The Greenhouse Effect on Earth
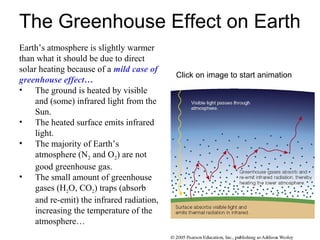
- 1. The Greenhouse Effect on Earth Earth’s atmosphere is slightly warmer than what it should be due to direct solar heating because of a mild case of greenhouse effect… • The ground is heated by visible and (some) infrared light from the Sun. • The heated surface emits infrared light. • The majority of Earth’s atmosphere (N2 and O2) are not good greenhouse gas. • The small amount of greenhouse gases (H2O, CO2) traps (absorb and re-emit) the infrared radiation, increasing the temperature of the atmosphere… Click on image to start animation
- 2. Water On Earth The condition is just right! • The combination of three factors: Distance to the Sun, the albedo, and the greenhouse effect, make it possible for water to stay on Earth. • N2 and O2 are not greenhouse gas. • Not much CO2 in the atmosphere. • Variable amount of H2O in the atmosphere…regulated by the temperature. The result is a mild greenhouse effect…not too hot, and not too cold, just the right temperature for most of the water to stay in liquid phase, and some to stay in gas phase in the atmosphere on the surface
- 3. Greenhouse Gases • The primary components of Earth’s atmosphere, N2 and O2 do not have absorption in the IR wavelength range, therefore, do not have a significant role in setting the surface temperature of the planet… • Greenhouse gas are efficient in absorbing IR light… The most important greenhouse gases are: – H2O – Water vapor. – CO2 – Carbon Dioxide – CH4 – methane The most abundant greenhouse gas in Earth’s atmosphere is water vapor. Most of the greenhouse heating of Earth’s atmosphere is due to Water vapor absorption of IR radiation emitted by Earth, and then transferring the energy to the surrounding air molecule
- 4. Source of Water The terrestrial planets were built from rock and planetesimals. No gases or water can condense at the high temperature near the Sun. So, where did the water on Earth come from? • The water on Earth (and other terrestrial worlds) most likely was brought over by the comets during the period of heavy bombardment about 4 billion years ago… • These water (and other gases) were trapped in the interior, and released by volcanic activities…by Outgassing • Mt. St Helen eruption, 2004!
- 5. The Atmosphere of Earth The atmosphere of Earth contains primarily N2 (77%) and O2 (21%). • What happened to all the CO2? • Where did all the O2 come from?
- 6. CO2 CO2 is a colorless gas… • condenses into solid form (dry ice) at -78°C in atmospheric pressure. • condenses into liquid at -57°C at pressure above 5.1 atmospheric pressure. Atmospheric CO2 is derived from (The sources…) • Volcanic outgassing • burning of organic matter • Respiration of living organisms • … CO2 can be stored in (The Sinks…) • Highly soluble in water: forms H2CO3 • Dissolved CO2 in water can interact with silicate minerals to form carbonated minerals… • …
- 7. Carbon Dioxide Cycle The mechanism by which Earth self-regulates its temperature is called the carbon dioxide cycle, or the CO2 cycle for short. Starting with the carbon dioxide in the atmosphere: • Volcanoes outgas CO2 into the atmosphere. • Atmospheric carbon dioxide dissolves in the oceans. • At the same time, rainfall erodes rocks on Earth’s continents and rivers carry the eroded minerals to the oceans. • In the oceans, the eroded minerals combine with dissolved carbon dioxide and fall to the ocean floor, making carbonate rocks such as limestone. • Over millions of years, the conveyor belt of plate tectonics carries the carbonate rocks to subduction zones, and subduction carries them down into the mantle. • As they are pushed deeper into the mantle, some of the subducted carbonate rock melts and releases its carbon dioxide, which then outgasses back into the atmosphere through volcanoes.
- 8. The CO2 Cycle The CO2 cycle acts as a thermostat that regulates the temperature of the Earth… If Earth warms up a bit, then • carbonate minerals form in the oceans at a higher rate. • The rate at which the oceans dissolve CO2 gas increases, pulling CO2 out of the atmosphere. • The reduced atmospheric CO2 concentration leads to a weakened greenhouse effect that counteracts the initial warming and cools the planet back down. If Earth cools a bit, • carbonate minerals form more slowly in the oceans. • The rate at which the oceans dissolve CO2 gas decreases, allowing the CO2 released by volcanism to build back up in the atmosphere. • The increased CO2 concentration strengthens the
- 9. Feedback Loop • Positive Feedback – Mechanisms that make things worse… – e.g., Increasing CO2 in the atmosphere leading to the release of more CO2 • Negative Feedback – Mechanisms that are self-correcting… – e.g., Increasing CO2 in the atmosphere leading to higher rate of CO2 removal, such as our CO2 cycle.
- 10. Plate Tectonics Plate tectonics plays an important role in the CO2 cycle in that it helps to carry the carbonate rocks into the mantle, which are then released again by volcanic activities. – Earth’s lithosphere is broken into pieces (the plates). – These plates float on top of the mantle, interacting with each other to produce the geological features we see and feel today. Click on image to start animation
- 11. Where Did O2 Come From? The most important source of O2 on Earth is Life and Photosynthesis. • • • • Photosynthesis converts CO2 to O2, and incorporates carbon into amino acids, proteins, and other components of living organisms. O2 will be depleted from the atmosphere very rapidly without a source. O2 is a very reactive chemical that likes to be combined with other elements through oxidation. For examples, CO2, H2O, FeO (rust) ⇒ That’s how we make fire! O2 Absorbs UV, which also transform some of the O2 into O3, which absorbs even more UV ⇒ O2 not only supports life, it also protect life! UV light can break the water molecules to release oxygen, but the contribution is small….
- 12. The Role of the Magnetic Field of Earth Another important characteristics of the Earth is its magnetic fields, which shield us from the bombardment of the high-energy charged particles, mostly from the Sun. • Without magnetic field, the high energy particles of solar wind can strip much of the Earth’s atmosphere by breaking the bounds between the atoms in the air molecules – N2 → N + N – O2 → O + O – H2 O → H + H + O • The lighter gases then have higher probability of acquiring velocity higher than escape velocity and escape from Earth!
- 13. Water On Earth in the Past Was it always like this on Earth? • Yes. Water was plentiful throughout most of Earth’s history, for about three billion years. • No! Geological evidences suggest that Earth used to be covered by ice about 600-700 million years ago ⇒ Snowball Phase. How did Earth recover from the snowball phase? • Once the water was frozen, CO2 can no longer be removed from the atmosphere by dissolving in water ⇒ interruption of the CO2 cycle. • Increased CO2 level in the atmosphere leads to stronger greenhouse effect, which warms the atmosphere. • Higher temperature melt the ice ⇒ restoration of the CO2 cycle.