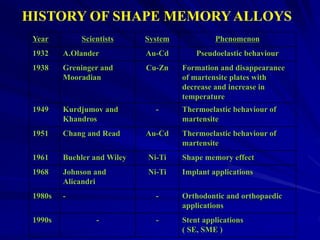
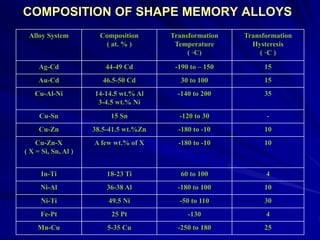
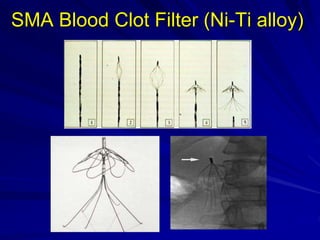
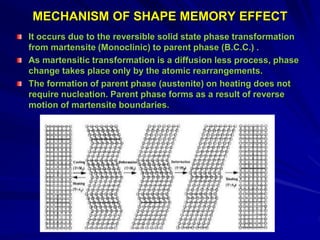
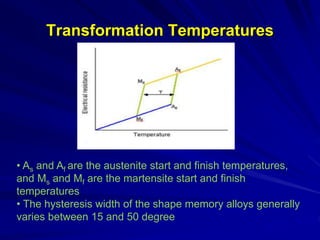
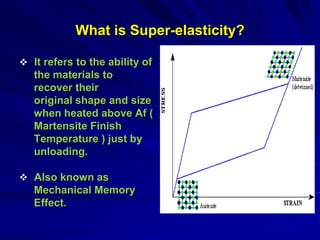
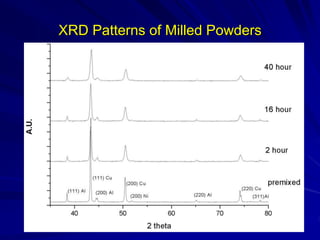
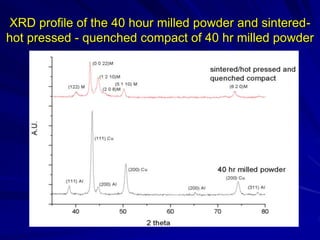
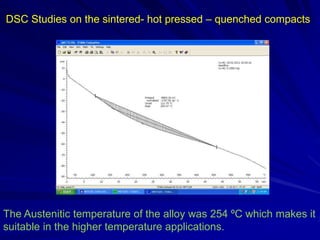
This document discusses smart materials, which are materials that can sense and respond to environmental stimuli. It classifies common smart materials and describes their properties and applications. Smart materials include piezoelectric materials, shape memory alloys, magnetostrictive materials, and active fluids. Shape memory alloys have the unique ability to return to their original shape after deformation by heating. Processing techniques like powder metallurgy and mechanical alloying can produce shape memory alloys with fine grain sizes and improved properties for applications.