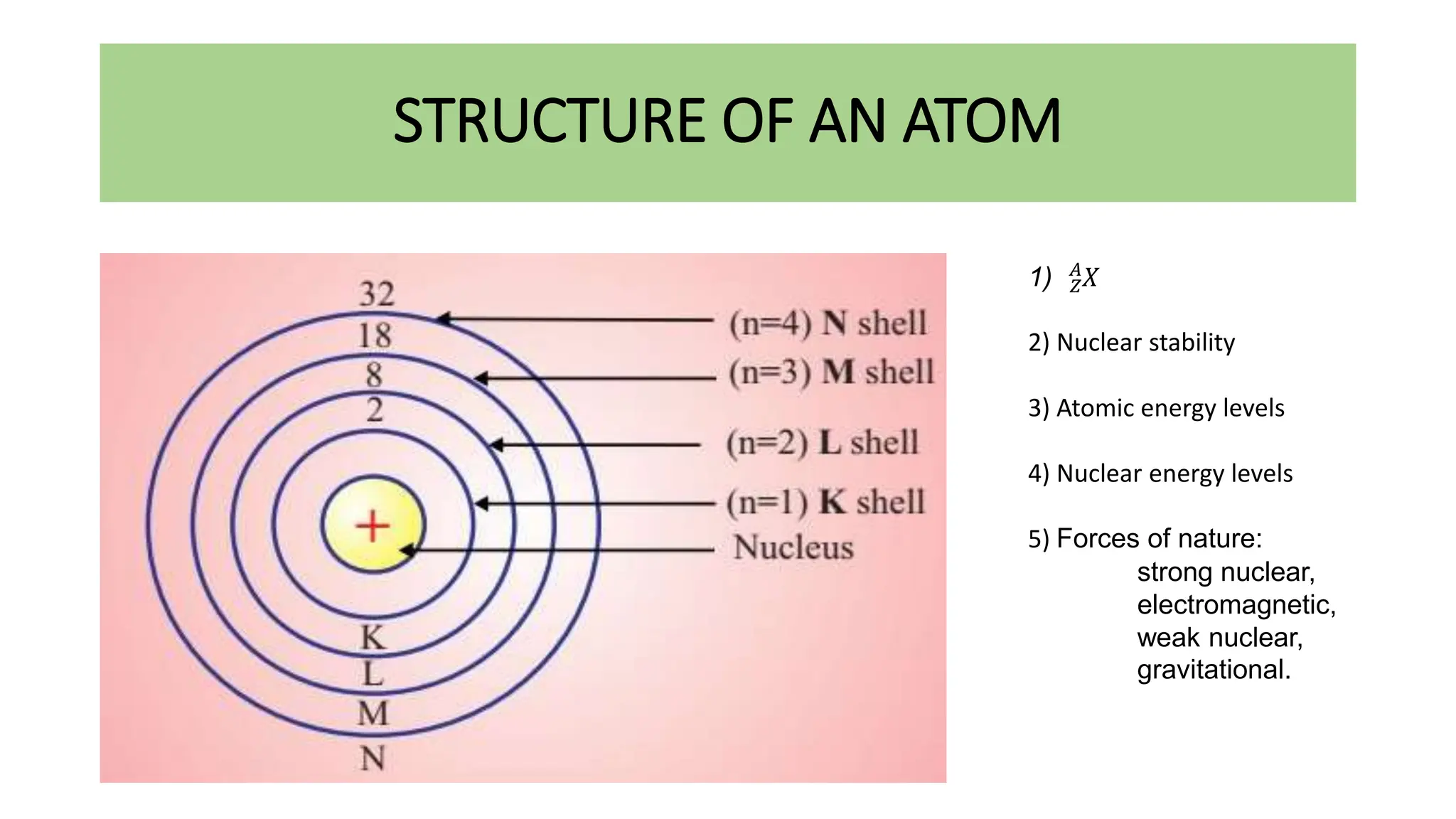
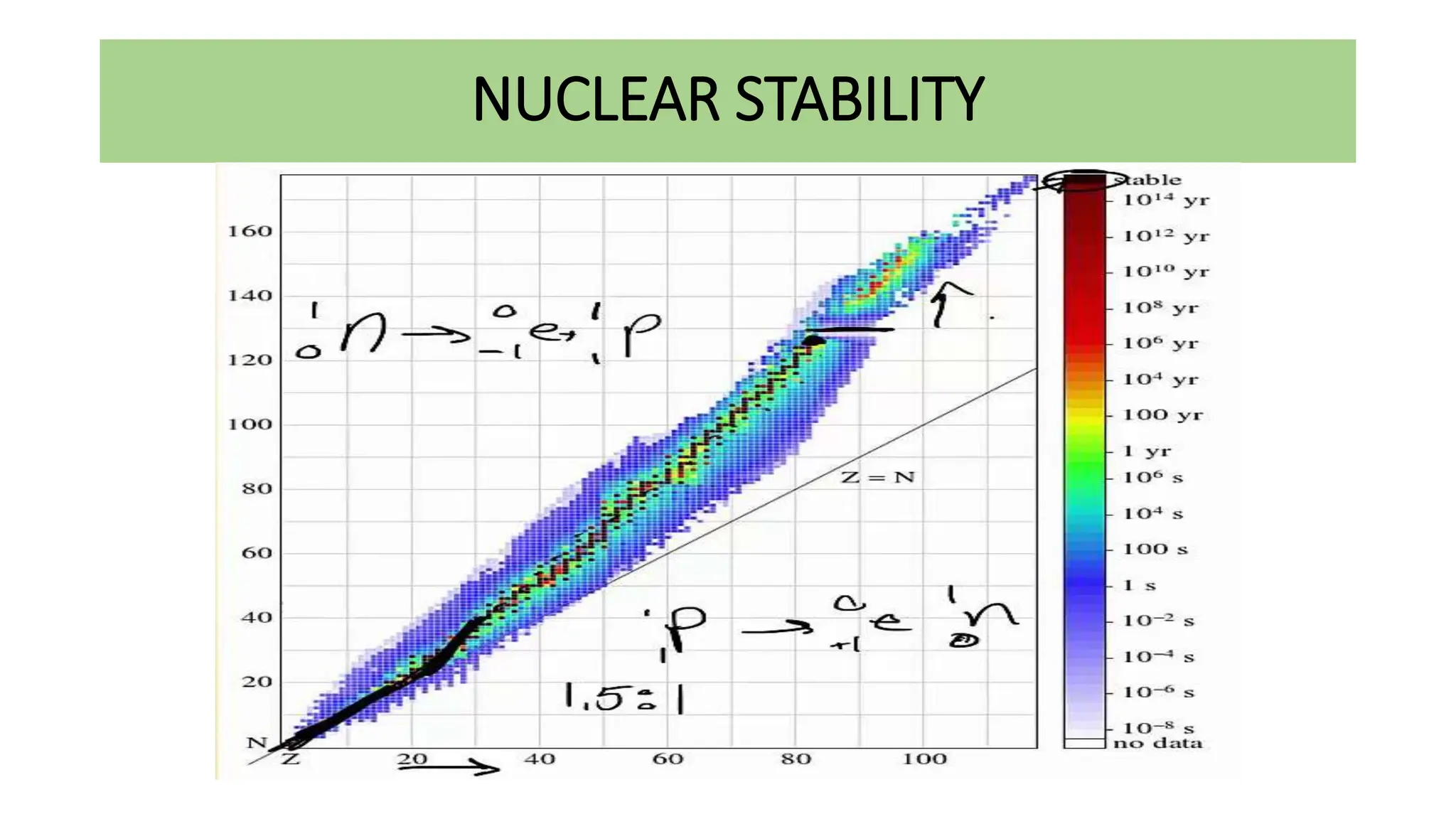
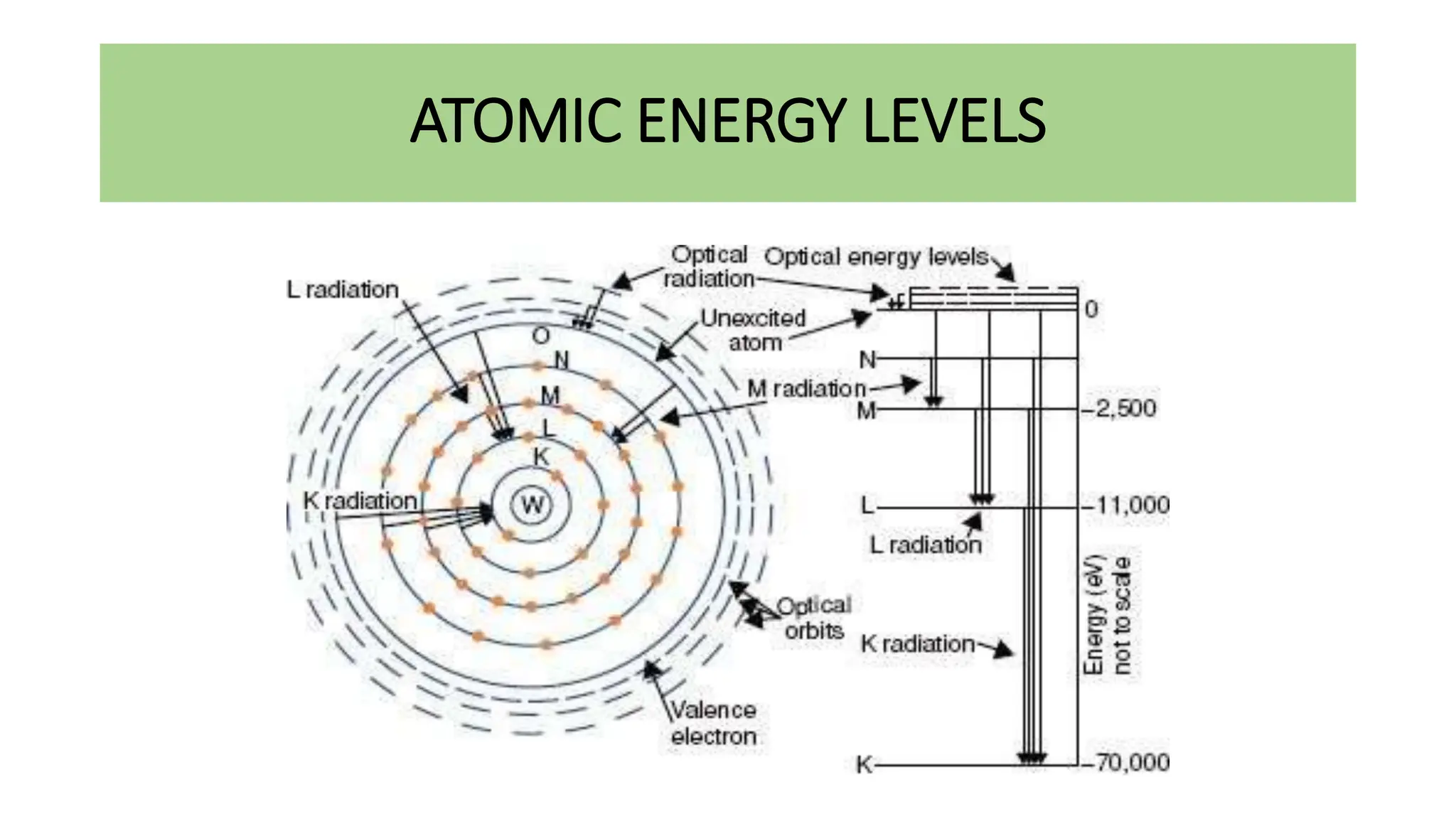
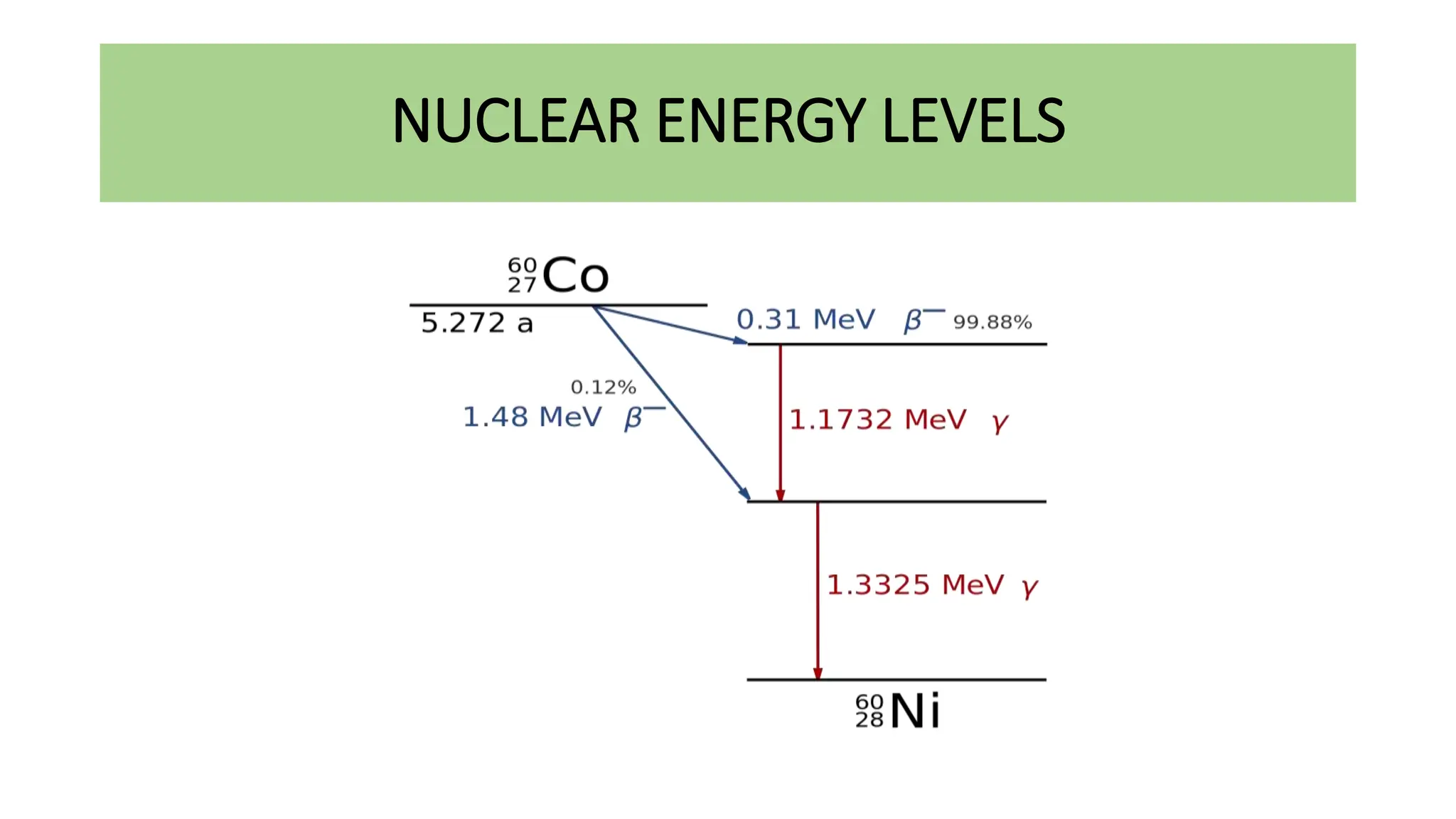
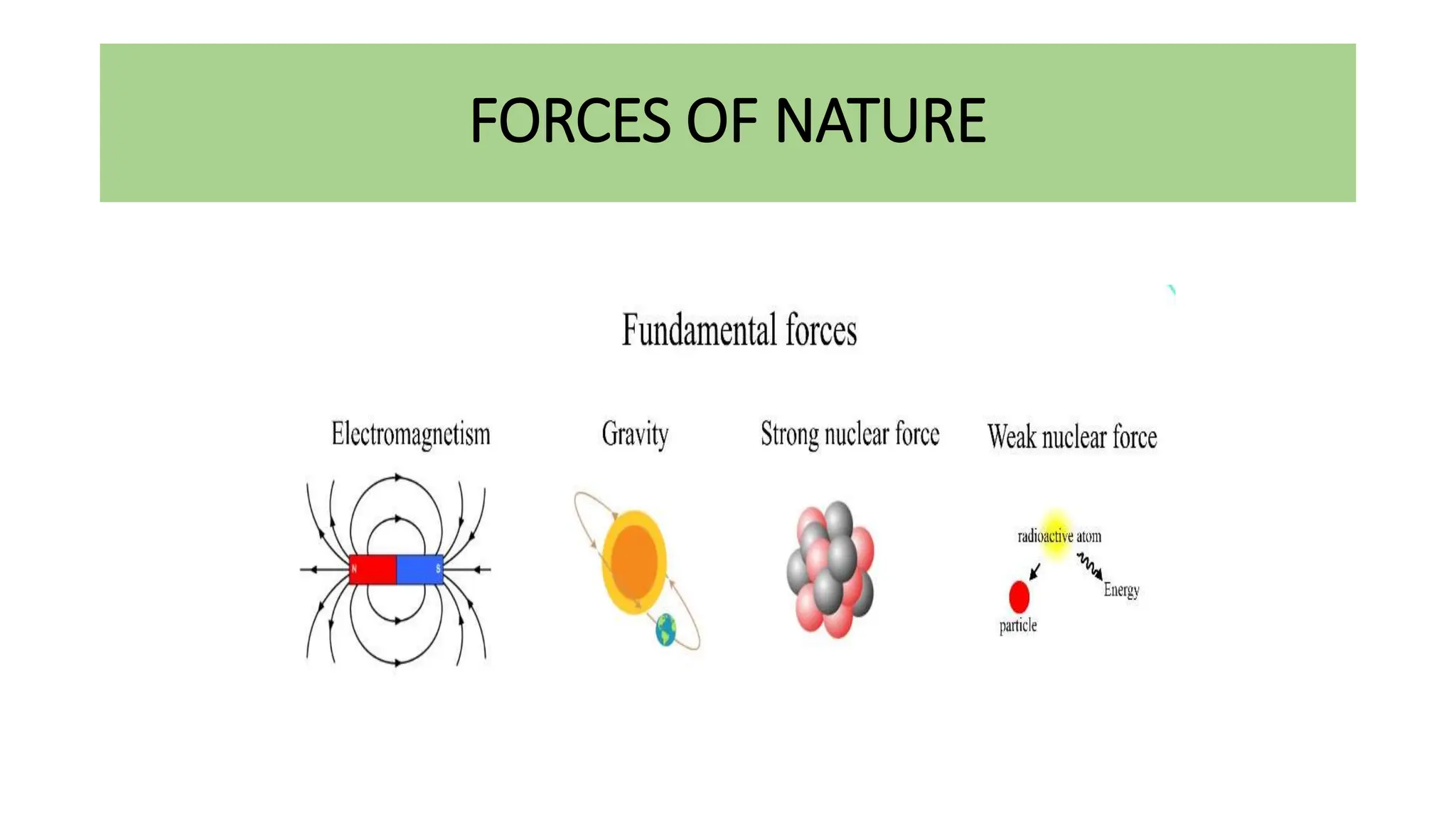
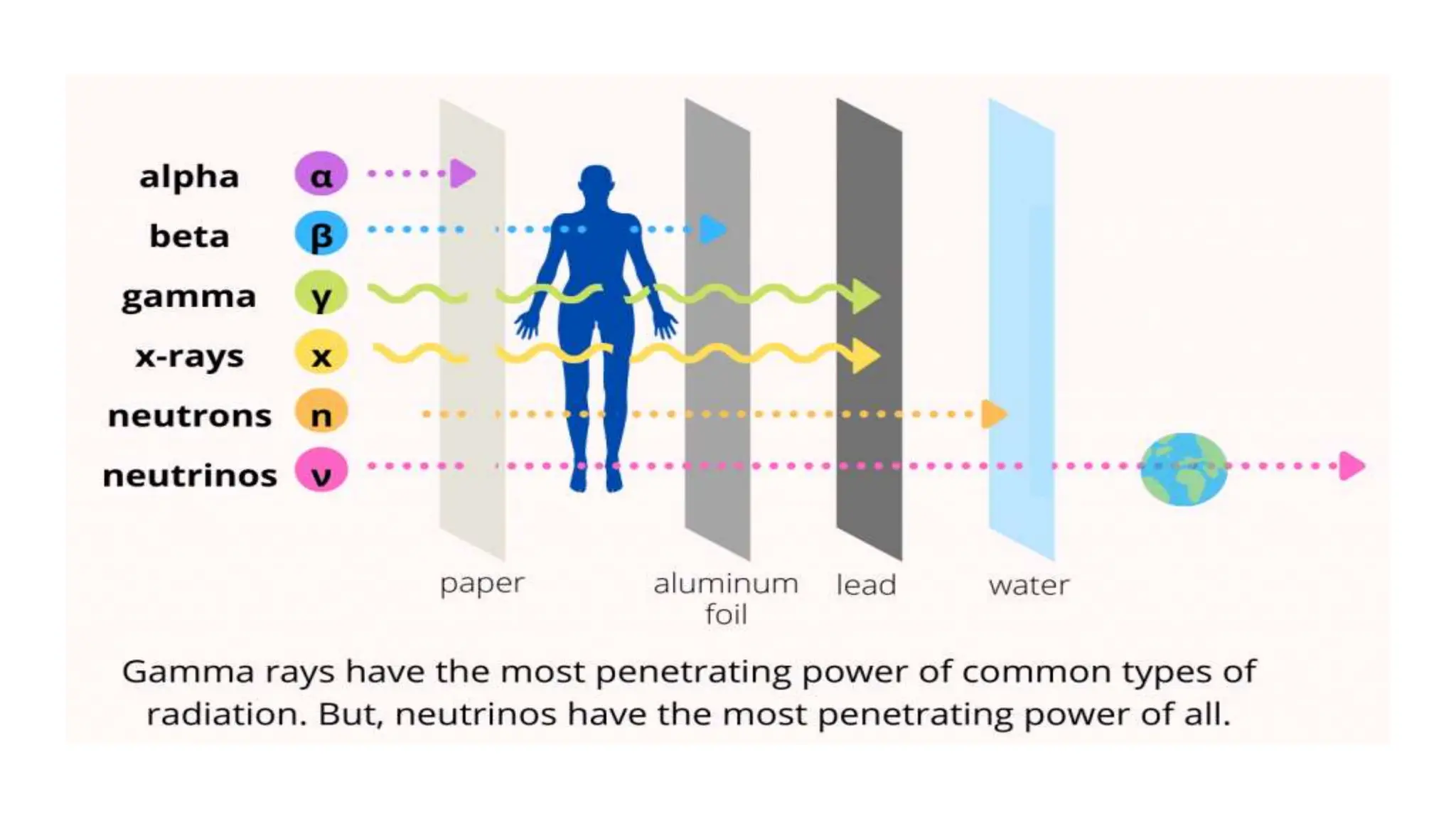
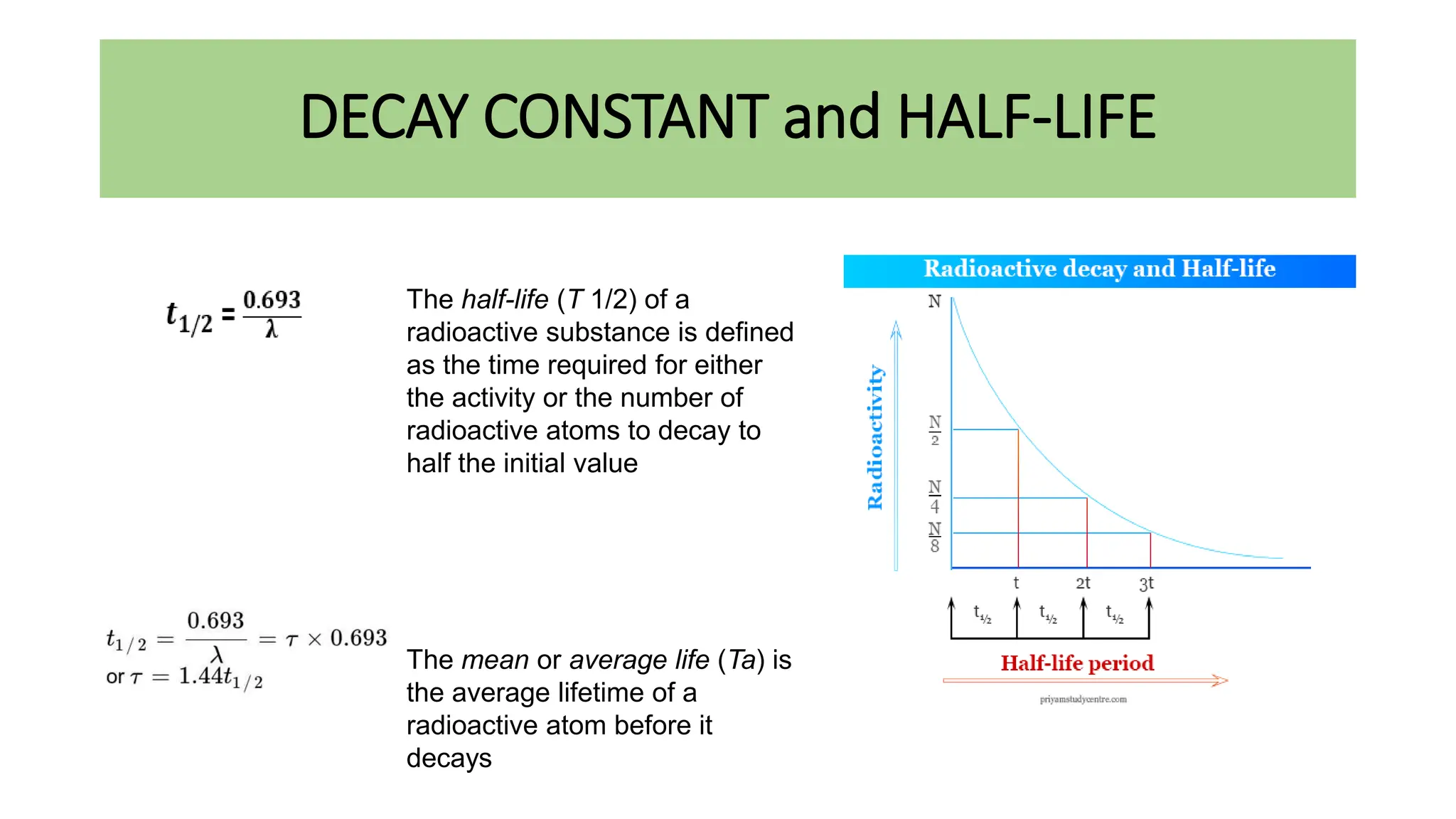
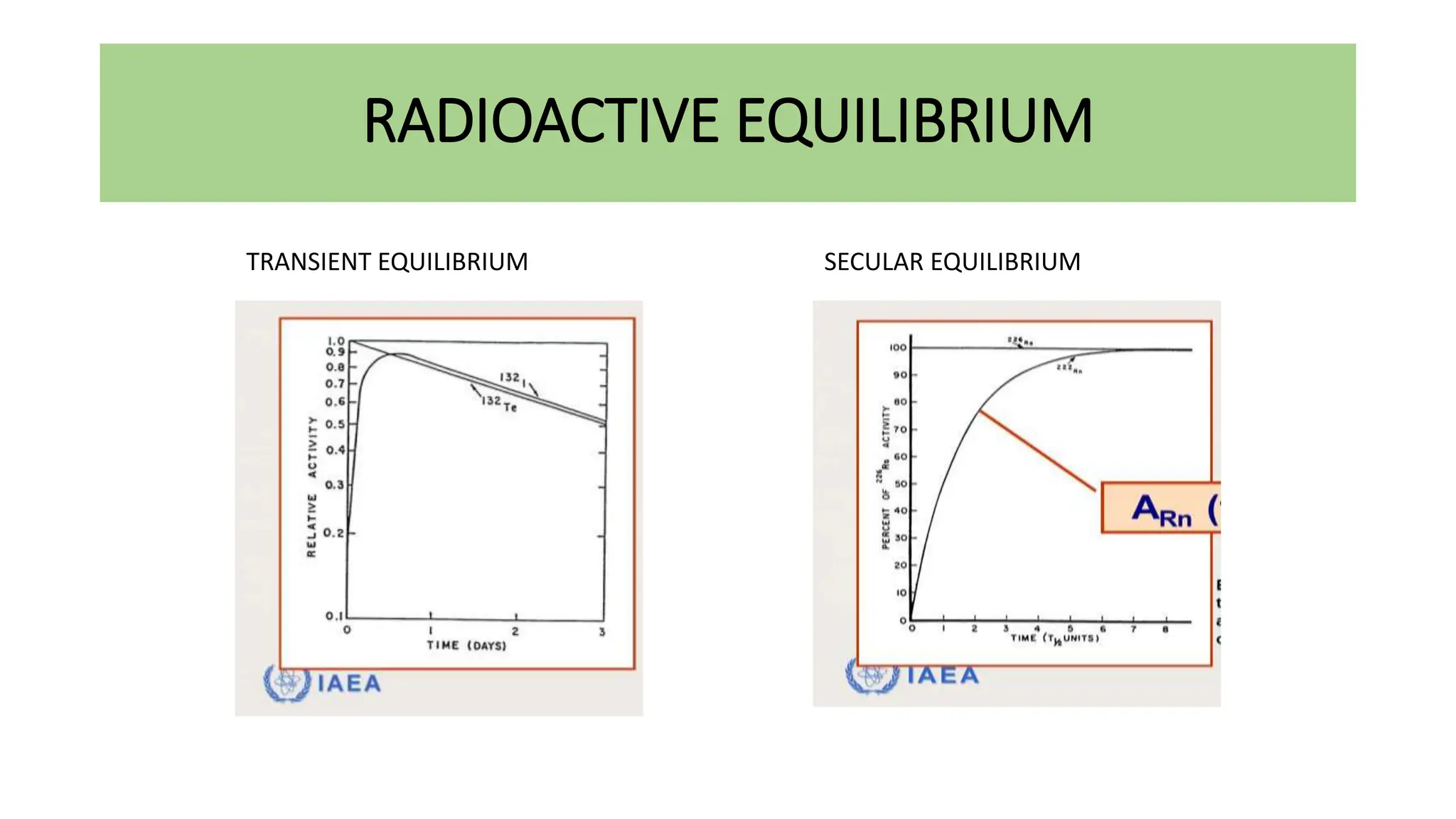
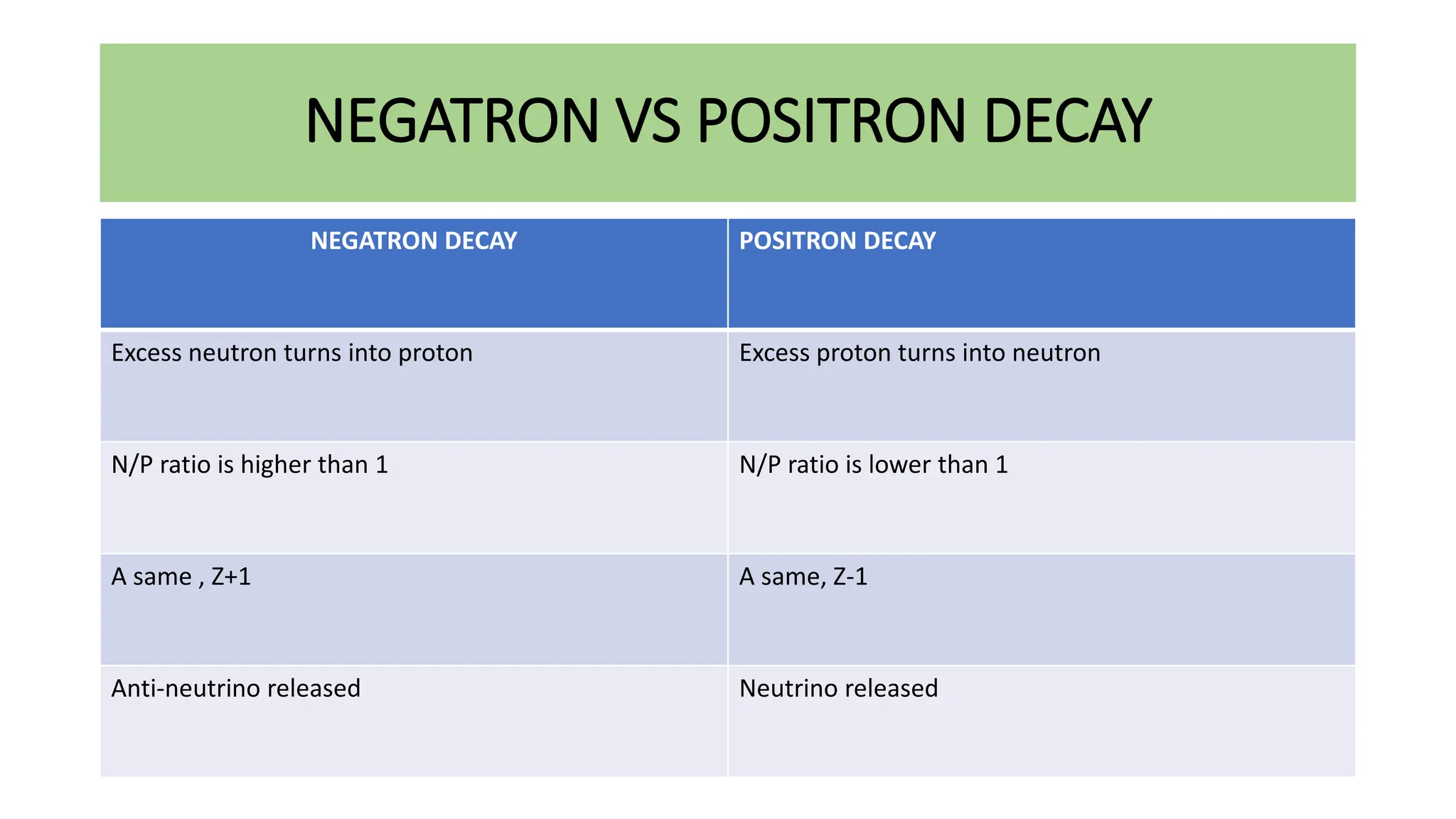
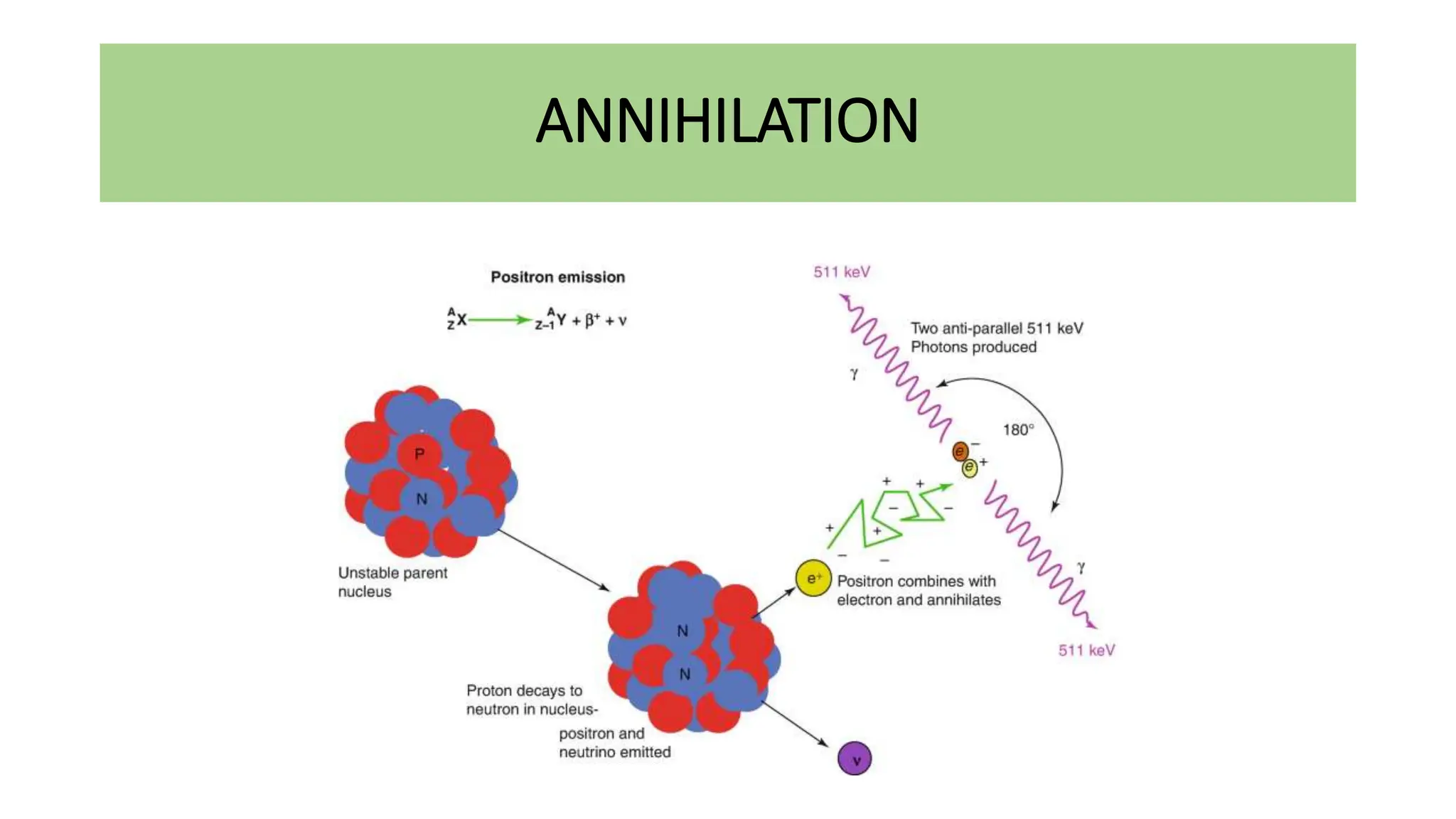
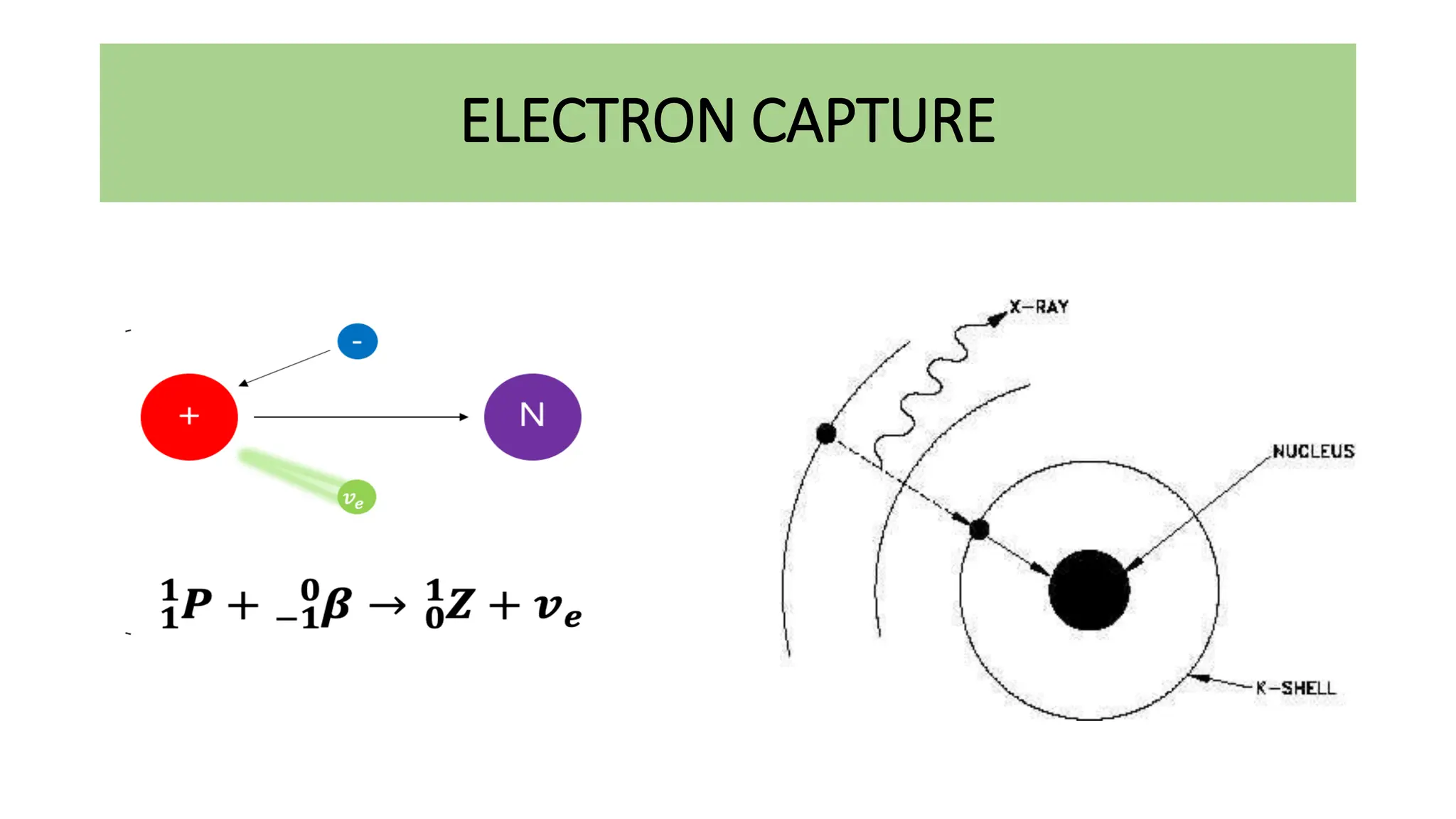
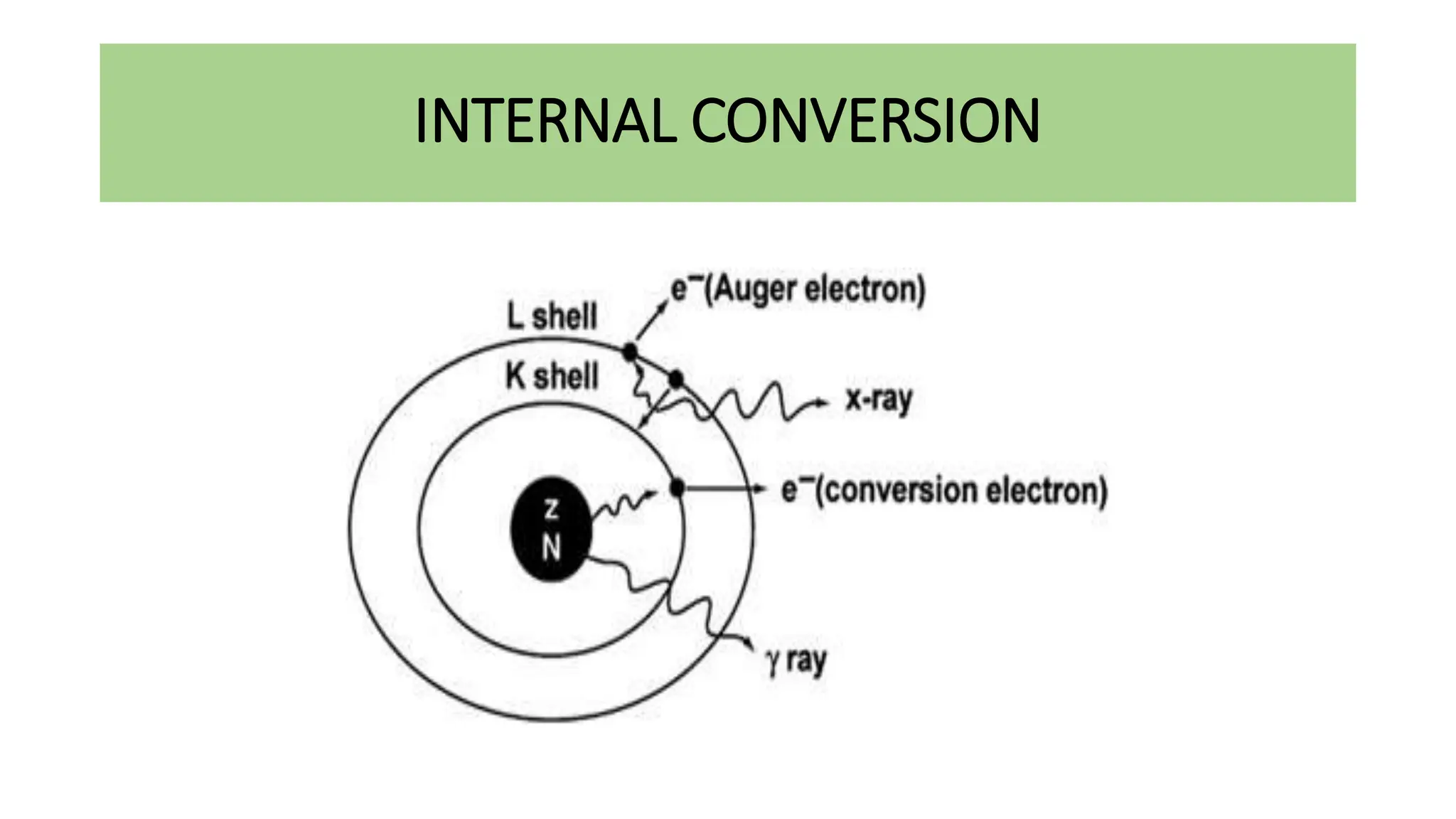
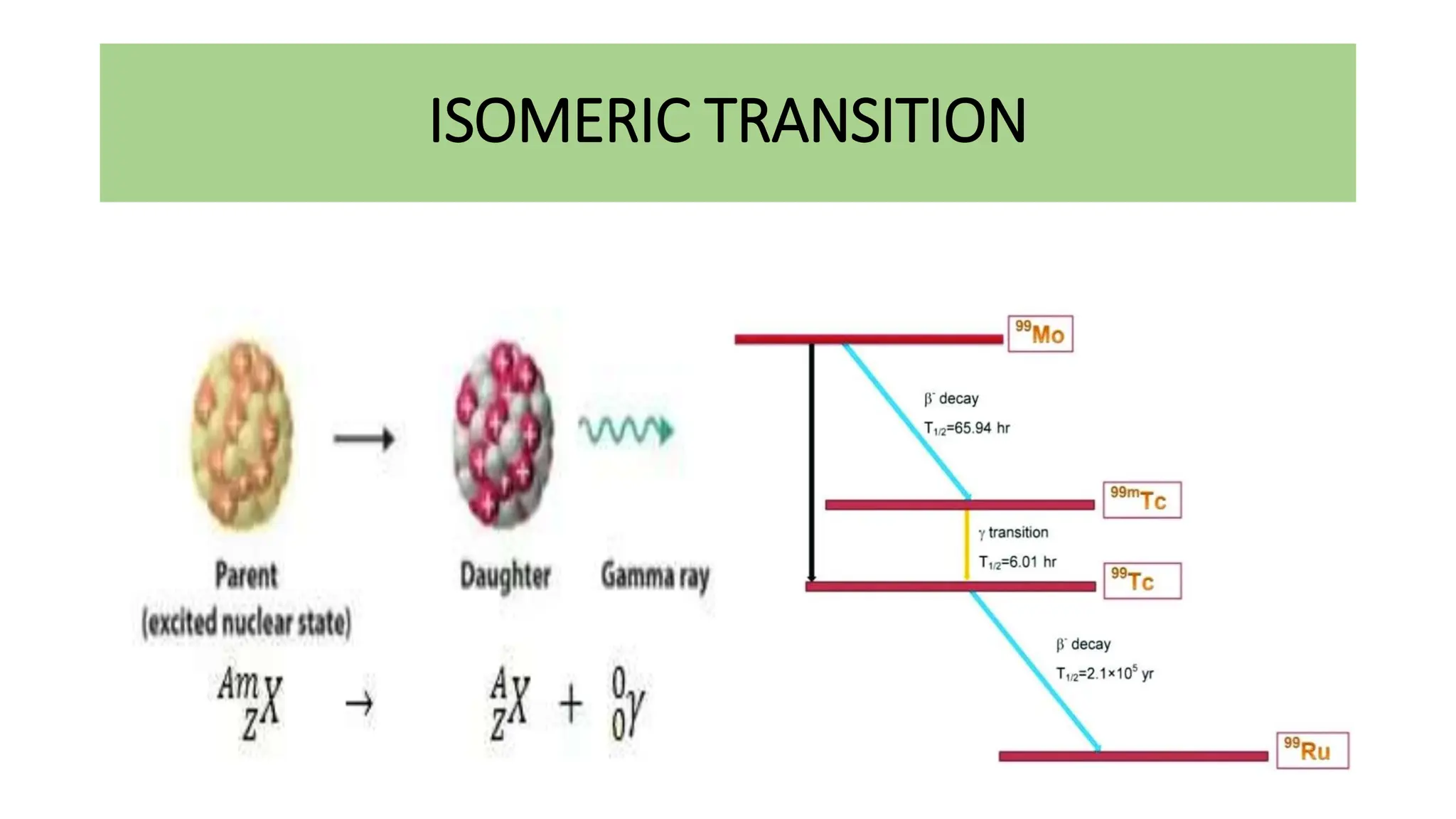
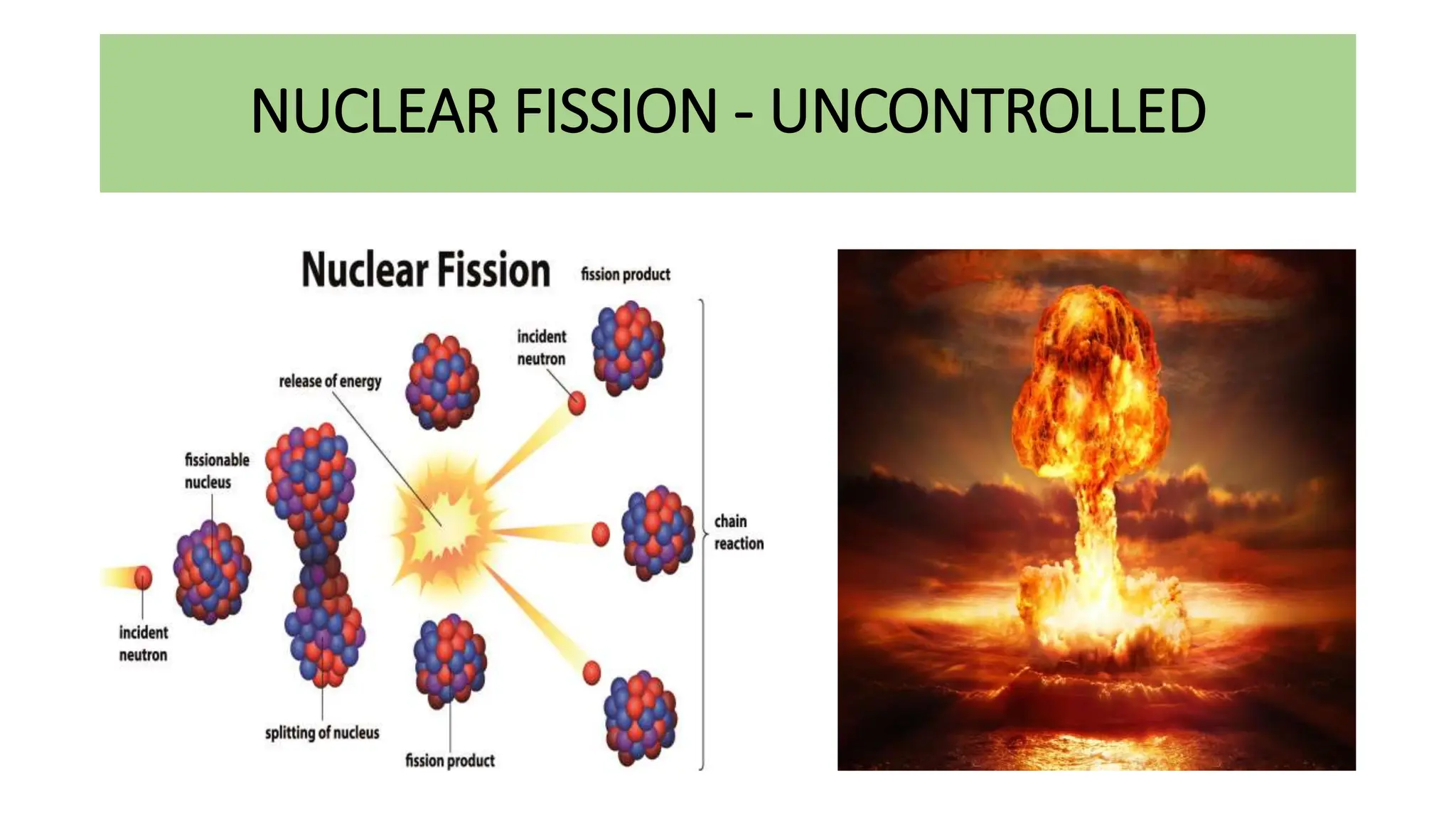
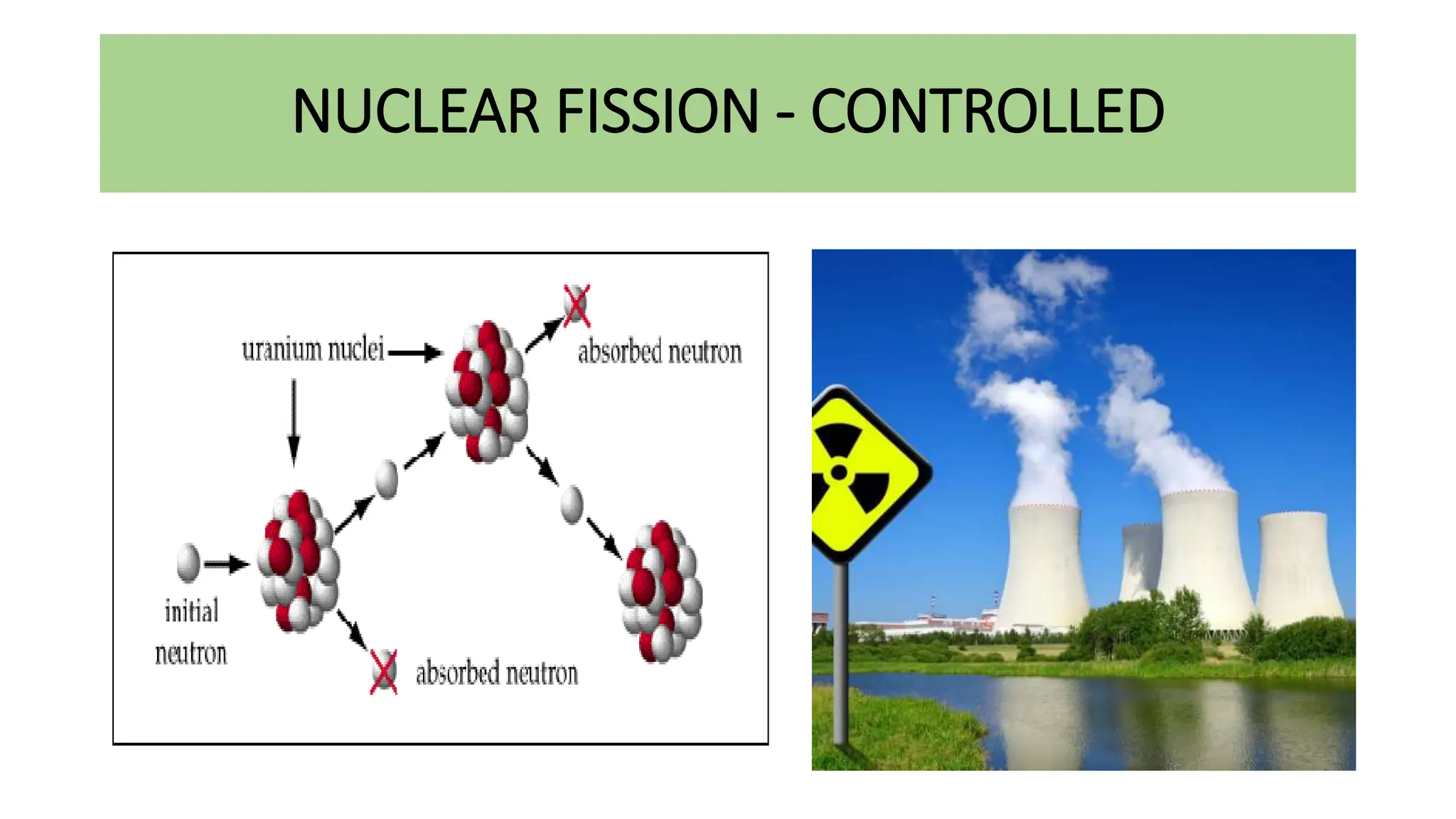
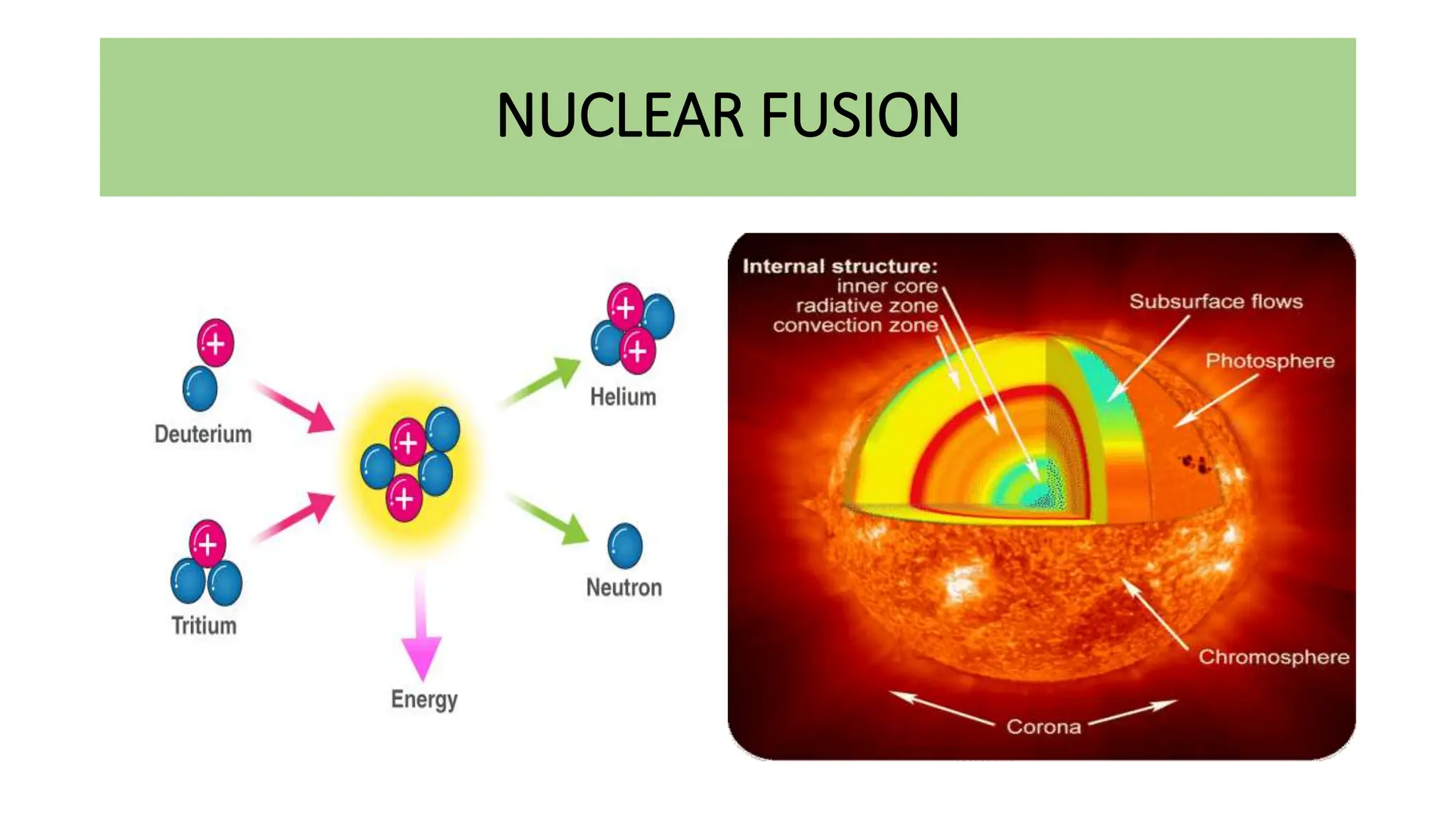
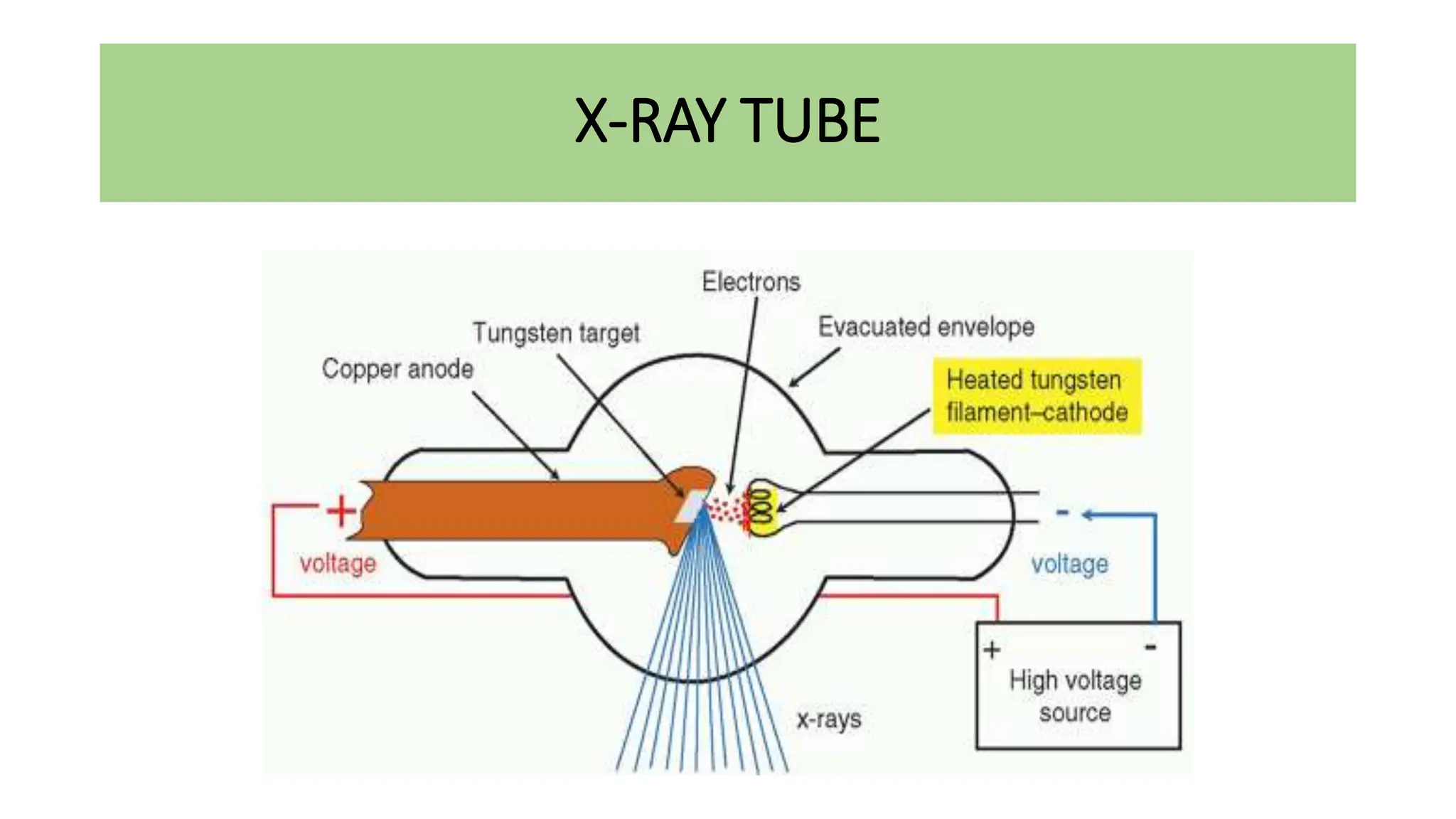
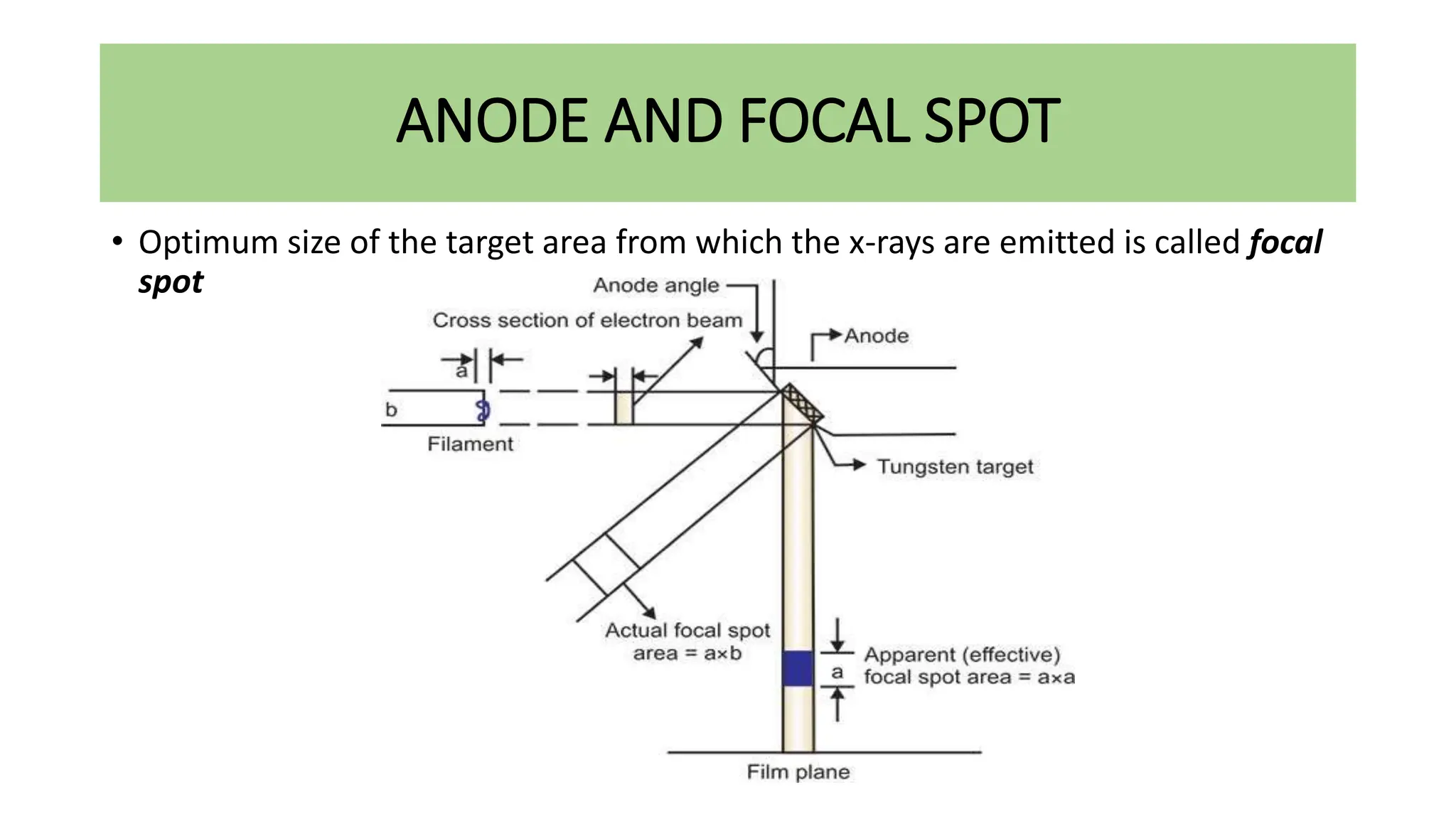
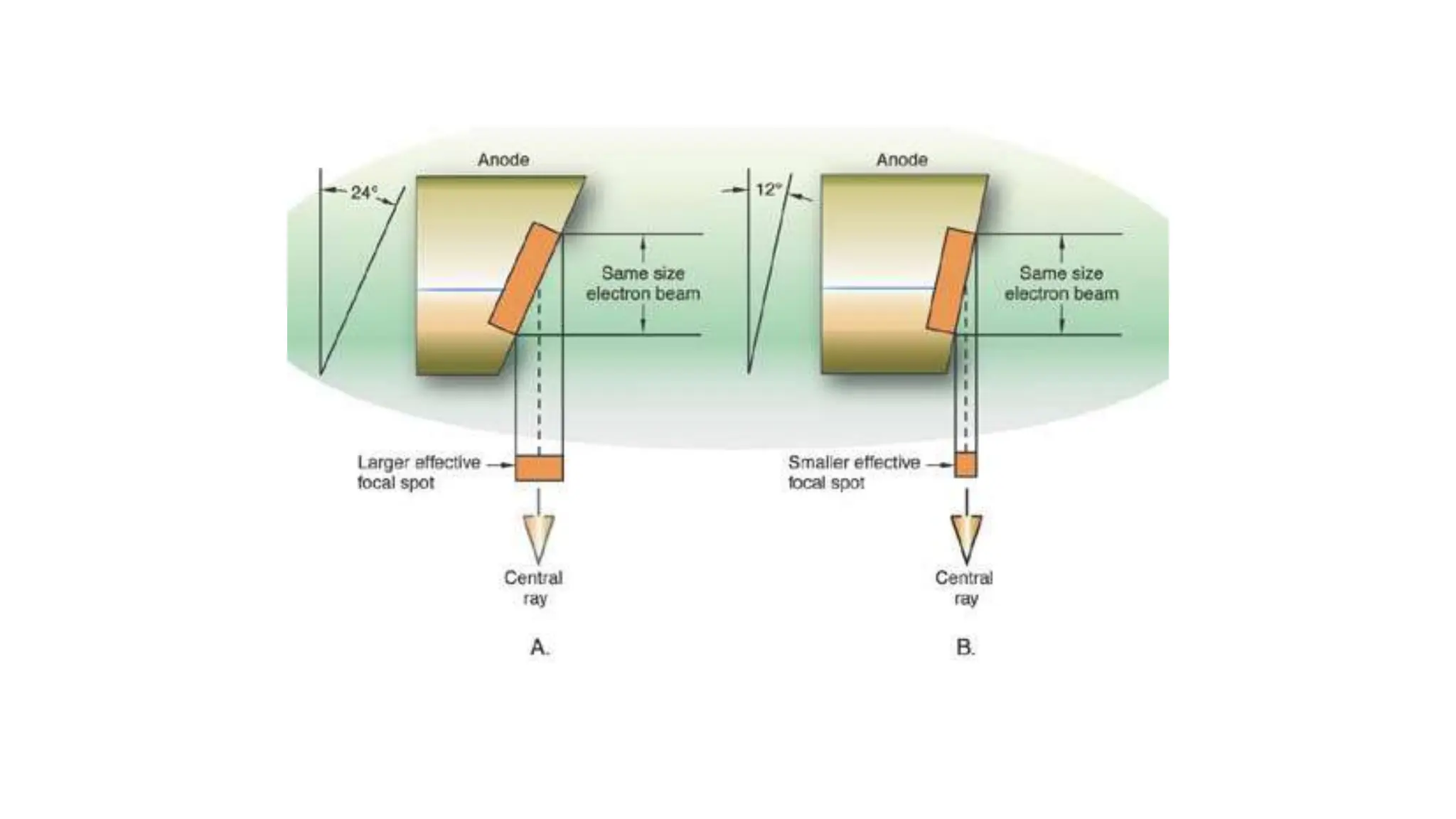
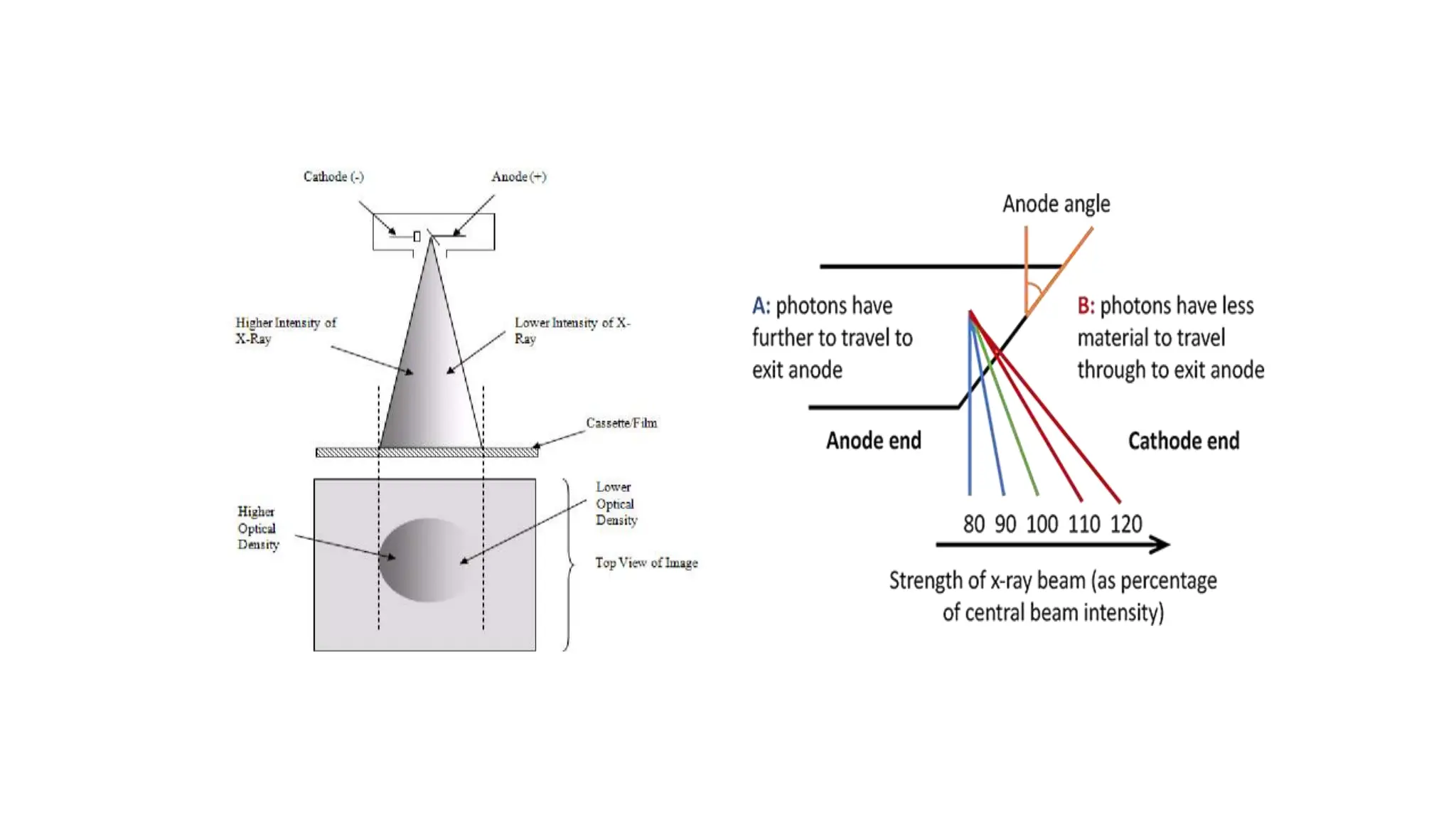
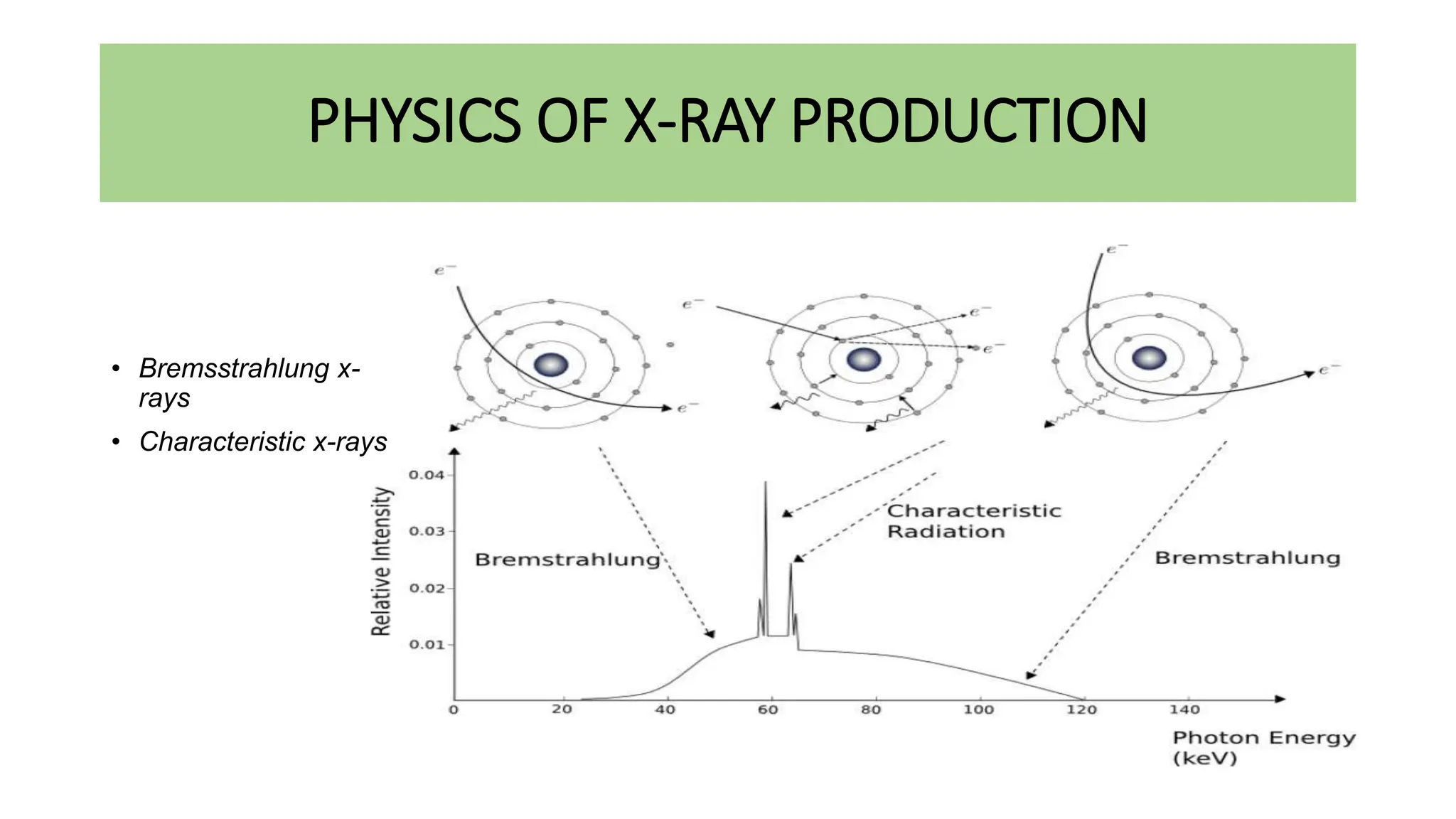
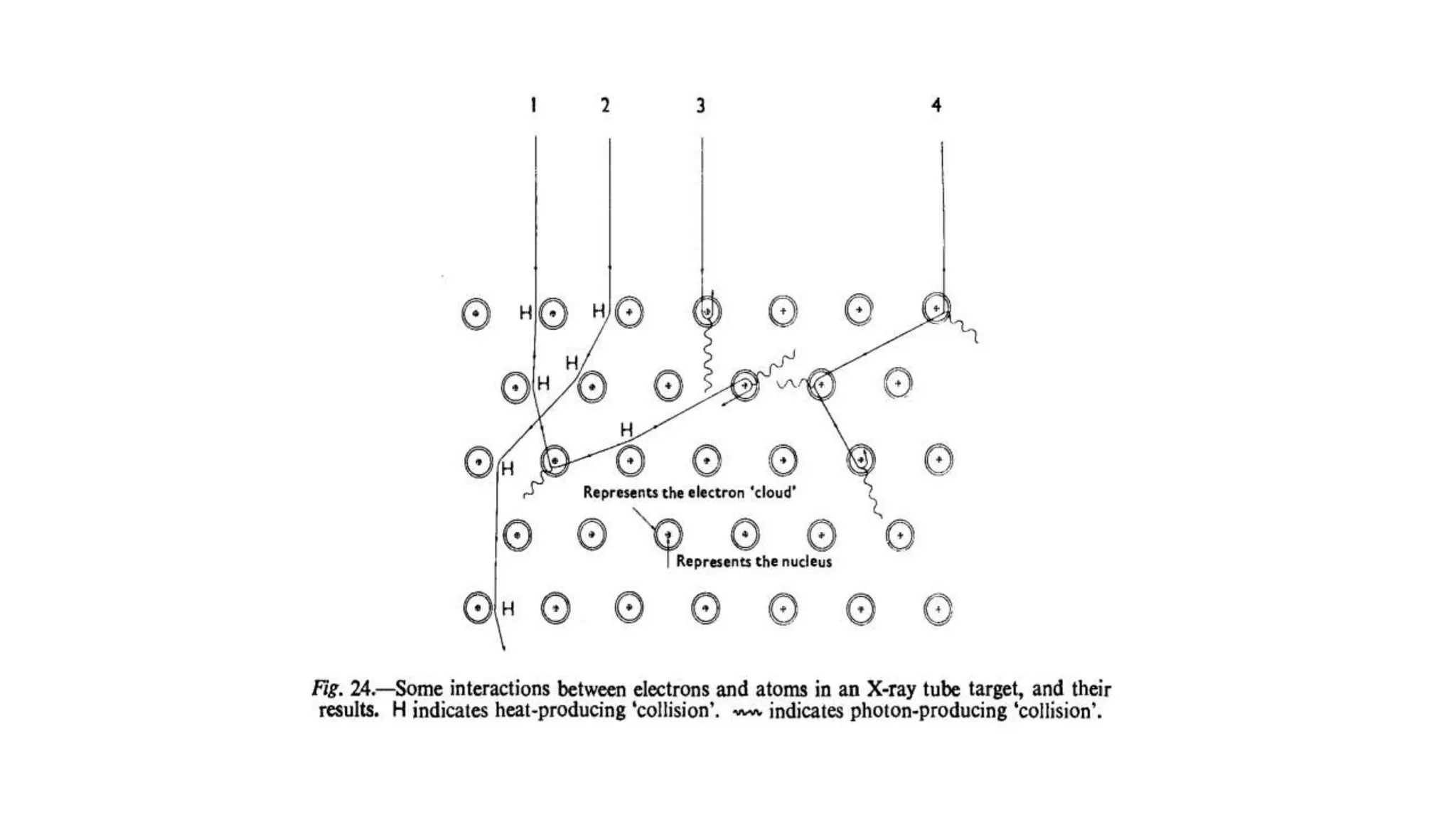
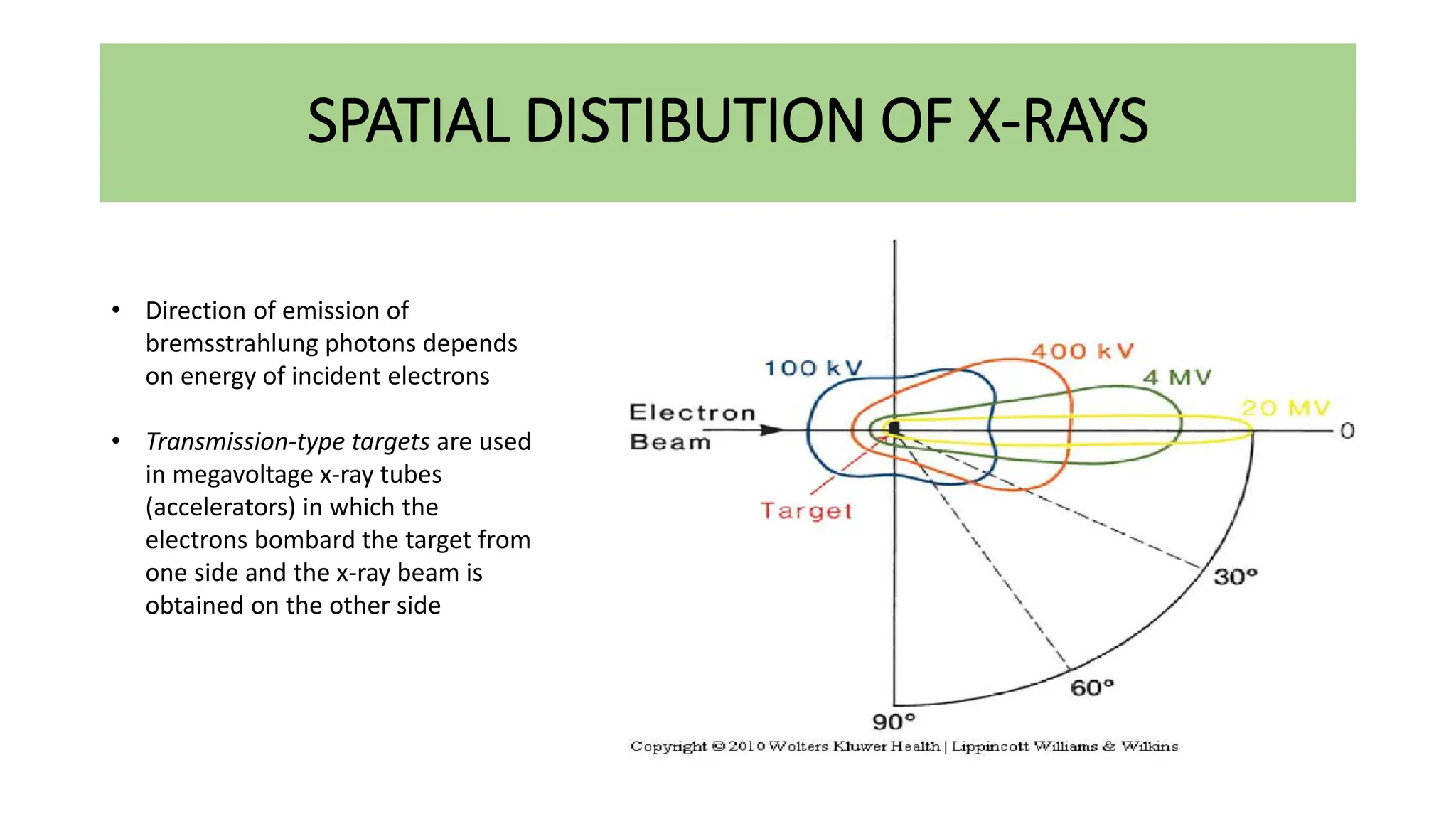
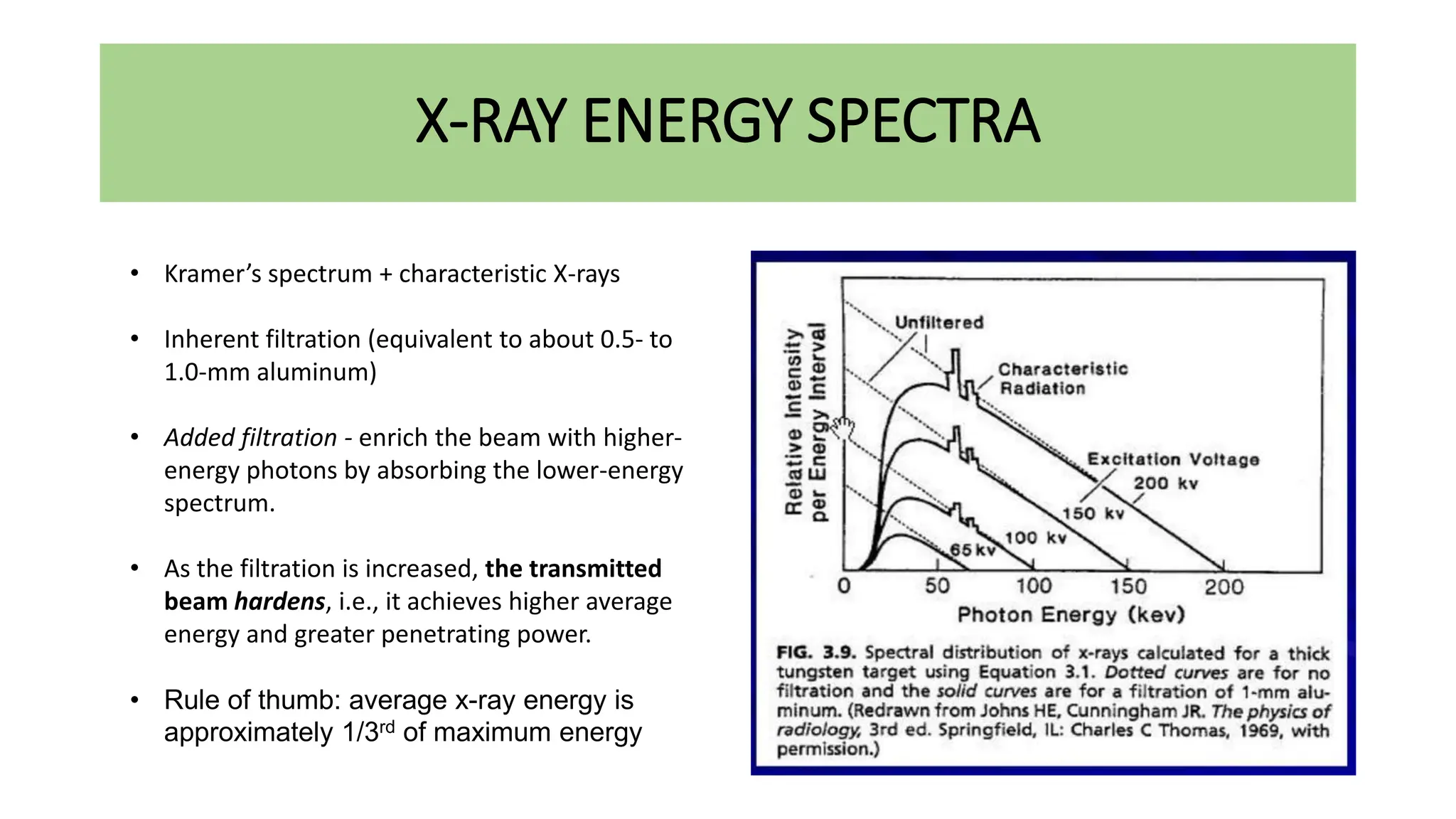
This document discusses radioactivity and the production of x-rays. It begins by describing the structure of atoms and different types of radioactive decay such as alpha, beta, and gamma rays. It then discusses concepts like decay constant and half-life. Different modes of radioactive decay like alpha and beta particle decay are explained. The document also covers nuclear reactions, fission, fusion, and how x-rays are produced when high-speed electrons interact with matter. It provides details on the components of an x-ray tube and the physics behind bremsstrahlung and characteristic x-ray production.