radiation, releasing electromagnetic radiation when heated that follows specific laws like Planck's quantum law, Wien's displacement law, and Stefan-Boltzmann law. Real blackbodies have opaque, closed surfaces that absorb radiation based on factors like temperature, emissivity, and surface area, and their radiation is used in applications like thermal imaging, optical sensors, and burglar alarms.
Blackbody Vs. graybody.pdf

It explains the difference between a blackbody and a gray body following Kirchhoff's law of thermal radiation.
MCQs of blackbody & Kirchhoff's law.pdf

It is a PowerPoint presentation on multiple choice questions and answers on Blackbody & Kirchhoff's law topics. It discusses all practical examples and numerical problems of the said concepts.
Planck quantum theory.pdf

This PowerPoint presentation explains Planck's quantum theory and Planck's constant topics briefly. It is now available on Instamojo for just Rs.10/-. Save a personalized copy of this presentation by visiting Instamojo. The link is below.
https://kameswariservices201831.myinstamojo.com/product/3439450/planck-quantum-theory-postulates
Kirchhoff's law (1).pdf

It is a PowerPoint presentation of Kirchhoff's law of thermal radiation. And it gives a brief idea of Kirchhoff's law, its applications, and examples. Moreover, it explains the terms absorptivity, emissive power, thermal equilibrium, and emissivity.
Kirchhoff's law.pdf

It discusses Kirchhoff's law and its examples. It explains blackbody emissions in thermal equilibrium conditions.
We are sorry to inform you about a small error in Kirchhoff's formula section of the above infographic. Kirchhoff's formula states that the absorptivity and emissivity of the body are equal in thermal equilibrium conditions. 'a' is the absorbing power of the body at a wavelength λ. And 'e' is the emissivity of the body at the same wavelength. So, we will write aλ=e but not aλ=eλ. We request you read it correctly while going through the PDF.
Kirchhoff's law formula and its derivation. Numerical problems.pdf

It discusses Kirchhoff's formula and its derivation. And it includes numerical problems of Kirchhoff's law. For a colorful e-book for only 10/- rupees on Kirchhoff's law, visit our store at;
https://kameswariservices201831.myinstamojo.com/product/3467221/kirchhoffs-law-of-thermal-radiations
Planck's constant.pdf

It describes the definition of Planck's constant. Planck constant helps compute the discrete energy changes of a body by relating to the frequency of the photon. Planck constant explains the proportionality relationship between the photon's energy and the frequency.
For more information on this topic, kindly visit our blog article at;
https://jayamchemistrylearners.blogspot.com/2022/08/plancks-constant-chemistry-learners.html
Numerical problems of Planck's quantum theory.pdf

It is a PowerPoint presentation on numerical problems of Planck quantum theory topic.
It includes 12 numerical problems with solved answers. Besides, this PowerPoint presentation has a mind map to remember all formulas of Planck quantum law.
MCQ on Planck constant.pdf

This document contains 10 multiple choice questions about Planck's constant. It provides explanations for why Planck's constant was introduced, its value in different units, and that it explains both the quantum and particle nature of light. The questions cover topics like the relationship between photon energy and frequency, experimental methods used to determine Planck's constant, and why LED lights are used in these calculations. Additional resources on Planck's constant and quantum theory are provided at the end.
Detailed light theories.pdf

It explains both wave & particle theories of light. It also includes Maxwell's mathematical equations and postulates of Planck quantum theory.
Theories of light.pdf

It explains the theories of light, including electromagnetic wave theory and Planck's quantum theory.
MCQ's on hydrogen spectrum.pdf

The document contains 10 multiple choice questions about the hydrogen spectrum. It discusses that the essential condition to observe the hydrogen spectrum is high temperature and low pressure. The hydrogen spectrum appears as distinct lines, with the prominent line being red with a wavelength of 656 nm. It is an example of a line spectrum and can be recorded using a spectroscope. Robert Bunsen and Gustav Kirchhoff discovered the hydrogen spectrum and spectroscope, while Isaac Newton first discovered the word "spectrum".
Rydberg equation.pdf

The Rydberg formula helps to determine the wavenumber or wavelengths of hydrogen spectral lines obtained in the hydrogen spectrum. Previously, Johann Jakob Balmer discovered an empirical formula to determine the wavelengths of hydrogen spectral lines obtained in the visible region of the hydrogen spectrum. As we all know, the hydrogen spectrum is not limited to the visible zone only. It occupies the ultraviolet and infrared parts of the electromagnetic spectrum also. Hence, the scientists' quests to determine the spectral positions of various spectral lines of the hydrogen spectrum finally came to an end with the Rydberg formula.
More from SaiKalyani11 (20)
Kirchhoff's law formula and its derivation. Numerical problems.pdf

Kirchhoff's law formula and its derivation. Numerical problems.pdf
Recently uploaded
Advanced Java[Extra Concepts, Not Difficult].docx![Advanced Java[Extra Concepts, Not Difficult].docx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Advanced Java[Extra Concepts, Not Difficult].docx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
This is part 2 of my Java Learning Journey. This contains Hashing, ArrayList, LinkedList, Date and Time Classes, Calendar Class and more.
CACJapan - GROUP Presentation 1- Wk 4.pdf

Macroeconomics- Movie Location
This will be used as part of your Personal Professional Portfolio once graded.
Objective:
Prepare a presentation or a paper using research, basic comparative analysis, data organization and application of economic information. You will make an informed assessment of an economic climate outside of the United States to accomplish an entertainment industry objective.
The History of Stoke Newington Street Names

Presented at the Stoke Newington Literary Festival on 9th June 2024
www.StokeNewingtonHistory.com
Azure Interview Questions and Answers PDF By ScholarHat

Azure Interview Questions and Answers PDF By ScholarHat
Digital Artifact 1 - 10VCD Environments Unit

Digital Artifact 1 - 10VCD Environments Unit - NGV Pavilion Concept Design
A Survey of Techniques for Maximizing LLM Performance.pptx

A Survey of Techniques for Maximizing LLM Performance
Top five deadliest dog breeds in America

Thinking of getting a dog? Be aware that breeds like Pit Bulls, Rottweilers, and German Shepherds can be loyal and dangerous. Proper training and socialization are crucial to preventing aggressive behaviors. Ensure safety by understanding their needs and always supervising interactions. Stay safe, and enjoy your furry friends!
RPMS TEMPLATE FOR SCHOOL YEAR 2023-2024 FOR TEACHER 1 TO TEACHER 3

RPMS Template 2023-2024 by: Irene S. Rueco
The simplified electron and muon model, Oscillating Spacetime: The Foundation...

Discover the Simplified Electron and Muon Model: A New Wave-Based Approach to Understanding Particles delves into a groundbreaking theory that presents electrons and muons as rotating soliton waves within oscillating spacetime. Geared towards students, researchers, and science buffs, this book breaks down complex ideas into simple explanations. It covers topics such as electron waves, temporal dynamics, and the implications of this model on particle physics. With clear illustrations and easy-to-follow explanations, readers will gain a new outlook on the universe's fundamental nature.
How to Add Chatter in the odoo 17 ERP Module

In Odoo, the chatter is like a chat tool that helps you work together on records. You can leave notes and track things, making it easier to talk with your team and partners. Inside chatter, all communication history, activity, and changes will be displayed.
Recently uploaded (20)
Liberal Approach to the Study of Indian Politics.pdf

Liberal Approach to the Study of Indian Politics.pdf
Azure Interview Questions and Answers PDF By ScholarHat

Azure Interview Questions and Answers PDF By ScholarHat
A Survey of Techniques for Maximizing LLM Performance.pptx

A Survey of Techniques for Maximizing LLM Performance.pptx
RPMS TEMPLATE FOR SCHOOL YEAR 2023-2024 FOR TEACHER 1 TO TEACHER 3

RPMS TEMPLATE FOR SCHOOL YEAR 2023-2024 FOR TEACHER 1 TO TEACHER 3
The simplified electron and muon model, Oscillating Spacetime: The Foundation...

The simplified electron and muon model, Oscillating Spacetime: The Foundation...
Pride Month Slides 2024 David Douglas School District

Pride Month Slides 2024 David Douglas School District
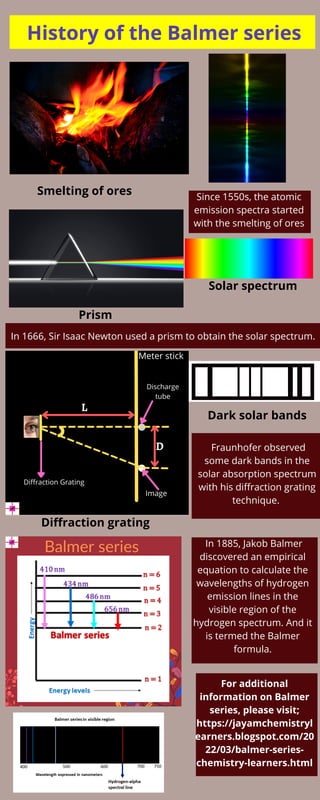
The history of the Balmer series
- 1. History of the Balmer series Smelting of ores Since 1550s, the atomic emission spectra started with the smelting of ores Prism Solar spectrum In 1666, Sir Isaac Newton used a prism to obtain the solar spectrum. Diffraction grating Dark solar bands Fraunhofer observed some dark bands in the solar absorption spectrum with his diffraction grating technique. In 1885, Jakob Balmer discovered an empirical equation to calculate the wavelengths of hydrogen emission lines in the visible region of the hydrogen spectrum. And it is termed the Balmer formula. For additional information on Balmer series, please visit; https://jayamchemistryl earners.blogspot.com/20 22/03/balmer-series- chemistry-learners.html