1. The document discusses various terms used to express the concentration of solutions such as percentage, molarity, molality, normality, and mole fraction. It provides examples and formulas to calculate these quantities.
2. Several numerical problems are given related to calculating concentration based on the mass or volume of components in a solution. This includes problems determining the percentage or mole fraction composition of mixed solutions.
3. Additional "HOTS" or higher-order thinking skills problems are presented involving multiple steps to determine volumes or densities based on given molarities, molalities, or percentages of solutions.





![Concentration of a solution:
(iii) Molarity (M) : It is the number of moles of solute present in one litre of solution. Its unit is mol/L. It is temperature
dependent. Molarity ( in mol/L) =
𝐌𝐨𝐥𝐞𝐬 𝐨𝐟 𝐬𝐨𝐥𝐮𝐭𝐞
𝐕𝐨𝐥𝐮𝐦𝐞 𝐨𝐟 𝐬𝐨𝐥𝐮𝐭𝐢𝐨𝐧 𝐢𝐧 𝐥𝐢𝐭𝐫𝐞
or, Msoln =
𝐧𝐬𝐨𝐥𝐮𝐭𝐞
𝐕𝐋
Example:
Molarity of NaOH solution is 1 M means 1 L of NaOH solution contains 1 mole = 40 g of NaOH crystals.
[Molarity of a solution is the number of milli moles of solute present in one milli litre of solution.]
(iv) Molality (m) : It is the number of moles of solute present in one kg ( or 1000g) of solvent. Its unit is mol/kg. It is
temperature independent. Molality (mol/kg) =
𝐌𝐨𝐥𝐞𝐬 𝐨𝐟 𝐬𝐨𝐥𝐮𝐭𝐞
𝐌𝐚𝐬𝐬 𝐨𝐟 𝐬𝐨𝐥𝐯𝐞𝐧𝐭 𝐢𝐧 𝐤𝐠
Example: Molality of ethyl alcohol solution is 1 m means 1 kg or 1000 g of water of 1 m ethyl alcohol solution contains 1 mole
or 46 g of ethyl alcohol.
**(v) Formality (F): It is the number of formula weights of the solute(ionic salt) present per litre of the solution. It is used in
case of ionic solid as solute in solution. We take the formula mass of an ionic solid crystal.
Example: Formality of NaCl solution is 1 F means 1L of NaCl solution contains 1 mole = 58.5 g of NaCl crystals.](https://image.slidesharecdn.com/solutionsclassxii-210623163445-230322025235-297921b1/85/solutionsclassxii-pdf-6-320.jpg)



![Example: What are the mole fractions of the components of the solution formed when 92 g glycerol (92 g/mol) is
mixed with 90 g water (18 g/mol)?
Components Step 1 Step 2 Step 3
Glycerol (A) Convert mass into moles
Mole =
mass in g
molar mass
= 92/92 = 1
mol
Total moles = 1+5 = 6
moles
(χA) =
𝐧𝐀
𝐧𝐀+𝐧𝐁
=
𝟏
𝟔
(Ans)
Water (B) Mole = 90 /18 = 5 mol (χB) =
𝐧𝐁
𝐧𝐀+𝐧𝐁
=
𝟓
𝟔
(Ans)
NUMERICAL PROBLEMS to Practices:
(1) Calculate the % composition in terms of mass of a solution obtained by mixing 300g of a 25% & 400 g of a 40%
solution by mass. [Ans: 33.57%, 66.43%].
(2) What volume of 95% by mass of sulphuric acid (density=1.85 g/mL ) & what mass of water must be taken to
prepare 100 mL of 15% by mass of sulphuric acid solution (density=1.10 g/mL) [Ans: 9.4mL, 92.6 g].
(3) Calculate the mole fraction of ethanol & water in a sample of rectified spirit which contains 95% ethanol by mass.
(At. Wt. C=12, H =1, O=16) [ Ans. ethanol = 0.88, water = 0.12]
(4) What volume of 10% (w/v) solution of Na2CO3 will be required to neutralise 100 mL of HCl solution containing 3.65
g of HCl. (At. wt. Na=23 , Cl = 35.5, H = 1) [Ans. 53.19 mL]
(5) Calculate the mole fraction of water in a mixture of 12 g water, 108 g acetic acid & 92 g of ethanol. [Ans.0.15]](https://image.slidesharecdn.com/solutionsclassxii-210623163445-230322025235-297921b1/85/solutionsclassxii-pdf-10-320.jpg)
![NUMERICAL PROBLEMS to Practices:
(6) The molality of a solution of ethanol in water is 1.55 m. How many grams of ethanol are dissolved in 2 kg of water?
[Ans. 142.6 g]
(7) Battery acid is 4.27 M H2SO4 aqueous solution of sulphuric acid of density 1.25 g/mL. Find its molality. [Ans 5.135 m]
HOTS
(8) How many mL of sulphuric acid of density 1.84 g/mL containing 95.6 mass % of H2SO4 should be added to one litre of
40 mass % solution of H2SO4 of density 1.31 g/mL in order to prepare 50 mass % solution of sulphuric acid of density 1.40
g/mL. [Ans : 166.2 mL of 95.6 mass%]
(9) Calculate the density of the sulphuric acid solution whose molarity is 10.8 M & molality is 92.6 m. [Ans: 1.17 g/mL]
(10)How many mL of a 0.1M HCl are required to react completely with 1 g mixture of Na2CO3 and NaHCO3 containing
equimolar amount of both? [Ans. 157.8 ml]
(11) When 10.0 mL of ethanol of density 0.7893 g/mL is mixed with 20.0 mL of water if density 0.997 g/mL at 25°C, the
final solution has the density of 0.9571 g/mL. Calculate the percentage change in volume on mixing. Also calculate the
molarity of the final solution. [Ans 3.1% contraction, 5.89 M]](https://image.slidesharecdn.com/solutionsclassxii-210623163445-230322025235-297921b1/85/solutionsclassxii-pdf-11-320.jpg)






![Numerical Problems to practice:
Q.1. One litre of water under a nitrogen pressure 1 atm dissolves 0.02g of nitrogen at 293 K.
Calculate Henry’s law constant. [Ans. 7.75 x 104 atm]
Q.2. Calculate the amount of CO2 dissolved at 4 atm in 1 dm3 of water at 298 K. The Henry’s law
constant for CO2 at 298 K is 1.67 K bar. [Ans. 5.85 g]
Q.3. The mole fraction of He gas in a saturated solution at 20° C is 1.25 X 10-6. Calculate the
pressure of He gas above the solution. (KH of Helium at 20° C = 144.98 K bar). [Ans. 0.181 bar]
Q.4 Henrys law constant for the molality of methane in benzene at 298 K is 4.27 x 105 mm Hg.
Calculate the solubility of methane in benzene at 298 K under 760 mm Hg. [Ans. 178 x 10-5]
Think: At same temperature hydrogen is more soluble than helium gas in water. Which of these
gases will have more KH value & why?](https://image.slidesharecdn.com/solutionsclassxii-210623163445-230322025235-297921b1/85/solutionsclassxii-pdf-18-320.jpg)



![Numericals for practice:
(i) Vapour pressure of chloroform (molar mass = 119.5 g/mol) & dichloromethane (molar mass = 55 g/mol) at 298
K are 200 mm & 415 mm Hg respectively. (i) calculate the vapour pressure of solution prepared by mixing 25.2 g
of chloroform & 40 g of dichloromethane at 298K & (ii) mole fraction of each component in solution & vapour
phase? (Ans. P = 347.9 mm Hg, Mole fraction in vapour phase of CHCl3 = 0.18 & CH2Cl2 = 0.82)
(ii) An aqueous solution containing 28 % by weight of a liquid A (molar mass = 140 g/mol) has vapour pressure of
0.210 bar at 300K. Calculate the vapour pressure of pure liquid.
(Vapour pressure of pure water at 300 K = 0.198 bar) [Ans.0.448 bar]
(iii) At 20℃, the vapour pressure of pure liquid A is 22 mm Hg & that of pure liquid B is 75 mm Hg. What is the
composition of the solution of these two components that has a vapour pressure of 48.5 mm Hg at this
temperature(assume ideal behaviour). [Ans: xA = xB = 0.5]
(iv) Two liquids A & B have vapour pressures of 0.658 bar & 0.264 bar respectively. In an ideal solution of the two,
calculate the mole fraction of A at which the two liquids have equal partial pressures. [Ans.0.286]
(v) The liquids X & Y form ideal solution having vapour pressures 200 & 100 mm Hg respectively. Calculate the
mole fraction of component X in vapour phase in equilibrium with an equimolar solution of the two. [Ans: 0.67]](https://image.slidesharecdn.com/solutionsclassxii-210623163445-230322025235-297921b1/85/solutionsclassxii-pdf-22-320.jpg)





![Numerical problems to practice:
(i) Vapour pressure of chloroform (molar mass = 119.5 g /mol) & dichloromethane (molar mass = 55 g/mol) at 298
K are 200 mm & 415 mm Hg respectively. (i) calculate the vapour pressure of solution prepared by mixing 25.2 g
of chloroform & 40 g of dichloromethane at 298K & (ii) mole fraction of each component in solution & vapour
phase? (Ans. P = 347.9 mm Hg, Mole fraction in vapour phase of CHCl3 = 0.18 & CH2Cl2 = 0.82)
(ii) An aqueous solution containing 28% by weight of a liquid A (molar mass = 140g/mol) has vapour pressure of
0.210 bar at 300K. Calculate the vapour pressure of pure liquid.
(Vapour pressure of pure water at 300 K = 0.198 bar. [Ans.0.448 bar]
(iii) At 20℃, the vapour pressure of pure liquid A is 22 mm Hg & that of pure liquid B is 75 mm Hg. What is the
composition of the solution of these two components that has a vapour pressure of 48.5 mm Hg at this
temperature. (assume ideal behaviour). [Ans: xA = xB = 0.5]
(iv) Two liquids A & B have vapour pressures of 0.658 bar & 0.264 bar respectively. In an ideal solution of the two,
calculate the mole fraction of A at which the two liquids have equal partial pressures. [Ans.0.286]
(v) The liquids X & Y form ideal solution having vapour pressures 200 & 100 mm Hg respectively. Calculate the
mole fraction of component X in vapour phase in equilibrium with an equimolar solution of the two. [Ans: 0.67]](https://image.slidesharecdn.com/solutionsclassxii-210623163445-230322025235-297921b1/85/solutionsclassxii-pdf-29-320.jpg)
![Numerical Problems : (continued)
(vi) An aqueous solution of is made by dissolving 10 g of glucose (molar mass = 180 ) in 90 g of
water at 303 K. If the vapour pressure of pure water at 303 K be 32.8 mm Hg, what would be the
vapour pressure of the solution? [Ans 32.44 mm]
(vii) At 298 K the vapour pressure of pure benzene is 0.256 bar & vapour pressure of pure toluene is
0.0925 bar. If the mole fraction of benzene in solutions 0.40, (i) find the total vapour pressure of
solution (ii) calculate the composition of the vapour in terms of male fraction.
[Ans: Ptotal = 0.158 bar, ybenzene= 0.648 & ytoluene = 0.352]
(viii) Two liquids X & Y on mixing form an ideal solution. The vapour pressure of this solution
containing 3 mol of X & 1 mol of Y is 550 mm of Hg. But when 4 mol of X & I mol of Y are mixed the
vapour pressure of solution becomes 560mm of Hg. What will be he vapour pressure of pure liquids
X and Y at this temperature? [Ans: X = 600 mm Y = 400mmof Hg].
HOTS
(ix) At a certain temperature, the vapour pressure (in mm of Hg) of CH3OH & C2H5OH solution is
represented by P= 119 x +135 where x is the mole fraction of CH3OH. Find the vapour pressures of
pure components at this temperature? [Ans: pmethanol=254 mm, pethanol=135mm]](https://image.slidesharecdn.com/solutionsclassxii-210623163445-230322025235-297921b1/85/solutionsclassxii-pdf-30-320.jpg)



![Problems to practice:
(1) The vp of water is 12.3 kPa at 300K. Calculate the v.p. of 1 molal solution of a solute in it. [Ans. 12.08kPa]
(2) The vapour pressure of pure benzene (molar mass = 78 g/mol) at a certain temperature is 262 bar. At the
same temperature, the vapour pressure of a solution containing 2 g of non volatile , non electrolytic solid in 100
g of benzene is 256 bar. What is the molecular mass of the solid? [Ans: 68.12 g/mol]
(3) The vapour pressure of a 5% aqueous solution of a non volatile organic substance at 373K is 745 mm Hg.
Calculate the molar mass of the solute. (Vapour pressure of water at 373 K is = 760 mm Hg) [Ans: 48 g/mol]
(4) What mass of a non-volatile solute urea (molar mass 60 g/mol) needed to be dissolved in 100 g of water in
order to decrease the vapour pressure of water by 25%? Calculate the molality of solution?
[Ans: 111 g, 18.5 m]
(5) The vapour pressure of water at 293 K is 0.0231 bar & the vapour pressure of solution of 108.24 g of a
compound in 1000 g of water at the same temperature is 0.0228 bar. Calculate the molar mass of the solute.
[Ans: 150 g/mol]](https://image.slidesharecdn.com/solutionsclassxii-210623163445-230322025235-297921b1/85/solutionsclassxii-pdf-34-320.jpg)



![Elevation of boiling point of a solution of non-volatile solute (𝜟𝑻𝒃)
PROBLEMS for practice:
(1) On dissolving 3.24 g of sulphur in 40 g of benzene , the boiling point of solution was higher than that of
benzene by 0.81 K. (Kb of benzene is 2.53 K kg/mol). What is the molecular formula of sulphur?
[Ans. S8]
(2) A solution of glycerol (molar mass = 92 g/mol) in water is prepared by dissolving some glycerol in 500 g of
water. This solution had a boiling point of 100.42℃ while pure water boils at 100℃ Find the mass of glycerol
dissolved in the solution? [Ans. 37.73 g]
(3) What is the elevation in boiling point is to be expected when 5 g of urea (molar mass = 60) are dissolved
in 75 g of it? (Kb of alcohol= 1.15 K kg/mol) [Ans. 1.28K)
(4) A solution of 12.5 g of a non electrolyte solid in 175 g of water gave the boiling point elevation of 0.70 K.
Calculate the molar mass of the substance (Kb of water = 0.52K kg/mol) [Ans. 53.06 ]](https://image.slidesharecdn.com/solutionsclassxii-210623163445-230322025235-297921b1/85/solutionsclassxii-pdf-38-320.jpg)




![(iii) Depression of freezing point of a solution of non volatile solute (𝚫𝐓𝐟)
Problems for practice:
(1) Calculate the mass of a compound (molar mass = 256 g /mol) to be dissolved in 75 g of benzene to lower its
freezing point by 0.48 K (Kf of benzene = 5.12 K kg /mol). [Ans. 1.8 g]
(2) 1.0 g of a non electrolyte solute dissolved in 50 g of benzene lowered the freezing pont of benzene by 0.40K. Find
the molar mass of solute (Kf of benzene = 5.12 K kg/mol). [Ans. 256 g/mol]
(3) Two aqueous solutions containing 7.5 g of urea (molar mass = 60) & 42.75 g substance X in 100 g water freeze at
same temperature. Calculate the molar mass of X. [Ans. 342 g/mol]
(4) An aqueous solution freezes at 272.07 K while pure water freezes at 273 K. Determine the molality & boiling
point of the solution. (Kb & Kf of water are 0.52 & 1.86 K kg/mol respectively) [Ans, 0.5 m, 373.256 K]
(5) In cold countries, water gets frozen causing damage to the car radiator. Ethylene glycol is used as an antifreeze.
Calculate the amount of ethylene glycol to be added to 4 kg of water to prevent freezing ̶6℃.
(Kf for water = 1.85 K/m) [Ans. 804.32 g]](https://image.slidesharecdn.com/solutionsclassxii-210623163445-230322025235-297921b1/85/solutionsclassxii-pdf-43-320.jpg)
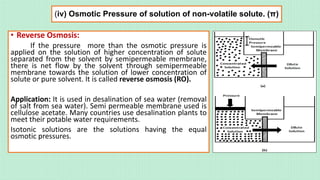
![(iv) Osmotic Pressure of solution of non-volatile solute. (π)
• Osmosis is the phenomenon of net movement of
solvent particles through from a region of higher
concentration of solvent (lower concentration of
solute) in solution to lower concentration of solvent
(higher concentration of solute) in solution. It is
temperature dependent. A semi permeable
membrane may be animal bladder like pig’s
bladder, parchment paper etc.
• Synthetic semi permeable is gelatinous
Cu2[Fe(CN)6].
• Osmotic pressure (π) of a solution is the minimum
pressure that must be applied on the solution of
higher concentration of solute just to prevent the
net flow of the solvent molecules into the solution
through the semipermeable membrane at a given
temperature. Osmotic pressure depends upon the
concentration of solution & its temperature.](https://image.slidesharecdn.com/solutionsclassxii-210623163445-230322025235-297921b1/85/solutionsclassxii-pdf-44-320.jpg)

![(iv) Osmotic Pressure of solution of non-volatile solute. (π)
[Osmotic pressure of a solution is the excess pressure that must be applied to a solution of higher concentration of
solute to prevent osmosis].
Experimentally, it is observed for a dilute solution of non-volatile solute osmotic pressure is proportional to the molarity
(C in mol L-1) of solution at a given temperature T in kelvin scale.
Hence, π = CRT =
𝐧𝐁
𝐕
𝐑𝐓 =
𝐰𝐁
𝐦𝐁
𝐑𝐓
𝐕
where C =
𝐧𝐁
𝐕
𝐦𝐨𝐥/𝐋
Thus we can calculate the molar mass of solute in solution as mB =
𝐖𝐁𝐑𝐓
𝛑 𝐕
Remember:
1 atm = 760 torr = 760 mm of Hg = 76 cm of Hg = 1.013 bar = 1.013 x 105 Pa & 1 bar = 0.987 atm).
Osmotic pressure method is the best method among other colligative property methods to calculate the
molar mass of macromolecules proteins, polymers etc because, π is calculated at room temperature &
molarity of solution is used in place of molality. So error in measurement is less. Also, for a dilute solution,
the magnitude of π value is appreciably large.](https://image.slidesharecdn.com/solutionsclassxii-210623163445-230322025235-297921b1/85/solutionsclassxii-pdf-46-320.jpg)




![OSMOTIC PRESSURE (Problems for practice):
(1) Calculate the osmotic pressure in Pa exerted by a solution prepared by dissolving 1.0 g of polymer of
molar mass 185,000 in 450 mL of water at 37℃. R = 8.314 J/mol/K. [Ans. 30.96 Pa]
(2) A 5% solution of cane sugar (malar mass = 342) is isotonic with 0.877% solution of urea. Calculate the
molar mass of urea. [Ans.59.99 g/mol]
(3) The osmotic pressure of human blood is 7.7 atm at 40℃. (a) What is the total concentration of all solute
in blood? (b) Assuming the concentration to be essentially the same as the molality, calculate the freezing
point of blood. (Kf = 1.86 K kg/mol) [ Ans.0.3 M , - 0.558℃]
(4) 600 mL of aqueous solution containing 2.5 g of a protein shows an osmotic pressure of 25 mm Hg at
27℃. Calculate the relative molar mass of protein. [Ans, 3119.8 g/mol]
HOTS
(5) At 10℃, the osmotic pressure of urea solution is 500 mm Hg. The solution is diluted & the temperature
is raised to 25℃, when the osmotic pressure becomes 105.3mm Hg. Calculate the extent of dilution.
[Ans. 5 times]](https://image.slidesharecdn.com/solutionsclassxii-210623163445-230322025235-297921b1/85/solutionsclassxii-pdf-52-320.jpg)





![Numerical problems using van’t Hoff’s factor :
(1) Calculate the boiling point of solution when 2 g of Na2SO4 (molar mass = 142) was dissolved in 50 g of water, assuming Na2SO4
undergoes complete ionisation. (Kb for water = 0.52 K kg/mol)
Solution: Weight of solute, wB = 2 g, molar mass mB = 142 g/mol, wA = 50 g, Kb = 0.52 K kg/mol
And Na2SO4 → 2Na+ + SO4
2- Here i = 3
We know, ΔTb = 𝐢
𝟏𝟎𝟎𝟎.𝐊𝐛 𝐰𝐁
𝐦𝐁 𝐰𝐀
=
𝟑 𝐱 𝟏𝟎𝟎𝟎 𝐱 𝟎.𝟓𝟐 𝐱 𝟐
𝟏𝟒𝟐 𝐱 𝟓𝟎
= 0.439 Boiling point of solution] Tb = 373 + 0.439 = 373.439 K (Ans.)
(2) A solution containing 3.1 g of BaCl2 (Molar mass = 208.3) in 250 g of water boils at 100.083℃. Calculate the Van’t Hoff’s factor and
molality of BaCl2 in this solution. (Kb for water = 0.52 K/m)
Solution: wB = 3.1 g, wA = 250 g, mB = 208.3, Kb = 0.52 K/m & ΔTb =100.083 – 100 = 0.083℃ = 0.083 K
We know, ΔTb = 𝐢
𝟏𝟎𝟎𝟎.𝐊𝐛 𝐰𝐁
𝐦𝐁 𝐰𝐀
𝐨𝐫, 0.083 =
𝐢 𝐱 𝟏𝟎𝟎𝟎 𝐱 𝟎.𝟓𝟐 𝐱 𝟑.𝟏
𝟐𝟎𝟖.𝟑 𝐱 𝟐𝟓𝟎
On calculation I = 2.68 (Ans.)
(3) 3.9 g of benzoic acid (molar mass = 122 g/mol) dissolved in 49 g of benzene shows a depression in freezing point of 1.62K. Calculate
the van’t Hoff factor & predict the nature of solute (associated or dissociated) ( Given: Kf of benzene = 4.9 K kg/mol)
Solution: Given : wB = 3.9 g, wA = 49 g, mB = 122, ΔTf = 1.62 K, Kf = 4.9K kg / mol
We know, ΔTf = 𝐢
𝟏𝟎𝟎𝟎.𝐊𝐟 𝐰𝐁
𝐦𝐁 𝐰𝐀
or 1.62 =
𝐢 𝐱 𝟏𝟎𝟎𝟎 𝐱 𝟒.𝟗 𝐱 𝟑.𝟗
𝟏𝟐𝟐 𝐱 𝟒𝟗
On calculation, value of i = 0.506
Since ’i’ < 1, solute benzoic acid is associated in benzene. (Ans.)](https://image.slidesharecdn.com/solutionsclassxii-210623163445-230322025235-297921b1/85/solutionsclassxii-pdf-58-320.jpg)
![Some Typical questions:
(i). Why are aquatic species more comfortable in cold water in comparison to warm water ?
(ii). Why is the vapour pressure of an aqueous solution of glucose lower than that of water ?
(iii) Why is ethylene glycol added in car radiator water in cold countries?
Problems for practice:
(1) A 1.2% (w/V) solution of NaCl (molar mass = 58.5) is isotonic with 7.2% (w/V) of glucose (molar mass = 180)
solution Calculate the degree of dissociation of NaCl solution. (Ans. degree of dissociation = 0.96
(2)0.6 mL of acetic acid (molar mass = 60) having density 1.06 g/mL is dissolved in 1 L of water. The depression in
freezing point observed for this strength of acid was 0.0205℃. Calculate the Van’t Hoff factor & dissociation
constant of the acid. (Kf = 1.86K kg/mol,) [Ans i = 1.041 & Ka = 1.86 x 10-5]
(3) Calculate the amount of KCl which must be added to 1 kg of water so that the freezing point is depresses by 2 K
(Kf = 1.86 K kg/mol & atomic masses: K = 39, Cl = 35.5) [ Ans.40.05 g]
(4) 0.01 m aqueous solution of K4[Fe(CN)6] freezes at – 0.062℃. What is the apparent %age of dissociation
(Kf for water = 1.86K kg/mol) [Ans. 77.7%]
(5) Calculate the normal freezing point of sea water sample containing 3.8 % NaCl & 0.12 & MgCl2 by mass.
(Kf for water = 1.86 K/m & atomic masses Na = 23 Cl = 35.5, Mg = 24) [Ans. – 2.59℃]](https://image.slidesharecdn.com/solutionsclassxii-210623163445-230322025235-297921b1/85/solutionsclassxii-pdf-59-320.jpg)
