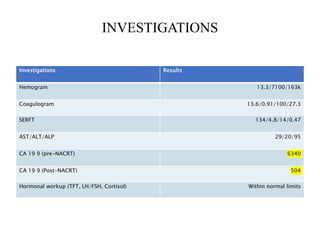
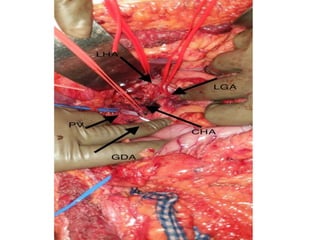
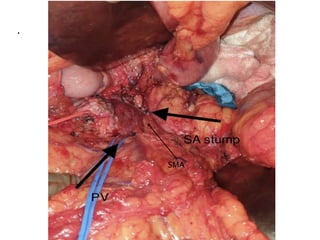
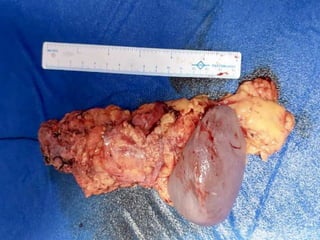
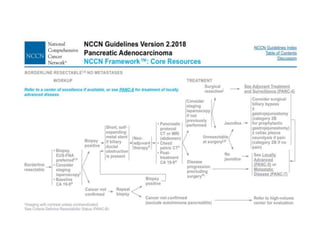
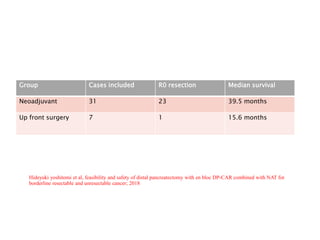
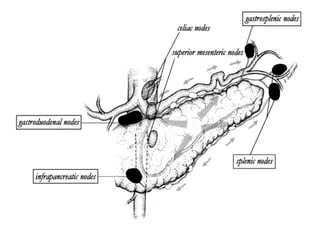
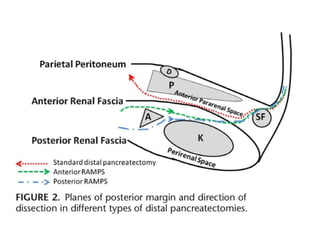
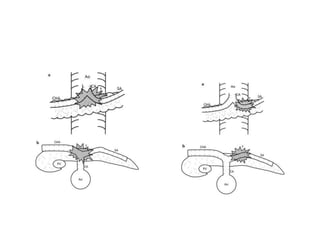
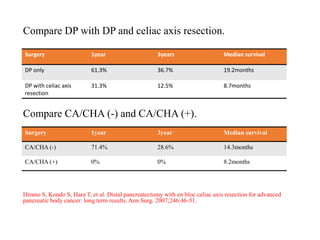
This document provides details of a case of a 45-year-old male diagnosed with locally advanced distal pancreatic cancer who underwent neoadjuvant chemotherapy and radiation followed by radical antegrade modular pancreatosplenectomy (RAMPS). The summary discusses the patient's history, diagnostic workup, treatment course, surgical approach including RAMPS technique, histopathology results, and conclusions about RAMPS being a good approach for distal pancreatic tumors with benefits like better lymph node yield and margin negative resection rates compared to other approaches.