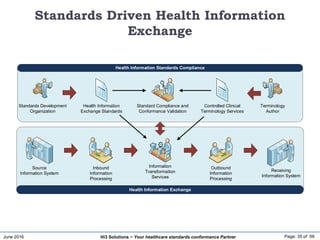
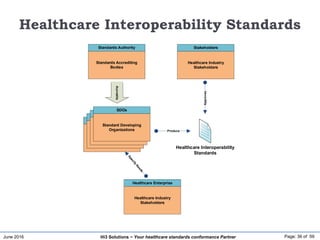
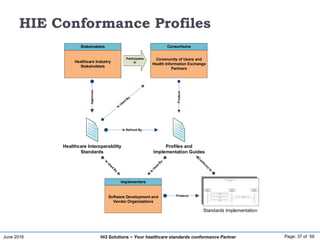
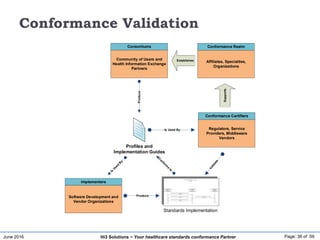
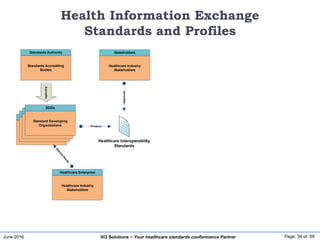
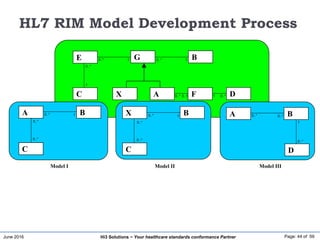
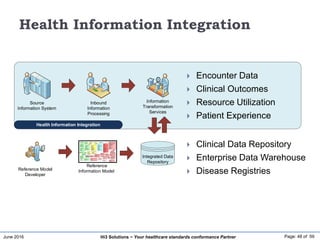
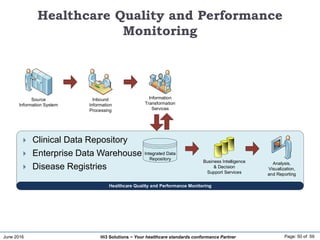
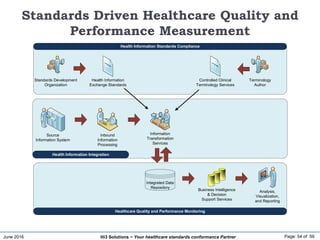
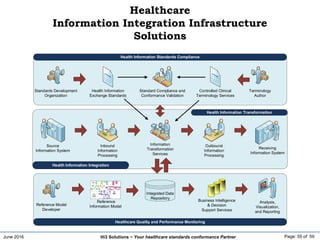
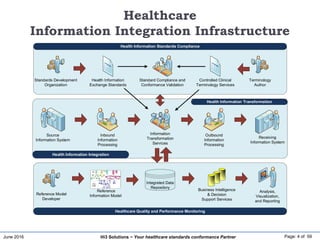
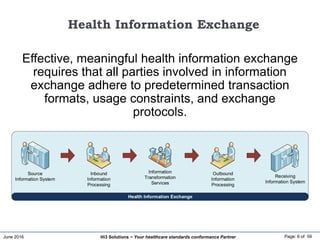
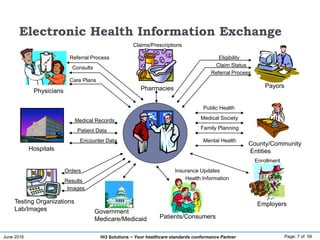
The document discusses the importance of healthcare information exchange, integration, and analytics for providing high-quality healthcare, emphasizing the need for standard compliance and technical infrastructure. It outlines various data exchange standards and clinical terminologies, as well as the role of standard development organizations in the healthcare sector. Additionally, it covers healthcare interoperability, conformance profiles, and compliance validation to improve the efficiency of data interchange among healthcare systems.









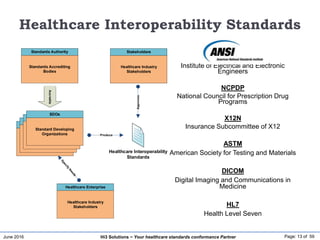


![June 2016 Page: 16 of 59Hi3 Solutions ~ Your healthcare standards conformance Partner
www.regenstrief.org/loinc [GO]
LOINC](https://image.slidesharecdn.com/hietechnicalinfrastructure-160627190103/85/Standards-Driven-Healthcare-Information-Integration-Infrastructure-16-320.jpg)
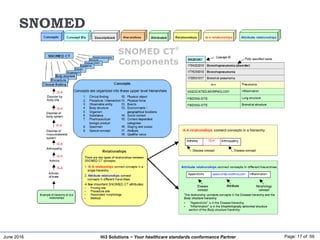


![June 2016 Page: 22 of 59Hi3 Solutions ~ Your healthcare standards conformance Partner
Conformance Profiles are an effective means of documenting our assumptions
about message structures
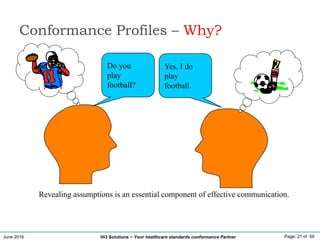
Conformance Profiles – Why?
Do you
use
HL7?
MSH
EVN
PID
[PD1]
[ { NK1 } ]
Yes, I
use
HL7.
MSH
EVN
PID
[ NK1 ]
OBX](https://image.slidesharecdn.com/hietechnicalinfrastructure-160627190103/85/Standards-Driven-Healthcare-Information-Integration-Infrastructure-22-320.jpg)

![June 2016 Page: 24 of 59Hi3 Solutions ~ Your healthcare standards conformance Partner
Conformance Profiles – What?
Use Case Model
Use Cases
Use Case Actors
Exchange Scenarios
Triggering Events
Receiver Responsibilities
Content Profiles
Element Usage and Cardinality
Coded Element Terminology Bindings
ud Use Case Model
Local Registry User
1.0 Immunization
History Query
2.0 Patient
Demographic
Update
3.0 Vaccine
Record Update Prov ider Organization
SIIS Registry
Administration
4.0 Immunization
Statistical Analysis
Trusted Third PartiesLocal Registry
Administration
SIIS Analysis Report
SIIS Analysis Report
SIIS Analysis Report SIIS Analysis Report
Update Confirmation
Update Confirmation
Query Response
Vaccine Record Update
Patient Information Update
Immunization History Request
sd Interactions
Requesting Registry
System
SIIS SIP Immunization
Information Exchange
System
Responding Registry
System
Vaccination Record Query (VXQ)
[Invalid VXQ Message]: General Acknowledgement (ACK)
[Valid VXR Message]: Vaccination Record Query (VXQ)
[No Matching Record]: Query Acknowledgement (QCK)
[Invalid QCK Message]: General Acknowledgement (ACK)
[Valid QCK Message]: Query Acknowledgement (QCK)
[Multiple Matching Records]: Vaccination Query Response (VXX)
[Invalid VXX Message]: General Acknowledgement (ACK)
[Valid VXX Message]: Vaccination Query Response (VXX)
[Single Matching Record]: Vaccination Query Response (VXR)
[Invalid VXR Message]: General Acknowledgement (ACK)
[Valid VXR Message]: Vaccination Query Response (VXR)](https://image.slidesharecdn.com/hietechnicalinfrastructure-160627190103/85/Standards-Driven-Healthcare-Information-Integration-Infrastructure-24-320.jpg)