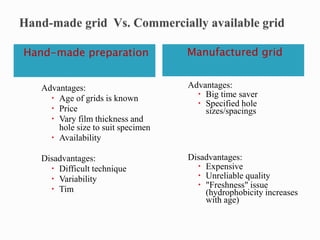
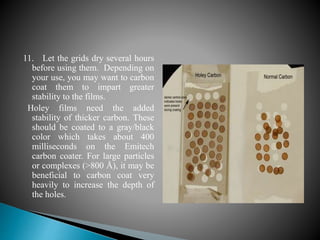
The document provides a comprehensive guide on preparing electron microscopy (EM) grids, detailing the various types of grids, their materials, methods for preparation, and the importance of cleaning protocols. It outlines the characteristics and applications of different support films, including formvar and pioloform, and specific techniques for coating grids to ensure high-quality specimens for transmission electron microscopy (TEM). Additionally, it highlights the precautions needed during preparation and chemical handling, emphasizing the critical role of films and grids in the preservation and observation of specimens under electron beams.