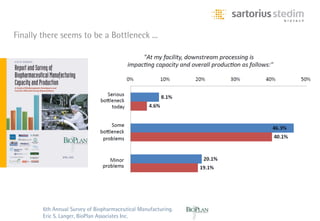
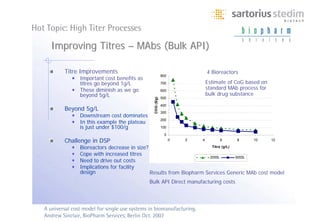
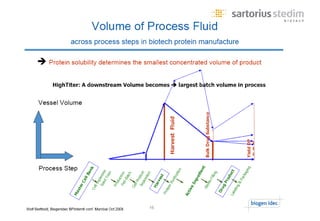
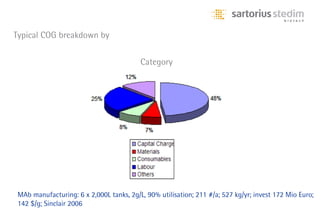
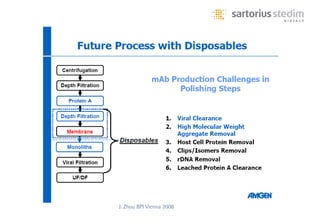
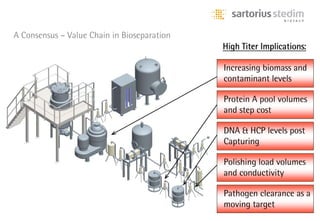
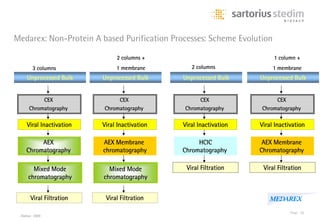
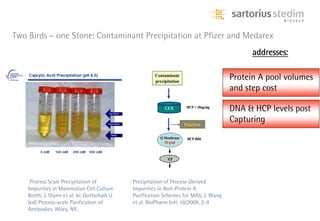
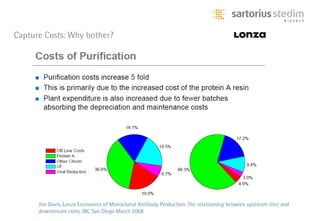
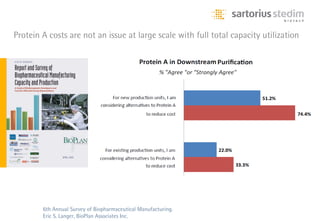
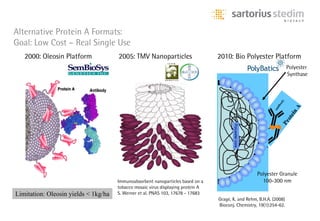
The document discusses the shift in downstream processing from a bottleneck to a driver of biomanufacturing. It notes that while high titers can reduce costs, downstream processing costs still dominate and present challenges to handle increased volumes and contaminant loads from improved processes. New technologies in filtration and chromatography using single-use disposables aim to improve throughput and capacity while reducing costs. The future of downstream processing involves revisiting past approaches with a focus on continuous improvement rather than identifying any single best solution.