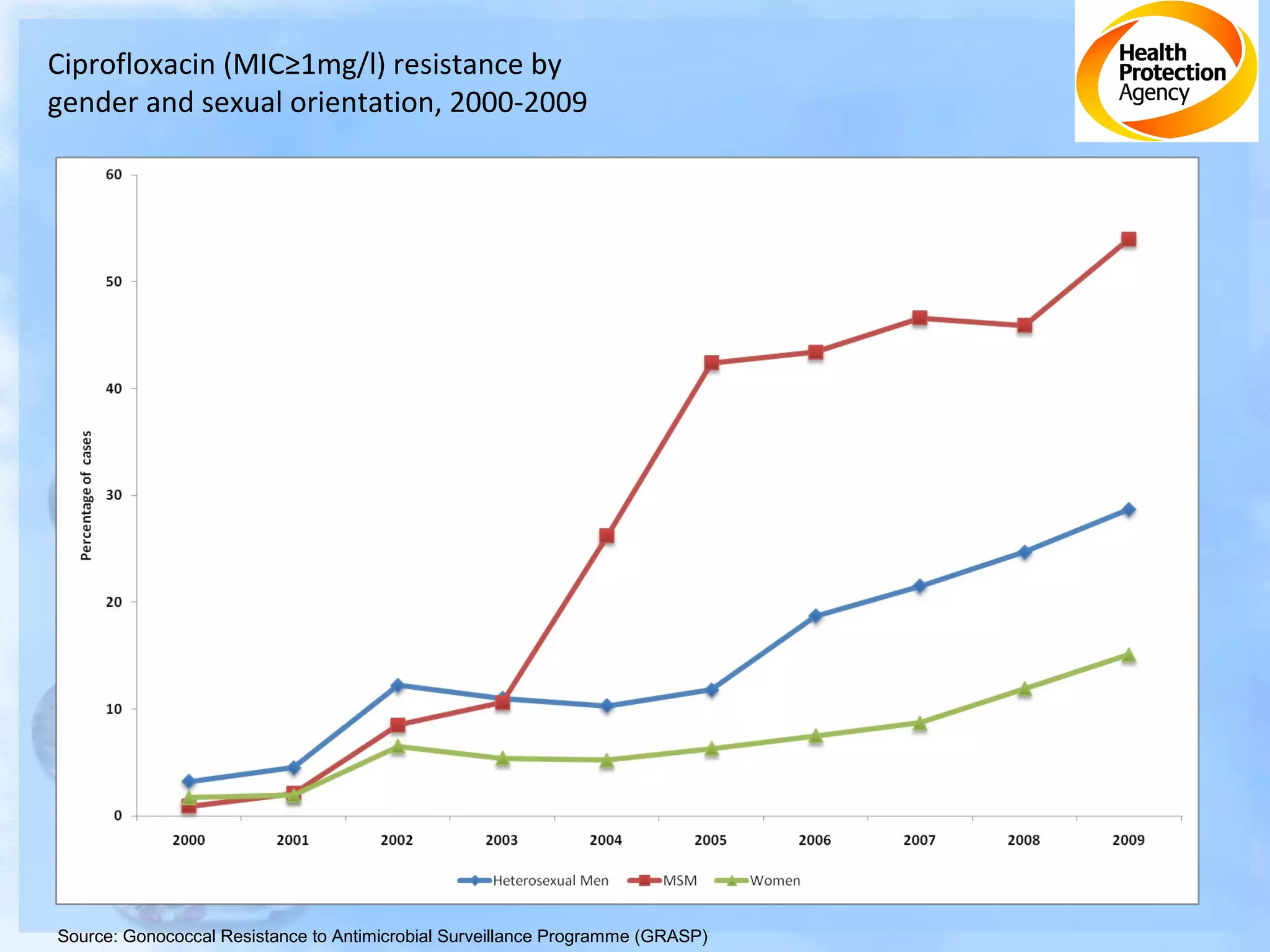
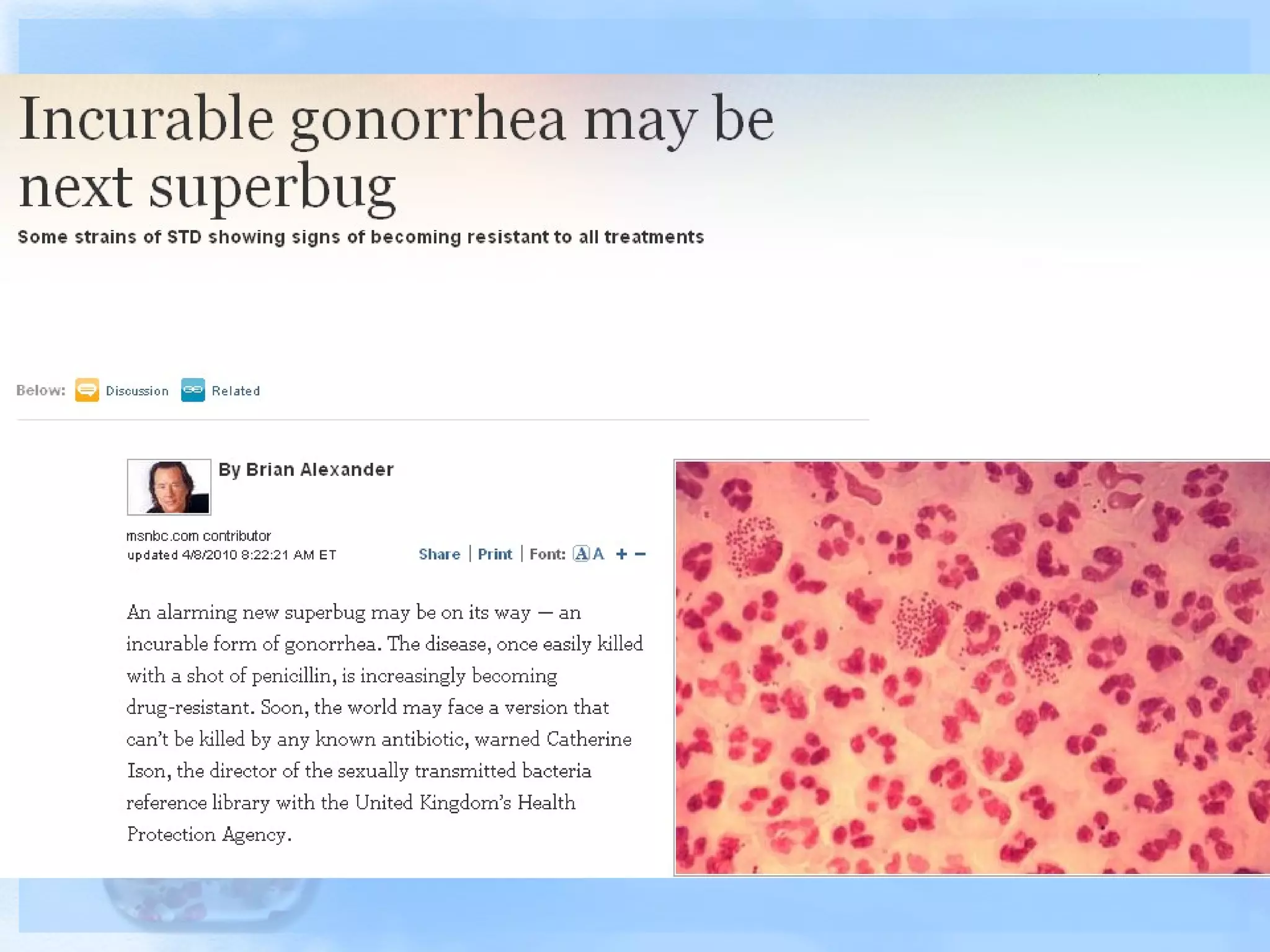
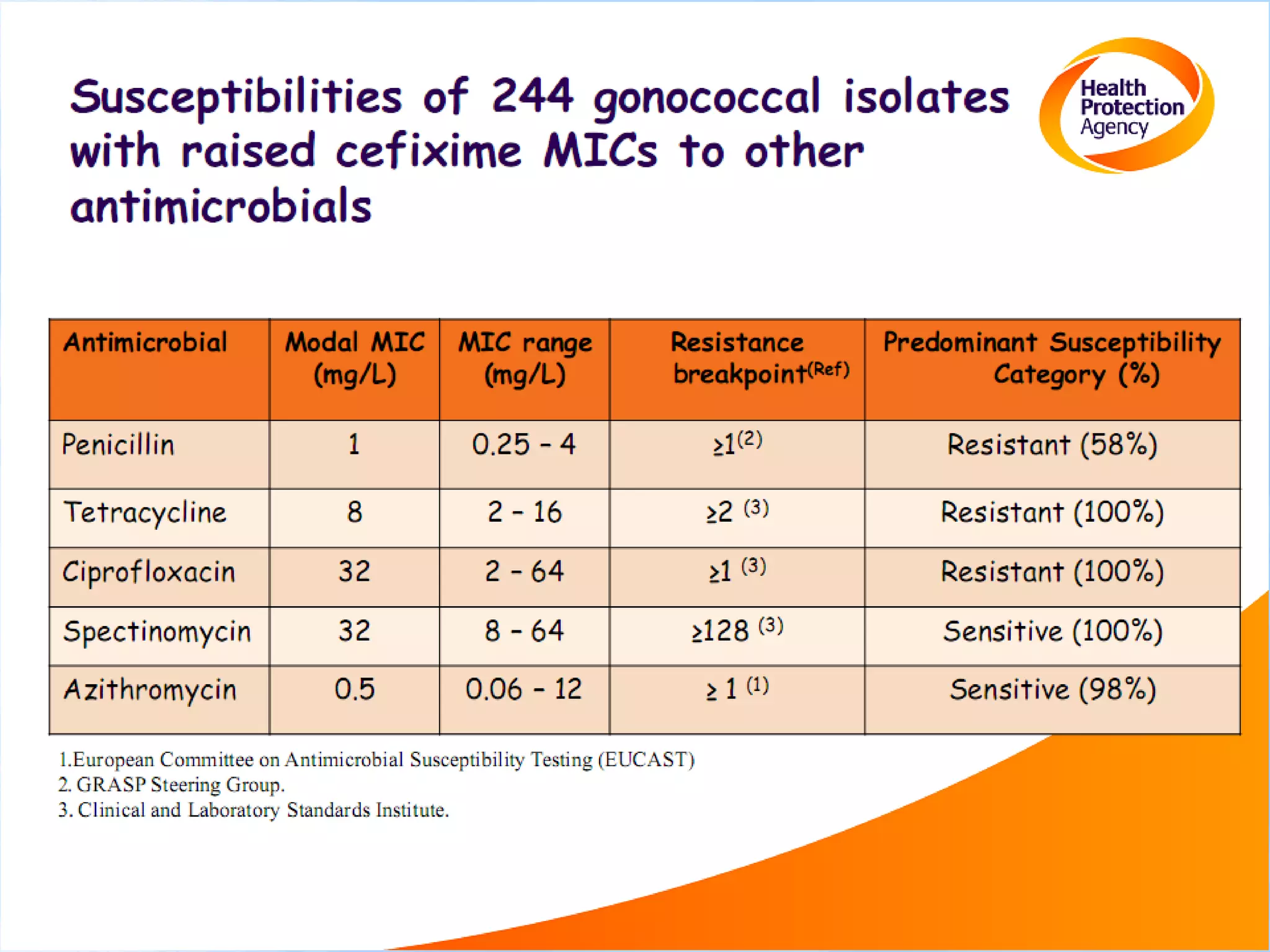
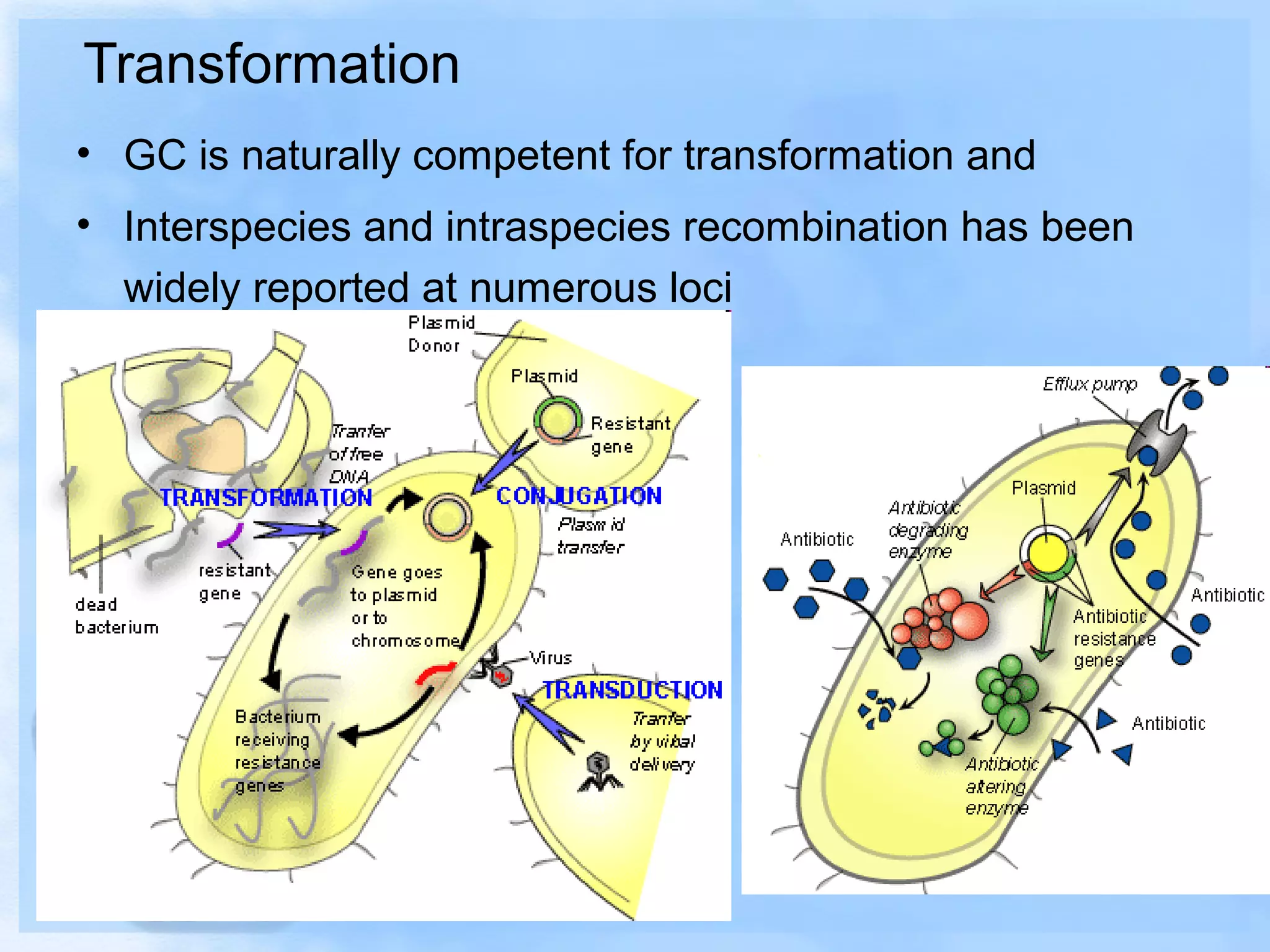
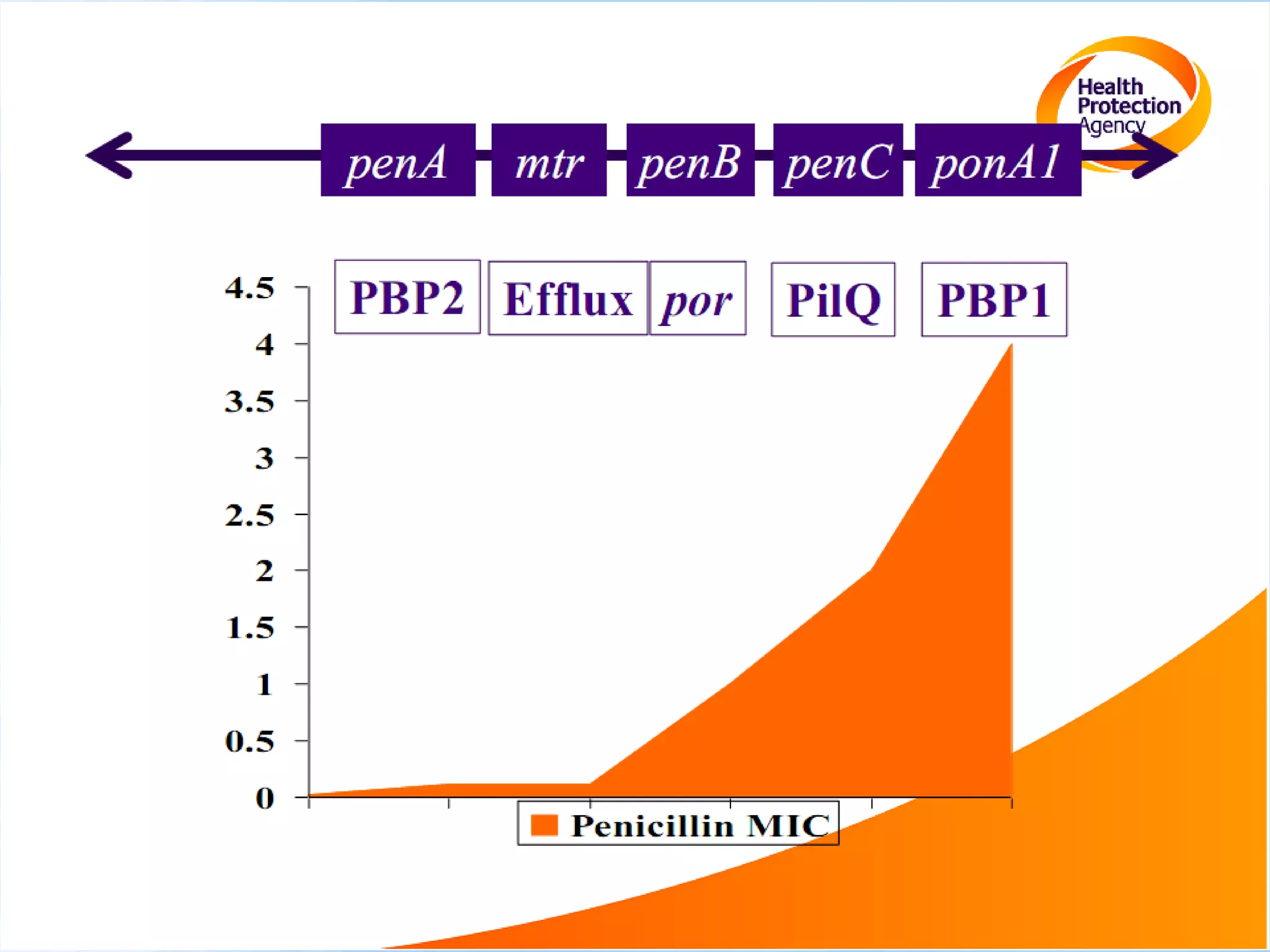
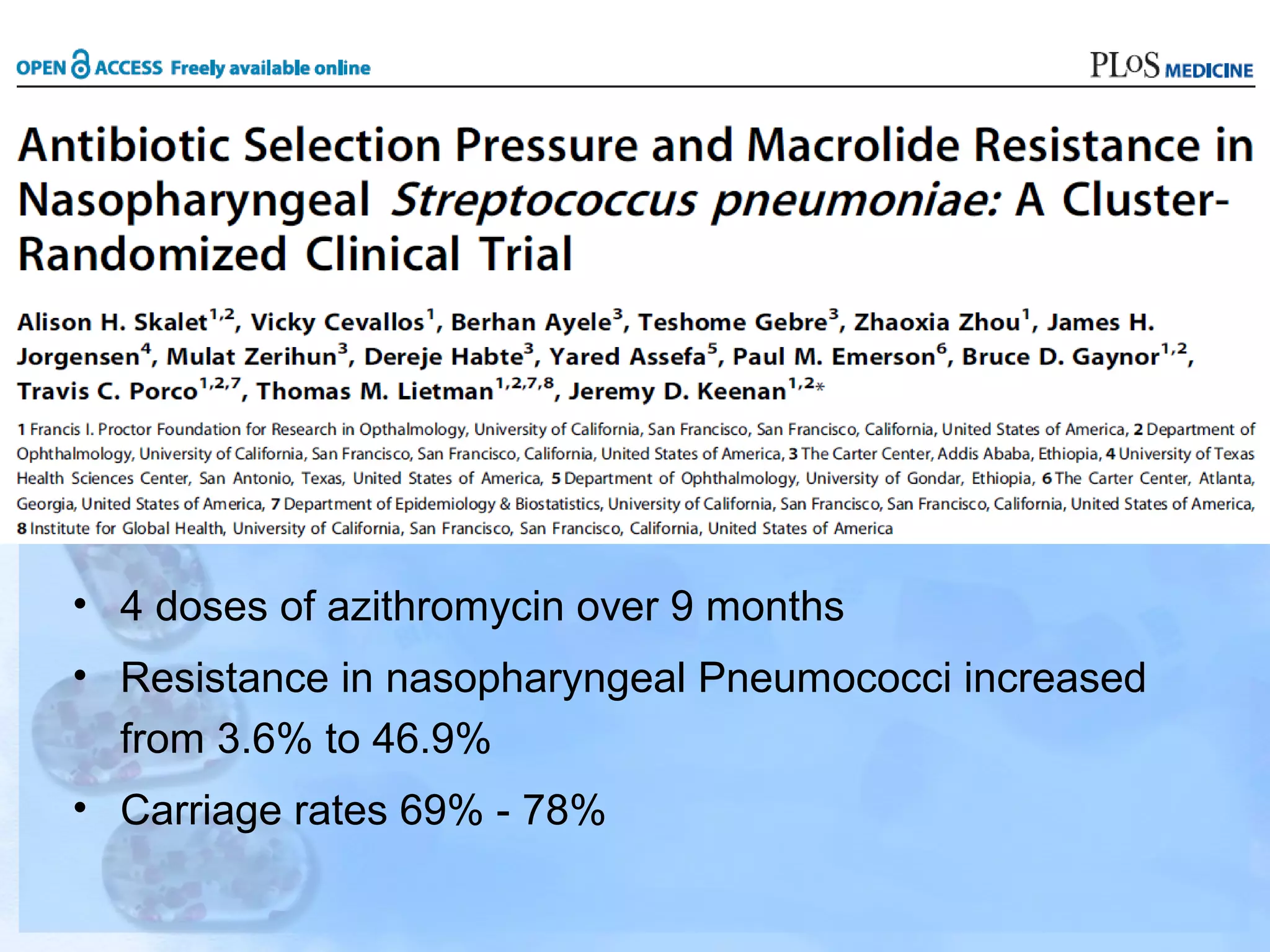
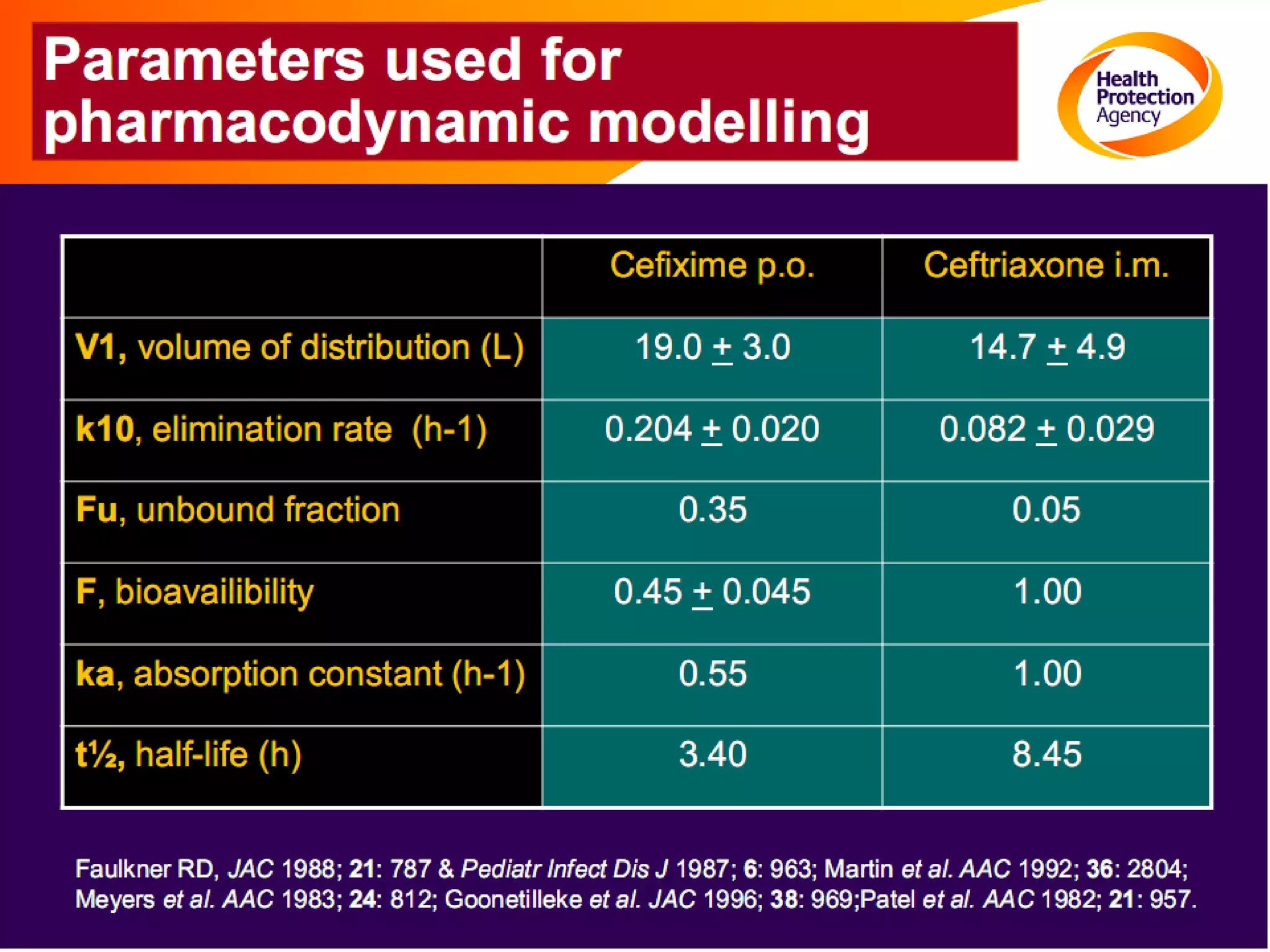
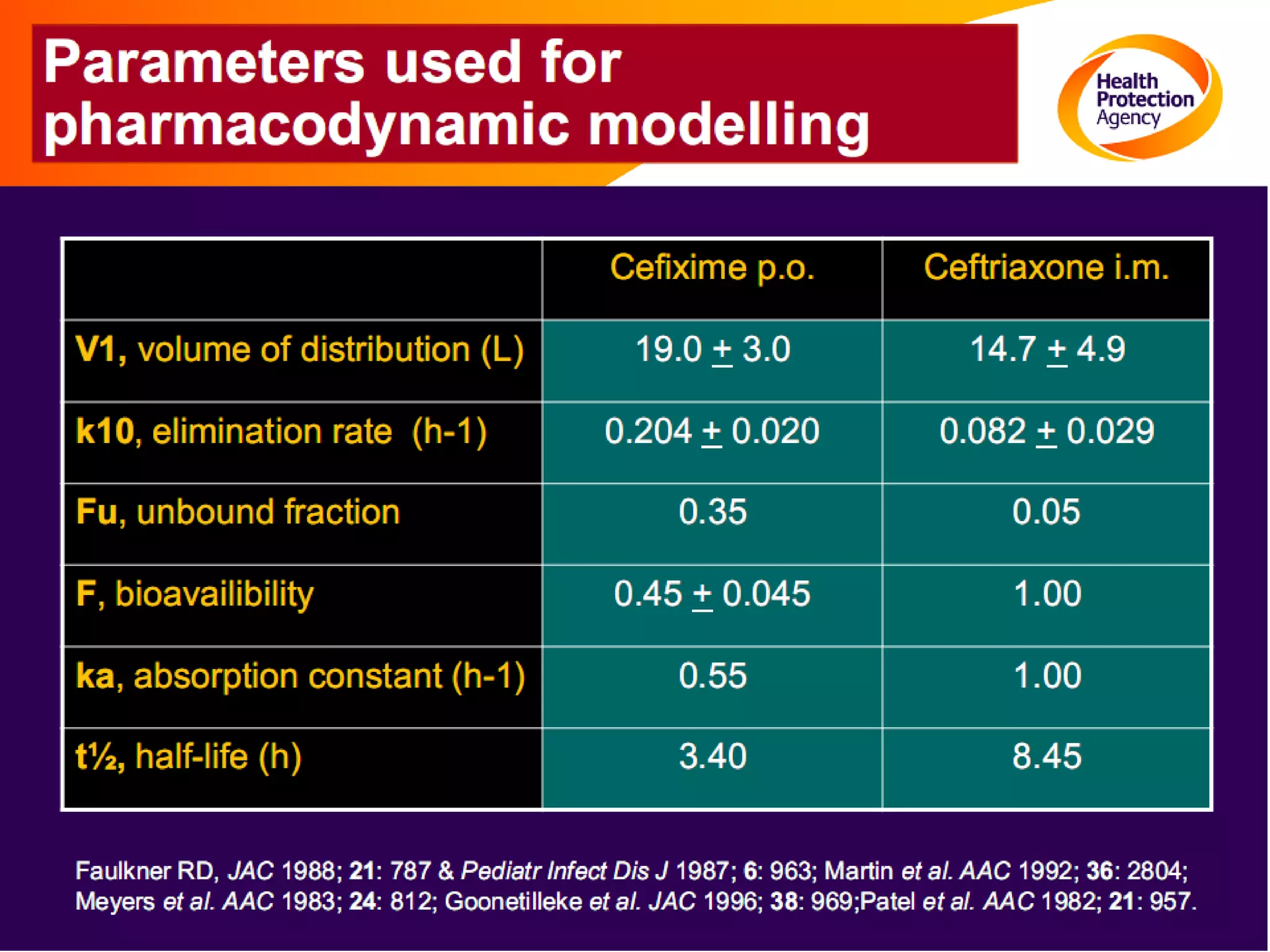
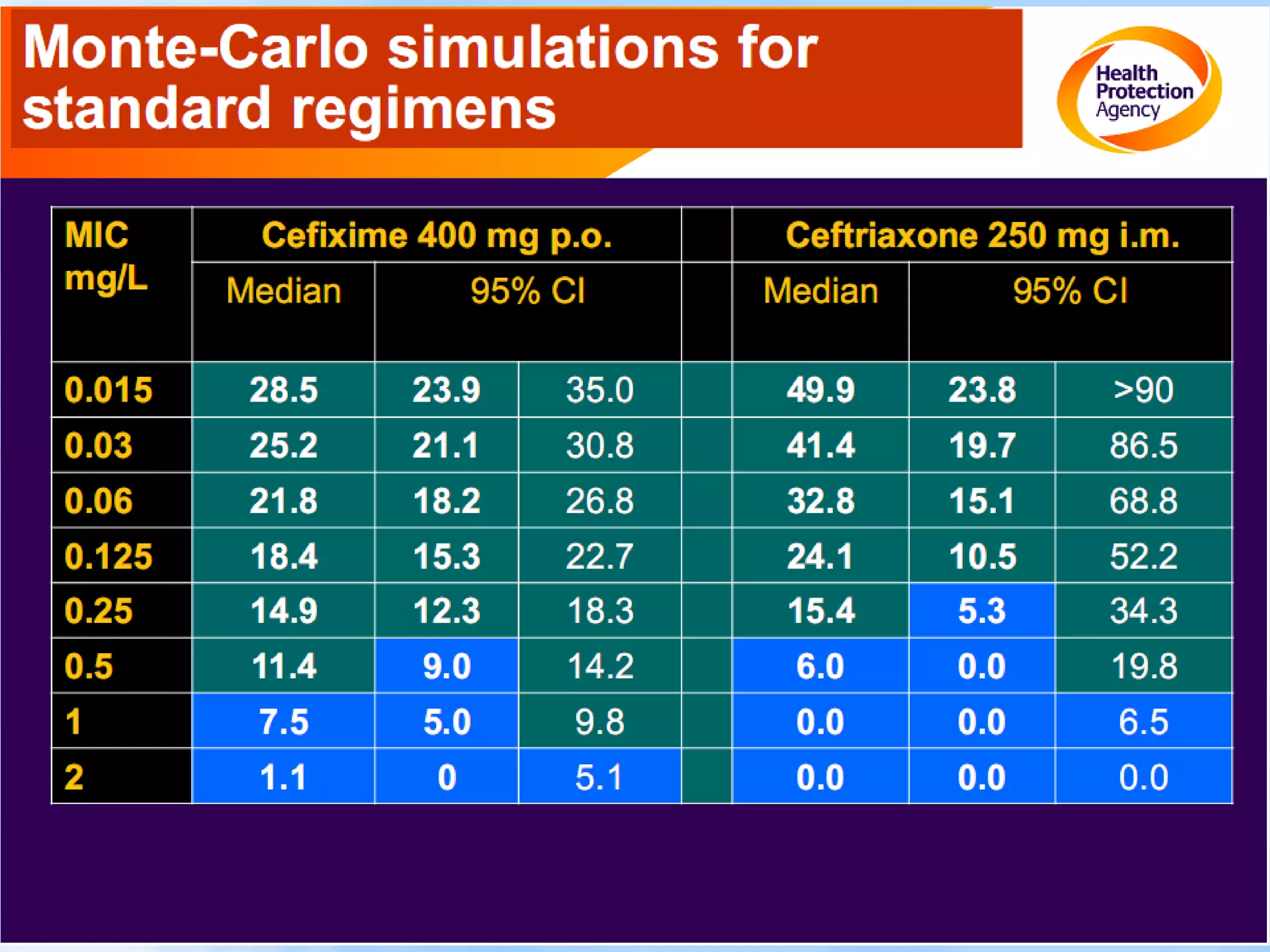
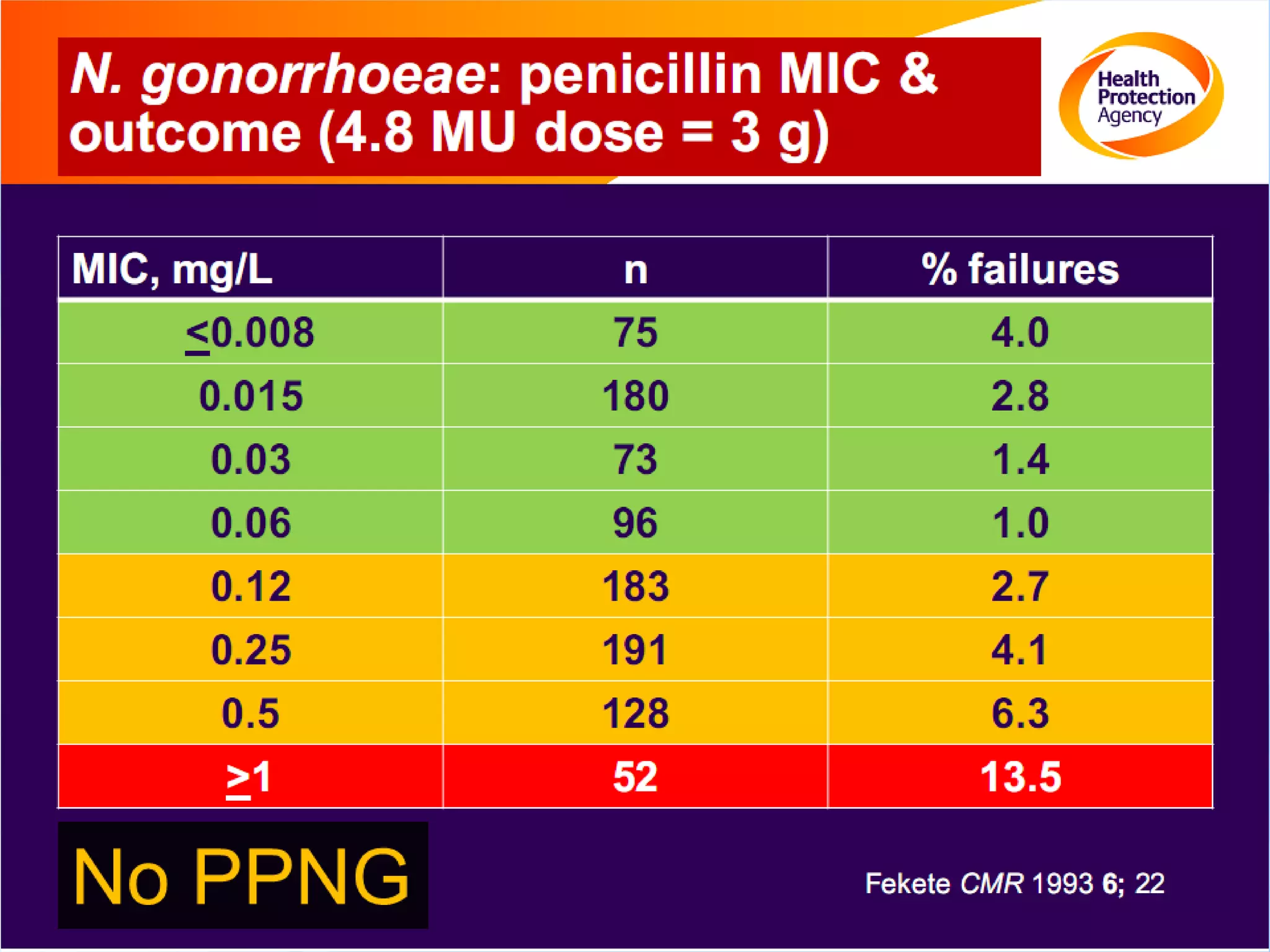
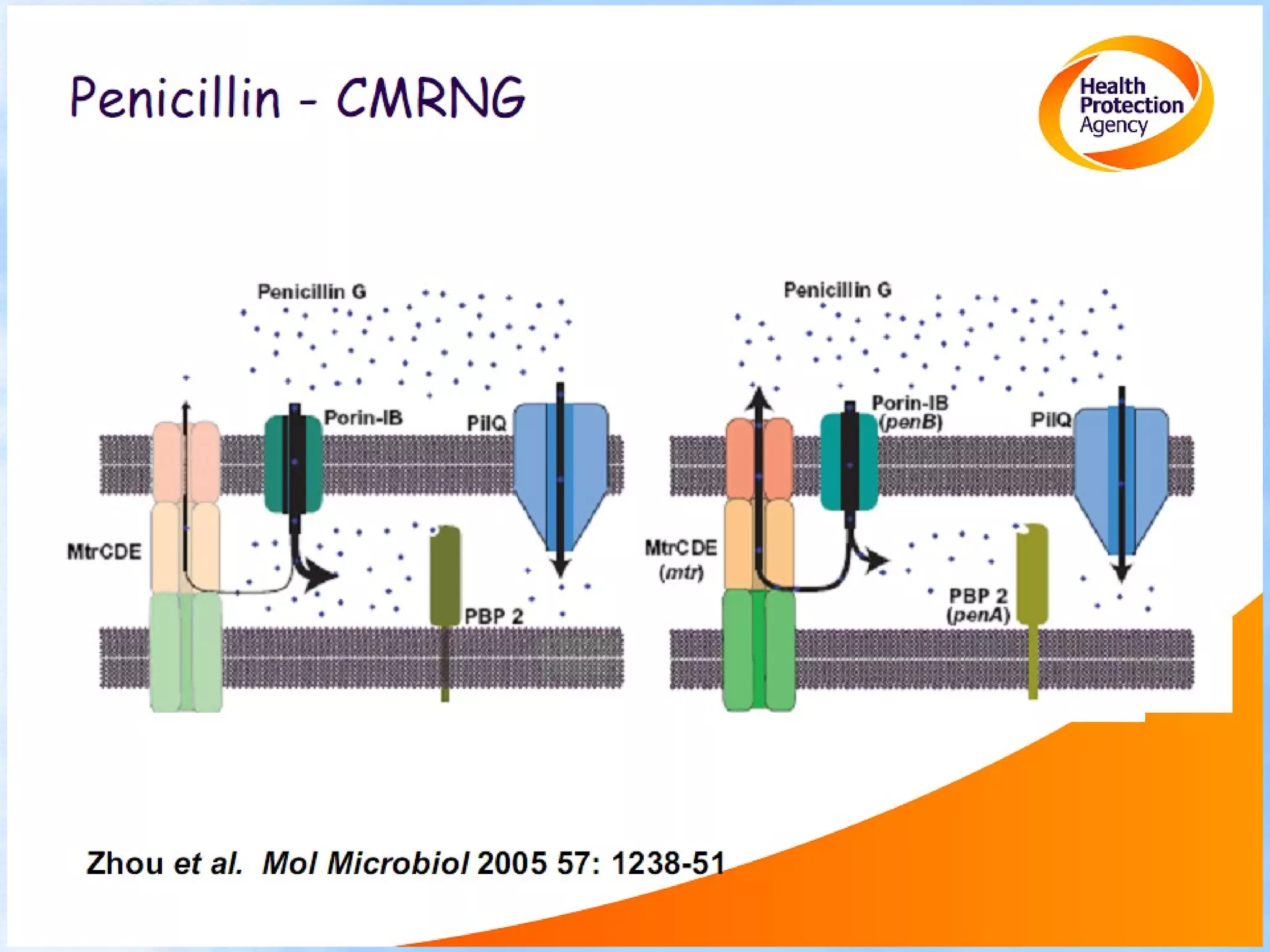
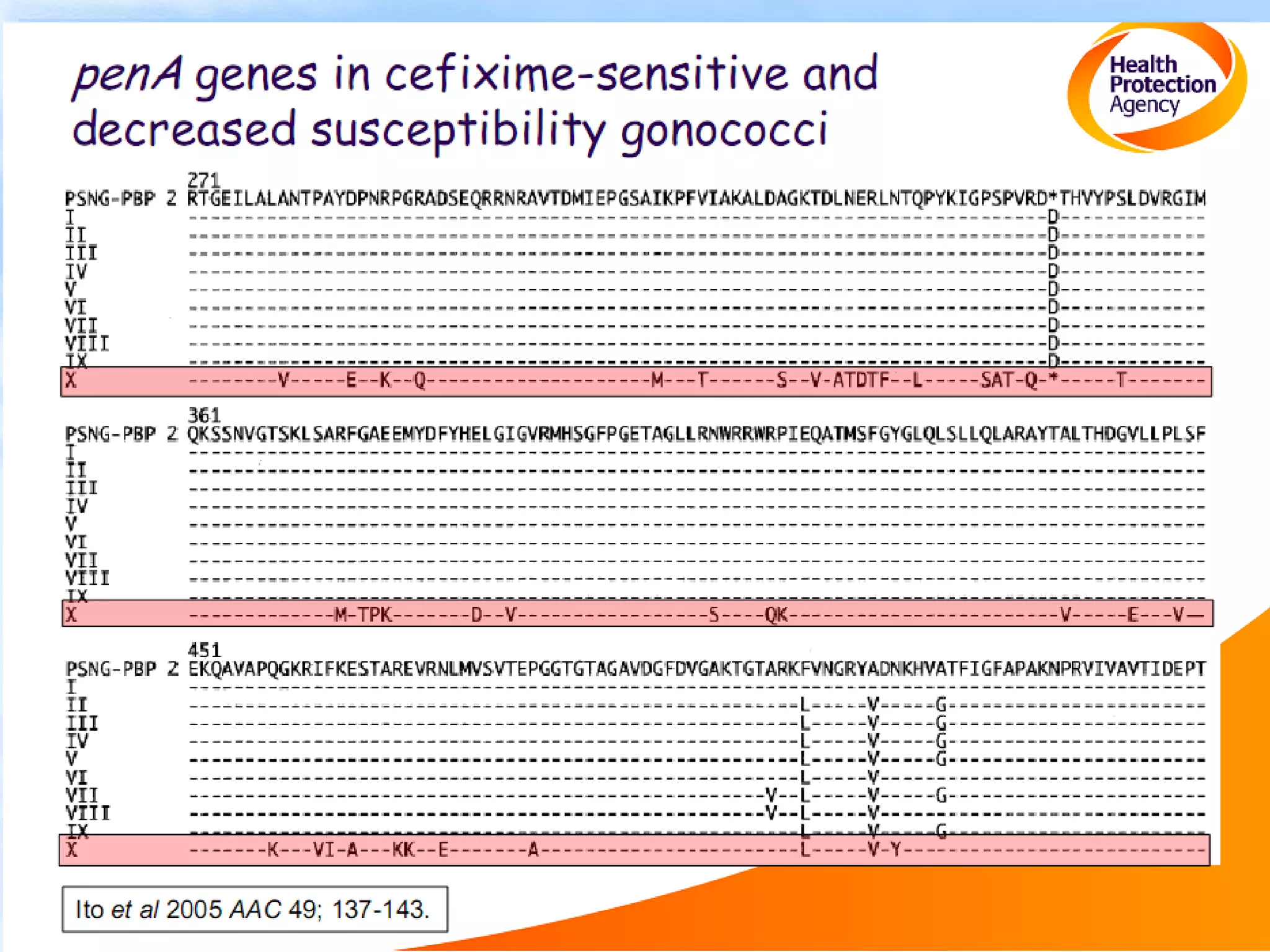
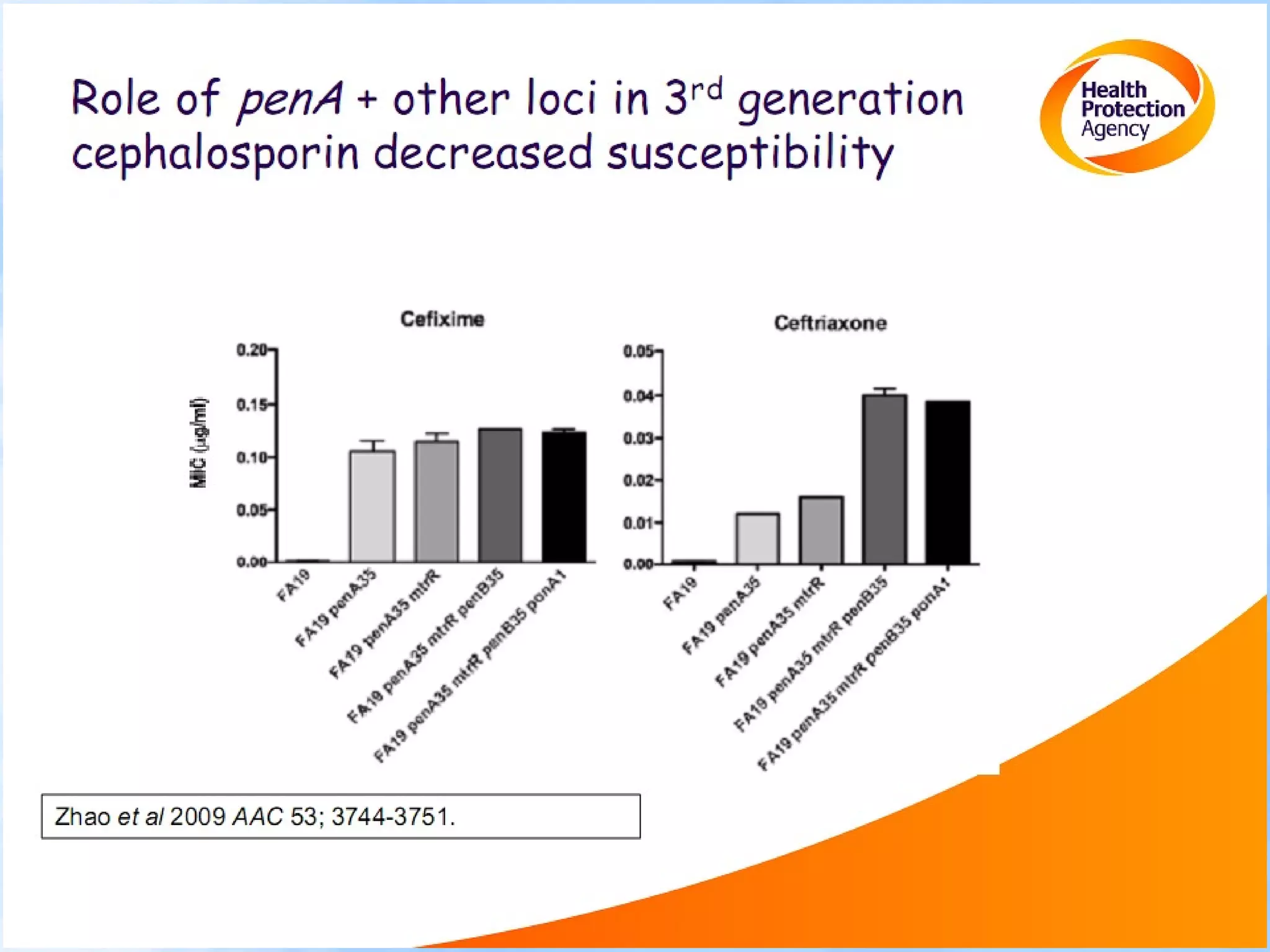
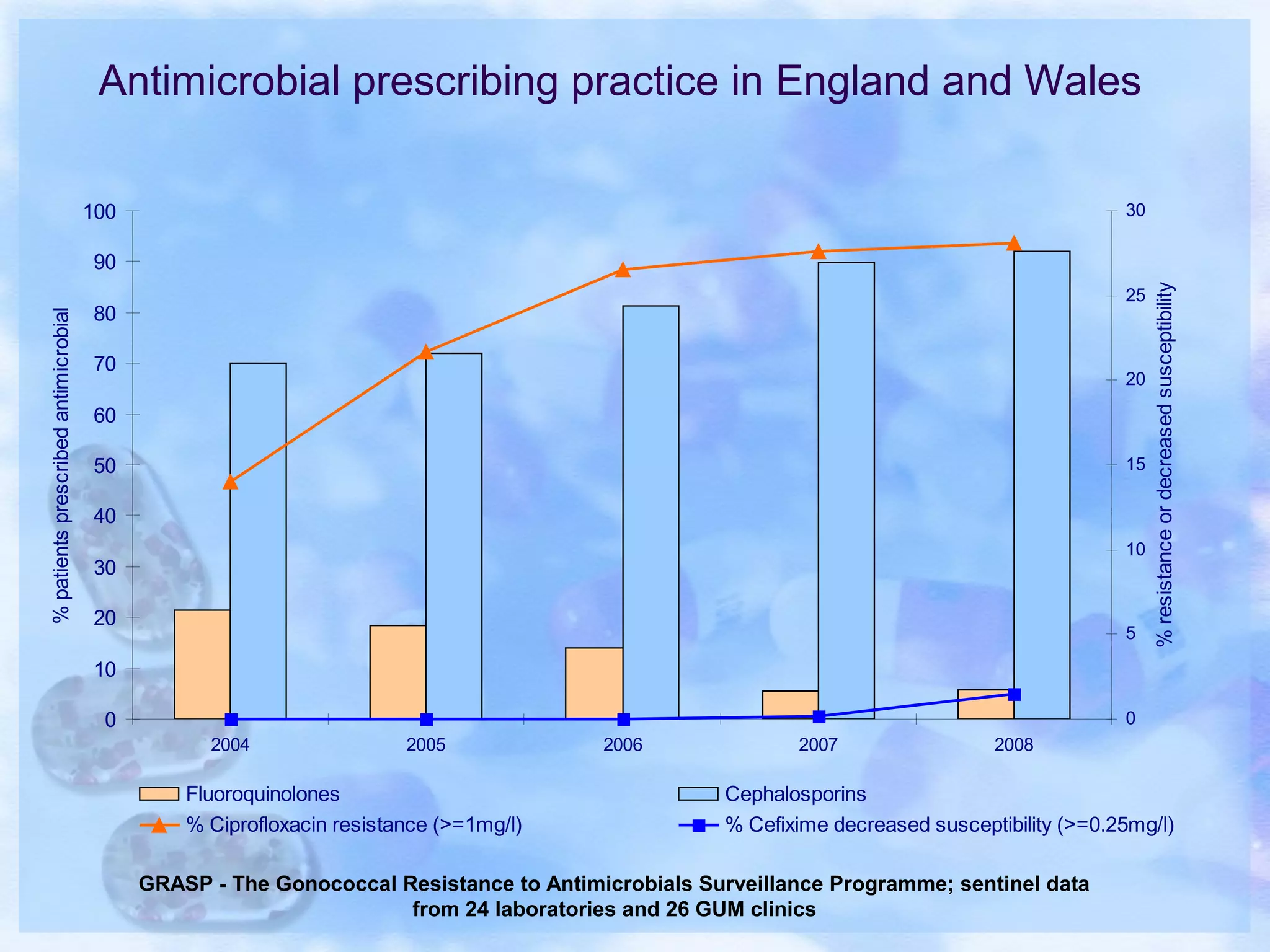
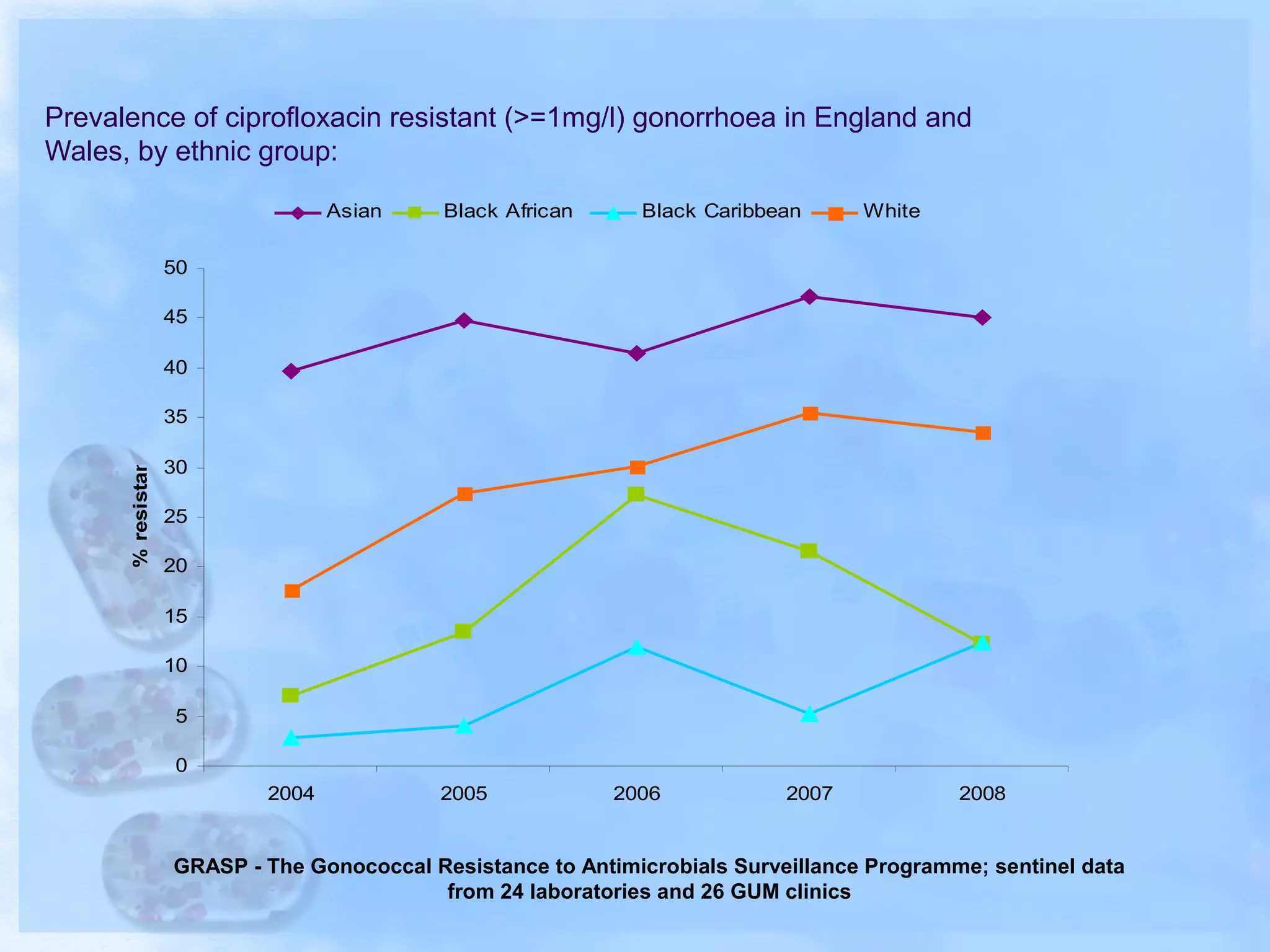
Antimicrobial resistance has been an ongoing issue since the discovery of early antimicrobial treatments. Resistance first emerged in the early 1900s in Neisseria gonorrhoeae and has since developed to nearly all classes of antimicrobials used to treat it. Resistance is now widespread globally to previously effective drugs. New treatment guidelines must consider emerging resistance patterns and combine antimicrobials to preserve effectiveness. Ongoing surveillance is also needed to monitor resistance trends and ensure optimal treatment strategies.