Solid supported Pd-PEPPSI-IPentCl-10
•Download as PPTX, PDF•
0 likes•146 views
This document summarizes research on immobilizing palladium N-heterocyclic carbene (NHC) catalysts on solid supports for use in heterogeneous catalysis. The researcher developed a new method to attach robust NHC ligands to a silica support through an aniline linker. Initial proof-of-concept reactions confirmed the activity of the immobilized catalyst before and after joining to the support. The researcher aims to develop this immobilization strategy further to enable efficient heterogeneous catalytic reactions while retaining the electronic properties of the NHC ligand.
Report
Share
Report
Share
Recommended
2019 tp-cpp-poster sces

Inorganic chem project- Synthesis of gadolinium catecholate for Lanthanide Separation, aiming to solve nuclear waste.
Misconceptions in Photocatalysis

Photocatalysis has now become an emerging scientific discipline due to its interdisciplinary nature. The wide range of research groups is now working on different aspects of photocatalysis worldwide. It is one of the technology the world looking forward to address environmental as well as energy related issues. Hence we can call it as a technology for the future or a dream technology! We need to overcome too many hurdles to implement this technology in real life. Like any other discipline there is a lot of misunderstanding/ misconceptions in photocatalysis.
Most frequently cited article in the field of photocatalysis is by Fujishima and Honda published in 1972 in nature and it has been cited by the photocatalytic community as an origin of photocatalysis. This aspect is not true at all. This article cannot be the origin of photocatalysis. This article only promoted photocatalytic studies. The author itself, actually, started a research career in the “boom” of photocatalytic studies initiated by this article.
This small presentation aims to deliver some misconceptions like above in photocatalysis. The entire presentation is based on different personal commentaries written by Jean Mary Hermann and Bunsho Ohtani. Some recent articles relevant to the topic are collected by the speaker itself and put it in one platform.
Recommended
2019 tp-cpp-poster sces

Inorganic chem project- Synthesis of gadolinium catecholate for Lanthanide Separation, aiming to solve nuclear waste.
Misconceptions in Photocatalysis

Photocatalysis has now become an emerging scientific discipline due to its interdisciplinary nature. The wide range of research groups is now working on different aspects of photocatalysis worldwide. It is one of the technology the world looking forward to address environmental as well as energy related issues. Hence we can call it as a technology for the future or a dream technology! We need to overcome too many hurdles to implement this technology in real life. Like any other discipline there is a lot of misunderstanding/ misconceptions in photocatalysis.
Most frequently cited article in the field of photocatalysis is by Fujishima and Honda published in 1972 in nature and it has been cited by the photocatalytic community as an origin of photocatalysis. This aspect is not true at all. This article cannot be the origin of photocatalysis. This article only promoted photocatalytic studies. The author itself, actually, started a research career in the “boom” of photocatalytic studies initiated by this article.
This small presentation aims to deliver some misconceptions like above in photocatalysis. The entire presentation is based on different personal commentaries written by Jean Mary Hermann and Bunsho Ohtani. Some recent articles relevant to the topic are collected by the speaker itself and put it in one platform.
Hot hole transfer from Ag nanoparticles to multiferroic YMn2O5 nanowires enab...

Plasmonic hot carriers with a nonthermal distribution of kinetic energies have opened up new avenues in photovoltaics, photodetection and photocatalysis. While several articles have reported ultrafast hot electron injection from coinage metals into n-type semiconductors across Schottky barriers and efficient subsequent utilization of injected hot electrons, reports of hot hole harvesting are comparatively rare due to the difficulty in forming Schottky junctions between p-type semiconductors and high work function metals. In this communication, we report the fabrication, characterization and theoretical calculations of a novel integrated multiferroic-plasmonic system comprising YMn2O5 nanowires decorated on their surface with Ag nanoparticles (NPs). A Schottky barrier for holes exists at the YMn2O5-Ag hetero-interface and hot holes were injected from Ag across this barrier. The synthesized hybrid along with bare Ag NPs were tested for Raman surface photocatalytic reduction of 4-NBT (4-nitrobenzenethiol) to DMAB (p, p′-dimercaptoazobenzene) where the composite demonstrated superior activity compared to the bare metal. Ultraviolet photoelectron spectroscopy (UPS) revealed a significantly reduced work function of the composite compared to the pristine Ag, indicative of more energetic hot electrons on the surface of the composite required for efficient photoreduction. Density functional theory (DFT)-based calculations revealed localization of molecular orbitals supportive of a possible hole transfer from YMn2O5 to Ag and a reorganization of electronic states beneficial for plasmon-induced charge carrier enhancement. DFT results also indicated a purely electronic contribution to the ferroelectric polarization of YMn2O5 over and above the ionic contribution, which originated from the magnetic polarization of O 2p states.
Rate limiting interfacial hole transfer in Sb2S3 solid state solar cells

view article: http://dx.doi.org/10.1039/C3EE43844A
Solid-state sensitized solar cells (SSCs) utilizing semiconductor absorbers overcome the issues of leakage and evaporation encountered in liquid-junction SSCs, and offer the potential for efficient, low cost photovoltaics. For widespread commercialization these solar cells require higher power conversion efficiency than is currently obtained with state-of-the-art devices. One critical component to this is the efficient extraction of photogenerated charges from the semiconductor absorber material. In this study, we decouple the two steps of hole transfer in the Sb2S3/CuSCN system: diffusion of holes in the Sb2S3 absorber layer, and transfer of these holes across Sb2S3–CuSCN interface. We find that interfacial transfer is the major limiting step in the thin (< 20 nm) Sb2S3 films used for high efficiency Sb2S3 photovoltaics. Decoupling of diffusion and interfacial transfer leads to a deeper understanding of the mechanism of hole transfer. This information has implications for the future design of semiconductor-based SSCs as it points to an important, often neglected interface, the absorber-hole conductor interface, which can play an important role in charge extraction.
Heterostructured nanocomposite tin phthalocyanine@mesoporous ceria (SnPc@CeO2...

Heterostructured tin phthalocyanine supported to mesoporous ceria was synthesized and used a
photocatalyst for CO2 reduction under visible light. The photoreduction CO2 activities of the
heterostructures were investigated in the presence of triethylamine as sacrificial agent. The developed
photocatalyst exhibited high catalytic activity for photoreduction of CO2 and after 24 hours of visible
light irradiation 2342 mmol g1 cat of methanol (fMeOH ¼ 0.0223 or 2.23%) and 840 mmol g1 cat of CO
(fCO ¼ 0.0026 or 0.26%) were obtained as the major reaction products. The methanol formation rate
(RMeOH) and CO formation rate (RCO) was found to be 97.5 mmol h1 g1 cat and 35.0 mmol h1 g1 cat
respectively. While under the identical experimental conditions mesoporous ceria (meso-CeO2) gave
only 316 mmol g1 cat of methanol (fMeOH ¼ 0.003 or 0.30%) and 126 mmol g1 cat CO (fCO ¼ 0.0004
or 0.04%) with product formation rate RMeOH ¼ 13.2 mmol h1 g1 cat and RCO ¼ 5.3 mmol h1 g1 cat.
Furthermore, the recovered catalyst showed consistent catalytic activity for at least five runs without any
significant loss in product yields
Lithium Sulfur Battery: Current Status and Future Prospects.

ECS 227th Meeting, Chicago, USA, 24-28th, May, 2015.
Photo-assisted oxidation of thiols to disulfides using cobalt ‘‘Nanorust’’ un...

Heterogeneous ‘‘Nanorust’’ containing cobalt oxide has been developed for the visible light assisted
oxidation of thiols to disulfides using molecular oxygen as an oxidant under alkaline free conditions and
therefore more environmentally friendly. Pyrolysis of heterogenized tetrasulfonated cobalt(II) phthalocyanine
(CoPcS) supported on mesoporous ceria (CeO2) transforms it into a novel heterogeneous ‘‘Nanorust’’
containing CoOx-C,N@CeO2 which exhibited higher catalytic activity than the homogeneous CoPcS as well
as the ceria immobilized CoPcS catalyst. Importantly, these catalysts could easily be recovered and recycled
for several runs, which makes the process greener and cost-effective.
2.9 Notes

Guided notes covering material from Topic 2.9 of the updated IB Biology syllabus for 2016 exams. Notes sequence and prompts are based on the Oxford IB Biology textbook by Allott and Mindorff.
Trap and Transfer. Two-Step Hole Injection Across the Sb2S3/CuSCN Interface i...

Trap and Transfer. Two-Step Hole Injection Across the Sb2S3/CuSCN Interface in Solid State Solar Cells. ACS Nano, 2013, ASAP.
DOI: 10.1021/nn403058f
In solid-state semiconductor-sensitized solar cells, commonly known as extremely thin absorber (ETA) or solid-state quantum dot sensitized solar cells (QDSCs), transfer of photogenerated holes from the absorber species to the p-type hole conductor plays a critical role in the charge separation process. Using Sb2S3 (absorber) and CuSCN (hole conductor), we have constructed ETA solar cells exhibiting a power conversion efficiency of 3.3%. The hole transfer from excited Sb2S3 into CuSCN, which limits the overall power conversion efficiency of these solar cells, is now independently studied using transient absorption spectroscopy. In the Sb2S3 absorber layer, photogenerated holes are rapidly localized on the sulfur atoms of the crystal lattice, forming a sulfide radical (S−•) species. This trapped hole is transferred from the Sb2S3 absorber to the CuSCN hole conductor with an exponential time constant of 1680 ps. This process was monitored through the spectroscopic signal seen for the S−• species in Sb2S3, providing direct evidence for the hole transfer dynamics in ETA solar cells. Elucidation of the hole transfer mechanism from Sb2S3 to CuSCN represents a significant step toward understanding charge separation in Sb2S3 solar cells, and provides insight into the design of new architectures for higher efficiency devices.
More Related Content
What's hot
Hot hole transfer from Ag nanoparticles to multiferroic YMn2O5 nanowires enab...

Plasmonic hot carriers with a nonthermal distribution of kinetic energies have opened up new avenues in photovoltaics, photodetection and photocatalysis. While several articles have reported ultrafast hot electron injection from coinage metals into n-type semiconductors across Schottky barriers and efficient subsequent utilization of injected hot electrons, reports of hot hole harvesting are comparatively rare due to the difficulty in forming Schottky junctions between p-type semiconductors and high work function metals. In this communication, we report the fabrication, characterization and theoretical calculations of a novel integrated multiferroic-plasmonic system comprising YMn2O5 nanowires decorated on their surface with Ag nanoparticles (NPs). A Schottky barrier for holes exists at the YMn2O5-Ag hetero-interface and hot holes were injected from Ag across this barrier. The synthesized hybrid along with bare Ag NPs were tested for Raman surface photocatalytic reduction of 4-NBT (4-nitrobenzenethiol) to DMAB (p, p′-dimercaptoazobenzene) where the composite demonstrated superior activity compared to the bare metal. Ultraviolet photoelectron spectroscopy (UPS) revealed a significantly reduced work function of the composite compared to the pristine Ag, indicative of more energetic hot electrons on the surface of the composite required for efficient photoreduction. Density functional theory (DFT)-based calculations revealed localization of molecular orbitals supportive of a possible hole transfer from YMn2O5 to Ag and a reorganization of electronic states beneficial for plasmon-induced charge carrier enhancement. DFT results also indicated a purely electronic contribution to the ferroelectric polarization of YMn2O5 over and above the ionic contribution, which originated from the magnetic polarization of O 2p states.
Rate limiting interfacial hole transfer in Sb2S3 solid state solar cells

view article: http://dx.doi.org/10.1039/C3EE43844A
Solid-state sensitized solar cells (SSCs) utilizing semiconductor absorbers overcome the issues of leakage and evaporation encountered in liquid-junction SSCs, and offer the potential for efficient, low cost photovoltaics. For widespread commercialization these solar cells require higher power conversion efficiency than is currently obtained with state-of-the-art devices. One critical component to this is the efficient extraction of photogenerated charges from the semiconductor absorber material. In this study, we decouple the two steps of hole transfer in the Sb2S3/CuSCN system: diffusion of holes in the Sb2S3 absorber layer, and transfer of these holes across Sb2S3–CuSCN interface. We find that interfacial transfer is the major limiting step in the thin (< 20 nm) Sb2S3 films used for high efficiency Sb2S3 photovoltaics. Decoupling of diffusion and interfacial transfer leads to a deeper understanding of the mechanism of hole transfer. This information has implications for the future design of semiconductor-based SSCs as it points to an important, often neglected interface, the absorber-hole conductor interface, which can play an important role in charge extraction.
Heterostructured nanocomposite tin phthalocyanine@mesoporous ceria (SnPc@CeO2...

Heterostructured tin phthalocyanine supported to mesoporous ceria was synthesized and used a
photocatalyst for CO2 reduction under visible light. The photoreduction CO2 activities of the
heterostructures were investigated in the presence of triethylamine as sacrificial agent. The developed
photocatalyst exhibited high catalytic activity for photoreduction of CO2 and after 24 hours of visible
light irradiation 2342 mmol g1 cat of methanol (fMeOH ¼ 0.0223 or 2.23%) and 840 mmol g1 cat of CO
(fCO ¼ 0.0026 or 0.26%) were obtained as the major reaction products. The methanol formation rate
(RMeOH) and CO formation rate (RCO) was found to be 97.5 mmol h1 g1 cat and 35.0 mmol h1 g1 cat
respectively. While under the identical experimental conditions mesoporous ceria (meso-CeO2) gave
only 316 mmol g1 cat of methanol (fMeOH ¼ 0.003 or 0.30%) and 126 mmol g1 cat CO (fCO ¼ 0.0004
or 0.04%) with product formation rate RMeOH ¼ 13.2 mmol h1 g1 cat and RCO ¼ 5.3 mmol h1 g1 cat.
Furthermore, the recovered catalyst showed consistent catalytic activity for at least five runs without any
significant loss in product yields
Lithium Sulfur Battery: Current Status and Future Prospects.

ECS 227th Meeting, Chicago, USA, 24-28th, May, 2015.
Photo-assisted oxidation of thiols to disulfides using cobalt ‘‘Nanorust’’ un...

Heterogeneous ‘‘Nanorust’’ containing cobalt oxide has been developed for the visible light assisted
oxidation of thiols to disulfides using molecular oxygen as an oxidant under alkaline free conditions and
therefore more environmentally friendly. Pyrolysis of heterogenized tetrasulfonated cobalt(II) phthalocyanine
(CoPcS) supported on mesoporous ceria (CeO2) transforms it into a novel heterogeneous ‘‘Nanorust’’
containing CoOx-C,N@CeO2 which exhibited higher catalytic activity than the homogeneous CoPcS as well
as the ceria immobilized CoPcS catalyst. Importantly, these catalysts could easily be recovered and recycled
for several runs, which makes the process greener and cost-effective.
2.9 Notes

Guided notes covering material from Topic 2.9 of the updated IB Biology syllabus for 2016 exams. Notes sequence and prompts are based on the Oxford IB Biology textbook by Allott and Mindorff.
Trap and Transfer. Two-Step Hole Injection Across the Sb2S3/CuSCN Interface i...

Trap and Transfer. Two-Step Hole Injection Across the Sb2S3/CuSCN Interface in Solid State Solar Cells. ACS Nano, 2013, ASAP.
DOI: 10.1021/nn403058f
In solid-state semiconductor-sensitized solar cells, commonly known as extremely thin absorber (ETA) or solid-state quantum dot sensitized solar cells (QDSCs), transfer of photogenerated holes from the absorber species to the p-type hole conductor plays a critical role in the charge separation process. Using Sb2S3 (absorber) and CuSCN (hole conductor), we have constructed ETA solar cells exhibiting a power conversion efficiency of 3.3%. The hole transfer from excited Sb2S3 into CuSCN, which limits the overall power conversion efficiency of these solar cells, is now independently studied using transient absorption spectroscopy. In the Sb2S3 absorber layer, photogenerated holes are rapidly localized on the sulfur atoms of the crystal lattice, forming a sulfide radical (S−•) species. This trapped hole is transferred from the Sb2S3 absorber to the CuSCN hole conductor with an exponential time constant of 1680 ps. This process was monitored through the spectroscopic signal seen for the S−• species in Sb2S3, providing direct evidence for the hole transfer dynamics in ETA solar cells. Elucidation of the hole transfer mechanism from Sb2S3 to CuSCN represents a significant step toward understanding charge separation in Sb2S3 solar cells, and provides insight into the design of new architectures for higher efficiency devices.
What's hot (19)
Hot hole transfer from Ag nanoparticles to multiferroic YMn2O5 nanowires enab...

Hot hole transfer from Ag nanoparticles to multiferroic YMn2O5 nanowires enab...
Rate limiting interfacial hole transfer in Sb2S3 solid state solar cells

Rate limiting interfacial hole transfer in Sb2S3 solid state solar cells
Heterostructured nanocomposite tin phthalocyanine@mesoporous ceria (SnPc@CeO2...

Heterostructured nanocomposite tin phthalocyanine@mesoporous ceria (SnPc@CeO2...
Lithium Sulfur Battery: Current Status and Future Prospects.

Lithium Sulfur Battery: Current Status and Future Prospects.
Photo-assisted oxidation of thiols to disulfides using cobalt ‘‘Nanorust’’ un...

Photo-assisted oxidation of thiols to disulfides using cobalt ‘‘Nanorust’’ un...
Trap and Transfer. Two-Step Hole Injection Across the Sb2S3/CuSCN Interface i...

Trap and Transfer. Two-Step Hole Injection Across the Sb2S3/CuSCN Interface i...
Viewers also liked
6º Congresso Estadual | Baldur Schubert

Apresentação de Baldur Schubert no 6º Congresso Estadual da APEPREM - 12 a 14/ABR/2010 - Campinas, SP
6º Congresso Estadual | Delubio Gomes

Apresentação de Delubio Gomes no 6º Congresso Estadual da APEPREM - 12 a 14/ABR/2010 - Campinas, SP
6º Congresso Estadual | Otoni Gonçalves Guimarães

Apresentação de Otoni Gonçalves Guimarães no 6º Congresso Estadual da APEPREM - 12 a 14/ABR/2010 - Campinas, SP
Viewers also liked (20)
The castile in capri at pelican landing naples florida

The castile in capri at pelican landing naples florida
Similar to Solid supported Pd-PEPPSI-IPentCl-10
Cucurbituril[7] Host - Viologen Guest Complexes: Electrochromic and Photochem...![Cucurbituril[7] Host - Viologen Guest Complexes: Electrochromic and Photochem...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Cucurbituril[7] Host - Viologen Guest Complexes: Electrochromic and Photochem...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
PhD Defense Presentation of Marina Freitag
Electro-kinetic Remediation of Soil

This presentation describes the removal of metal contaminants from soil using electro-kinetic remediation process
Computationally Driven Characterization of Magnetism, Adsorption, and Reactiv...

Metal organic frameworks (MOFs) are a class of nanoporous materials that are com- posed of metal-containing nodes connected by organic linkers. The study of MOFs has grown in importance due to the wide range of possible node and linker combinations, which allow tailoring towards specific applications. This work demonstrates that the- ory can complement experiment in a way that advances the chemical understanding of MOFs. This thesis contains the results of several investigations on three different areas of MOF research: 1) magnetism, 2) CO2 adsorption, and 3) catalysis.
Organic Semiconductor 

Organic semiconductor:
Difference b/w inorganic and organic semiconductor
Organic Semiconductor
Thin metal oxide/organic semiconductor
Motivation
Challenges
Overcome
Improvement in organic semiconductor device
Representation of organic electronic device
Transition metal oxide buffer layer
Comparison b/w organic electronics & silicon
Advantage metal oxide/organic semiconductor
Disadvantage metal oxide/organic semiconductor
Future of metal oxide/organic semiconductor
Conclusion
Thank you
Preeti Choudhary
chaudharypreeti1997@gmail.com
https://www.linkedin.com/in/preeti-choudhary-266414182/
https://t.me/physiwaves
https://t.me/barcphysics
https://t.me/phyiscswaves
Effect of metal dopant on photocatalytic performance of TiO2 nano particles 

This presentation will give the fundamental knowledge on photo- catalytic study on metal doped TiO2 semiconductors to the readers
Materials of pharmaceutical plant construction- Pharmaceutical Engineering

B. Pharmacy Second year, third semester
Electrokinetic Remediation of Heavy Metal contaminated soil

Removal of heavy metals from soil using low voltage electric current.Most economical and efficient method
TiO2 Nanomaterial

introduction to TiO2, importance of TiO2, synthesis method of TiO2 nanoparticle, properties of TiO2 nanoparticle, application of TiO2 nanoparticle.
Effect of Carbon and MgAl Oxide Supports and Potassium Promoter on Higher Alc...

Higher alcohol synthesis from syngas was studied over MoS2 during the induction period by varying the support, carbon (C) and MgAl mixed oxide (MMO), and the presence of the K promoter. MoS2 on C and MMO supports produced longer chain hydrocarbons compared to unsupported MoS2. Potassium is known to shift product distribution from hydrocarbons to higher alcohols. Before potassium addition, the C support was selective towards methanol and hydrocarbons while the MMO support was predominantly selective towards hydrocarbons. After K addition, the MMO support produced primarily higher alcohols, whereas the C support remained selective towards hydrocarbons with enhanced alcohol selectivity.
Similar to Solid supported Pd-PEPPSI-IPentCl-10 (20)
Cucurbituril[7] Host - Viologen Guest Complexes: Electrochromic and Photochem...![Cucurbituril[7] Host - Viologen Guest Complexes: Electrochromic and Photochem...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Cucurbituril[7] Host - Viologen Guest Complexes: Electrochromic and Photochem...](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Cucurbituril[7] Host - Viologen Guest Complexes: Electrochromic and Photochem...
Computationally Driven Characterization of Magnetism, Adsorption, and Reactiv...

Computationally Driven Characterization of Magnetism, Adsorption, and Reactiv...
Effect of metal dopant on photocatalytic performance of TiO2 nano particles 

Effect of metal dopant on photocatalytic performance of TiO2 nano particles
Materials of pharmaceutical plant construction- Pharmaceutical Engineering

Materials of pharmaceutical plant construction- Pharmaceutical Engineering
Electrokinetic Remediation of Heavy Metal contaminated soil

Electrokinetic Remediation of Heavy Metal contaminated soil
Effect of Carbon and MgAl Oxide Supports and Potassium Promoter on Higher Alc...

Effect of Carbon and MgAl Oxide Supports and Potassium Promoter on Higher Alc...
Solid supported Pd-PEPPSI-IPentCl-10
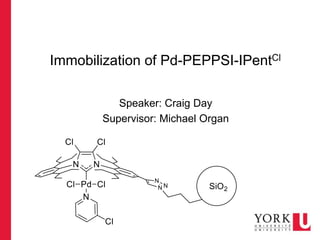
- 1. Immobilization of Pd-PEPPSI-IPentCl Speaker: Craig Day Supervisor: Michael Organ
- 2. 2010 Nobel Prize Winners http://www.nobelprize.org/ Cooper, T. W. J.; Campbell, I. B.; Macdonald, S. J. F. Angew. Chem. Int. Ed. 2010, 49, 80822 • 2010: 22% of all reactions in pharma are Pd-catalyzed couplings • 2013: Buchwald-Hartwig amination most common reaction in pharma
- 3. General catalytic cycle 3 Oxidative addition • Improved by electron rich metal Transmetallation • Ease impacted by reactivity of nucleophilic partner Reductive elimination • Reverse of oxidative addition • Improved by sterically bulky ligand
- 4. Phosphines N-Heterocyclic Carbenes (NHCs) 4 • σ-donating ligands - Increase e- density around metal centre - Easier oxidative addition - Difficult reductive elimination • Easily oxidized • Measured with Tolman cone angle • Electron-rich, neutral σ-donoring ligand • Steric and electronic properties are tuneable • Forms a very stable metal complex • Measured with percent buried volume
- 5. PEPPSI • Pyridine-Enhanced Precatalyst Preparation Stabilization Initiation Pd-PEPPSI-IPr Pd-PEPPSI-IPent Pd-PEPPSI-IPentCl 5 Increasing activity
- 6. Transition to heterogenous catalysis • Solid Support − Economical (recovery of metal and ligand) − Environmentally friendly − Reduce contamination • Flow Applications − Improved heat transfer − Efficent mass transport and mixing − Faster and safer reactions − Ease of scale-up SiliaCat® immobilized DPP-Pd complex6 • Silica − Cheap, widely available and studied • Phosphines − Easy to synthesize − Good catalyst − Suspected dissociation mechanism − Leaching issues
- 7. NHC Immobilization (1) (2) (3) 1. Collinson, J.-M.; Wilton-Ely, J. D. E. T.; Díez-González, S. Chem. Commun. 2013, 49 (97), 11358–11360 2. Mennecke, K.; Kirschning, A. Synthesis 2008, (20), 3267–3272. 3. Martínez, A.; Krinsky, J. L.; Peñafiel, I.; Castillón, S.; Loponov, K.; Lapkin, A.; Godard, C.; Claver, C. Catal. Sci. Technol. 2014, 5 (1), 310–319. 7 Alters backbone of NHC Not attached to NHC ligand Impacts steric properties Made in-situ
- 8. Objective • Advantages: − Robust − Tethered unit distant from metal centre − High yielding synthesis − Electronic properties of metal assumed to be the same − Preform metal complex for characterization 8
- 9. 1st generation: construction of aniline segments 9
- 10. Synthesis of protected immidazolium salt 10
- 11. Final steps to immobilization 11 Flow applications • Screened in flow (Negishi) • Active for 14 hours
- 12. Alkylation and decomposition 12 IPr IPent
- 13. Reduction 13 Reducing agent Equiv. Solvent Conversion (%) Yield (%) Borane 1.2 THF 100 41 + decomp. NaCNBH3 1.1 CH2Cl2 100 49 + decomp. Na(OAc)3B H 1.4 CH2Cl2 86 86
- 14. 2nd generation: construction of aniline segments 14
- 16. Summary • Robust, simple approach targeting complex anilines • Proof of concept confirmed for transformations before joining segments 16
- 17. Acknowledgement • Supervisor − Dr. Organ • Committee − Dr. Orellana − Dr. Wilson • Lab members − Greg Price, PhD 17
Editor's Notes
- Heck – Pioneered pd-cross coupling Negishi – investigated cross coupling partners (ZnR) Suzuki – investigated the catalytic capability of pd-cross couplings