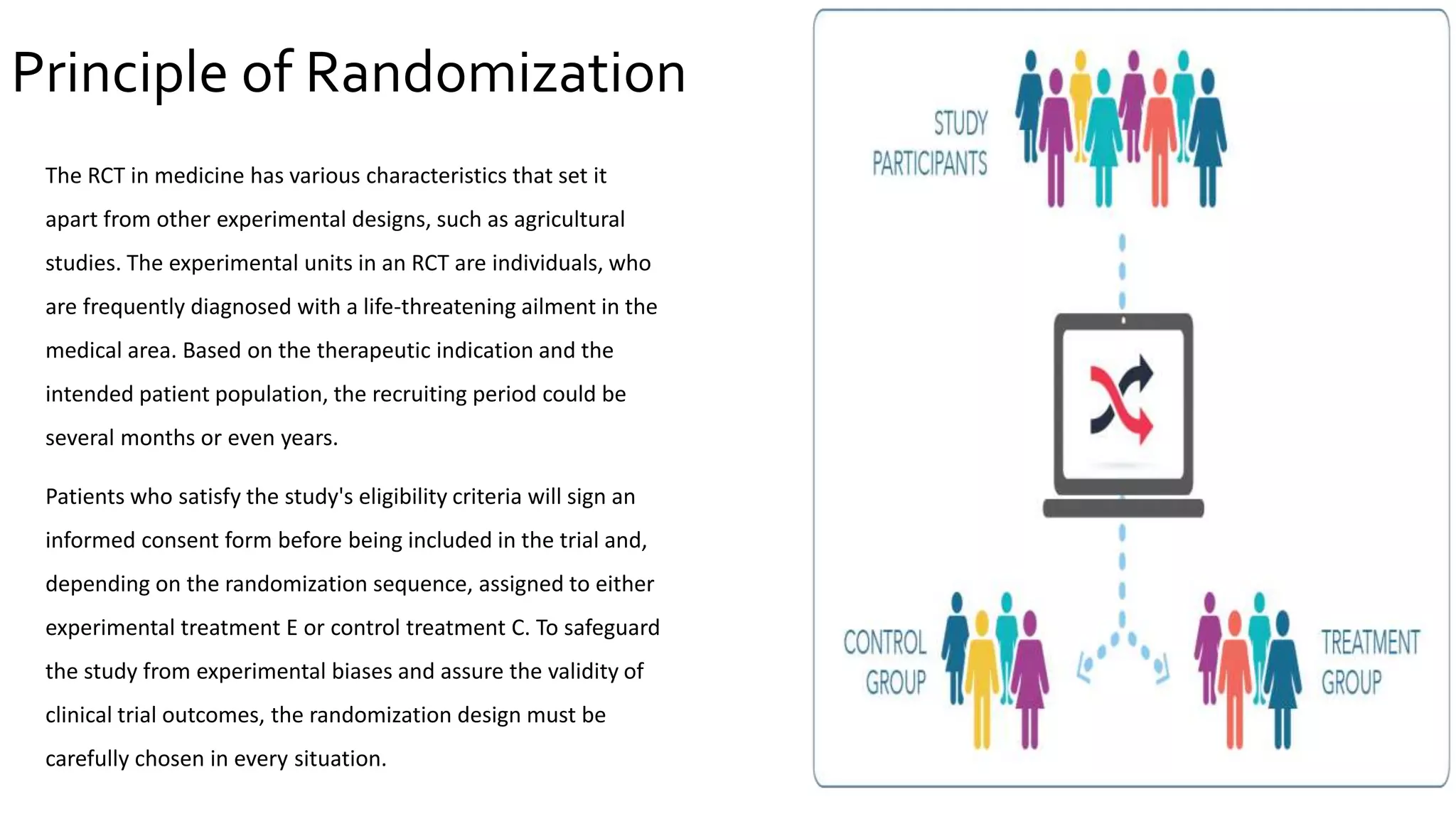
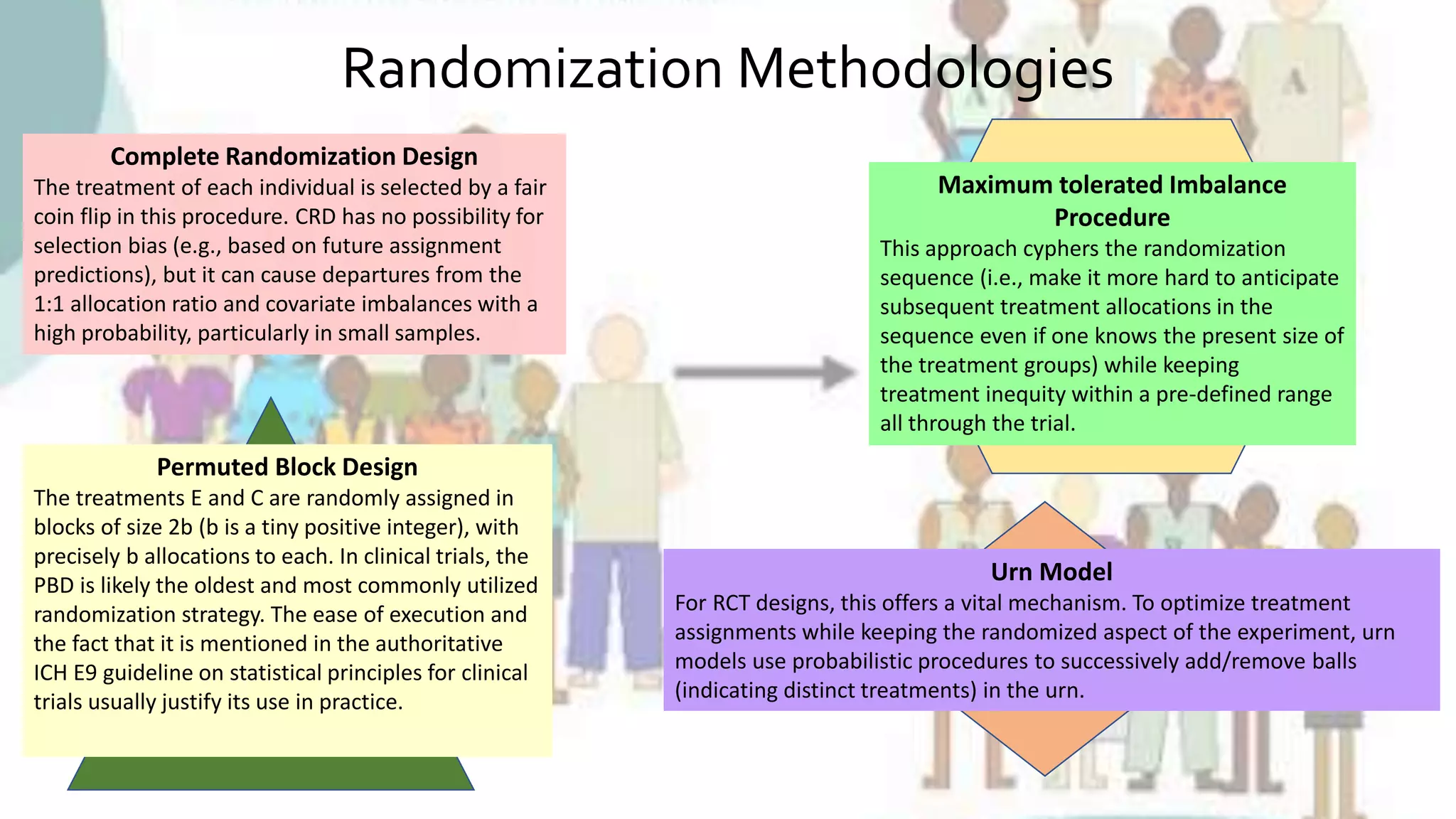
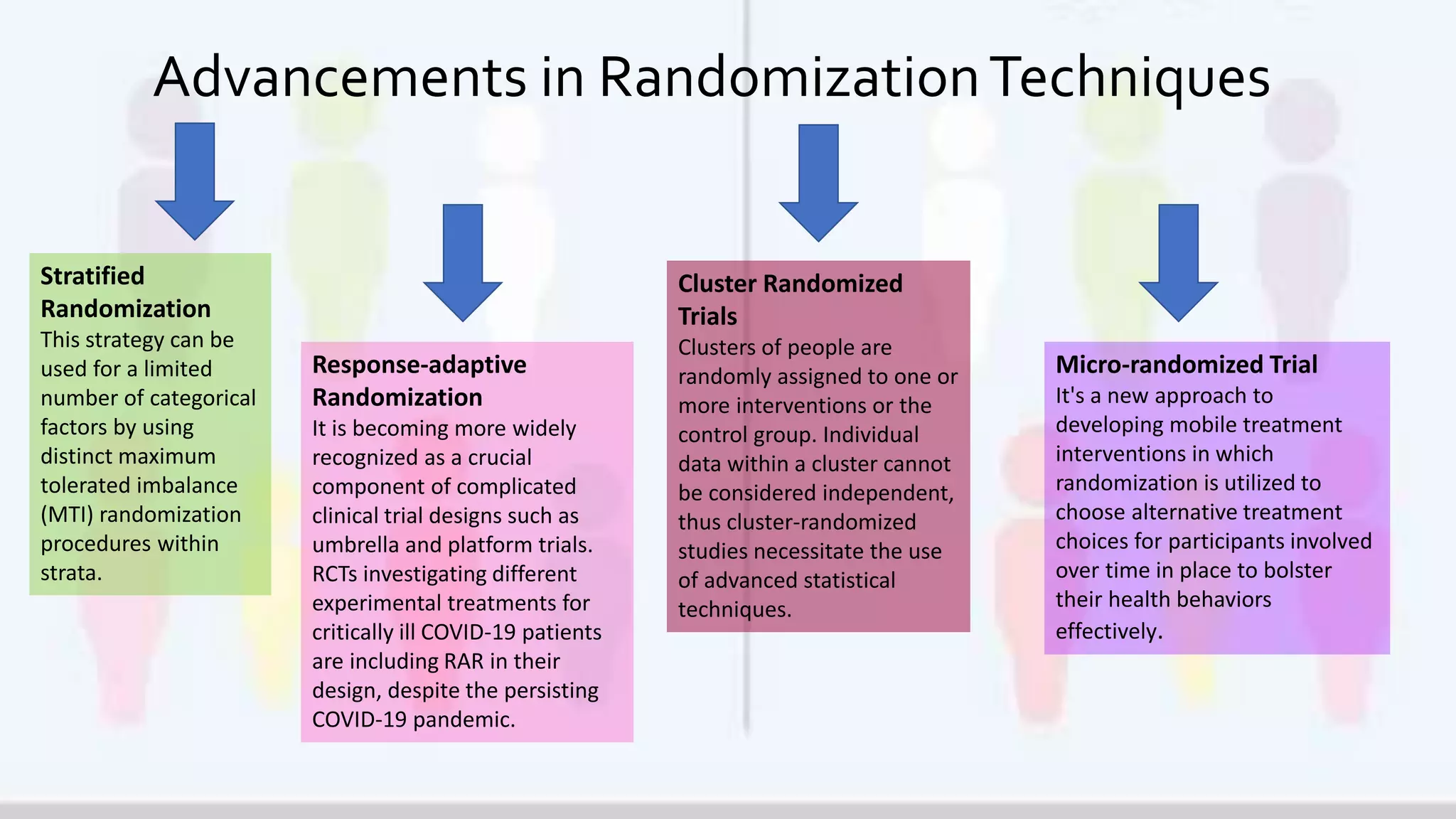
The document outlines the significance of randomization in clinical trials, emphasizing its role in establishing the clinical efficacy of treatments through randomized controlled trials (RCTs). It details various randomization methodologies, including complete randomization and stratified randomization, as well as highlights the challenges such as biases and the importance of proper study design for reliable results. Ultimately, RCTs are deemed essential for generating high-quality data critical in guiding evidence-based medicine and regulatory approvals.