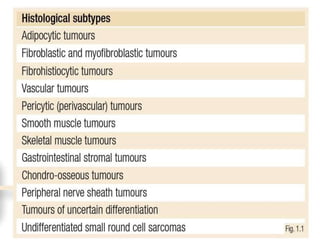
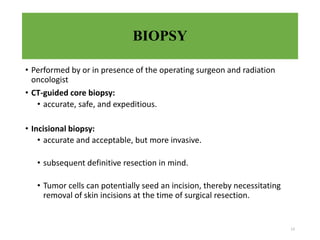
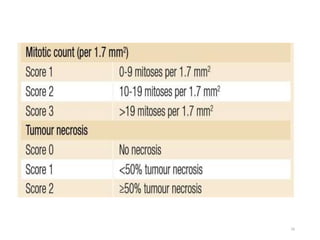
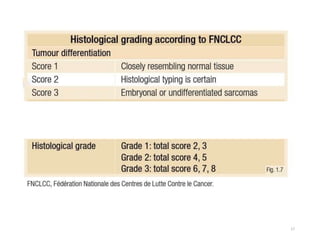
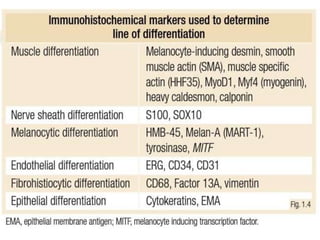
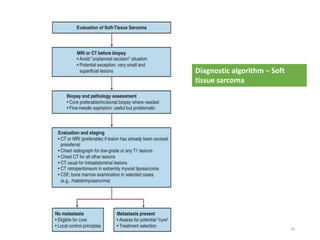
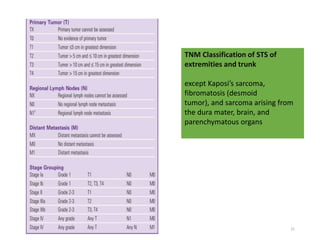
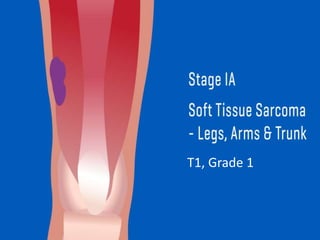
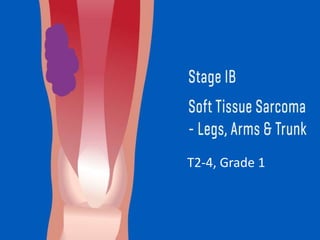
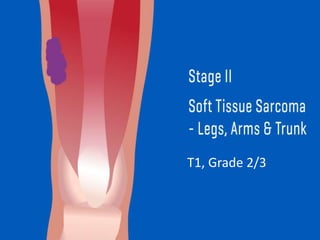
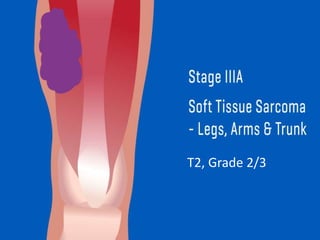
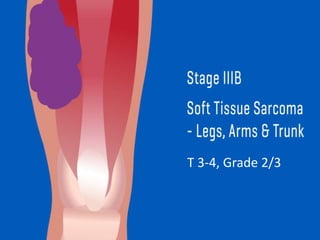
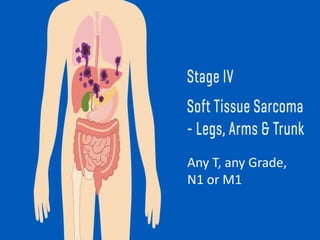
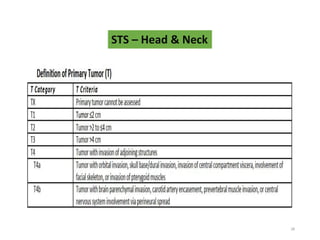
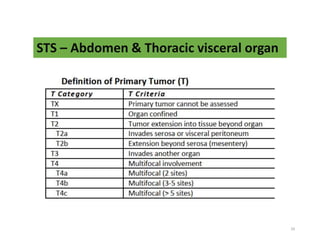
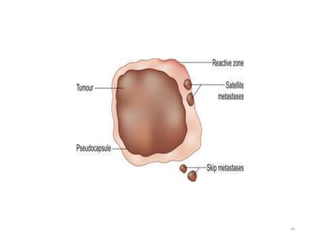
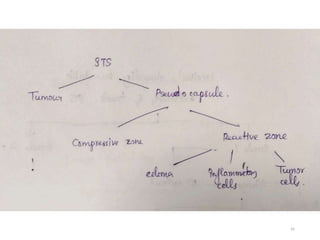
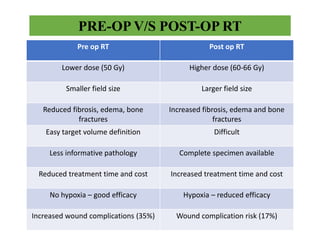
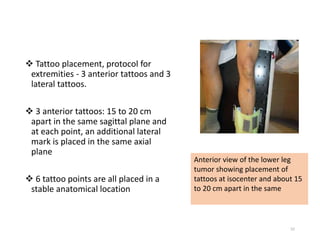
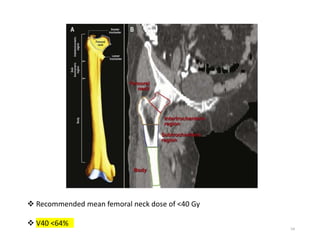
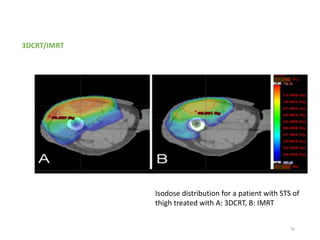
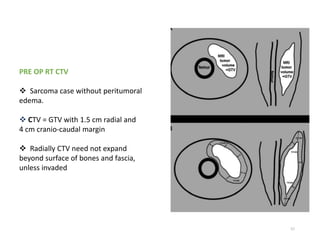
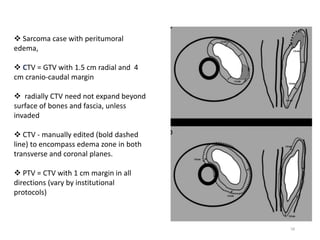
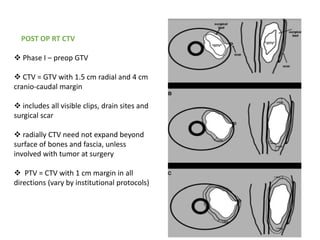
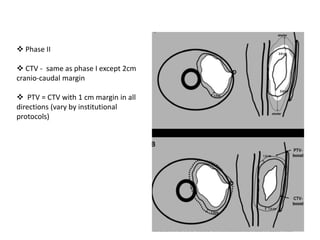
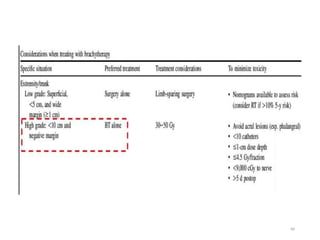
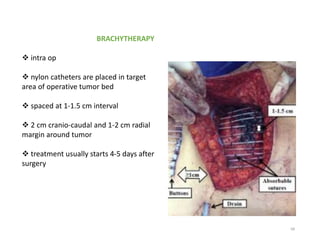
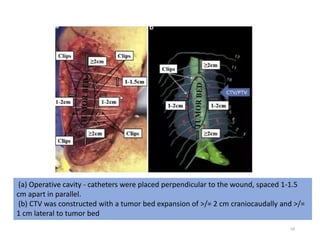
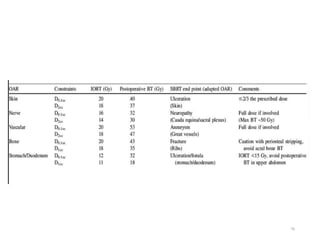
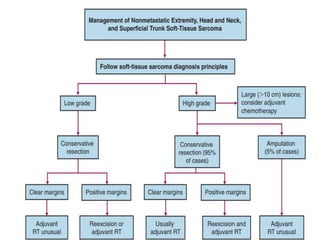
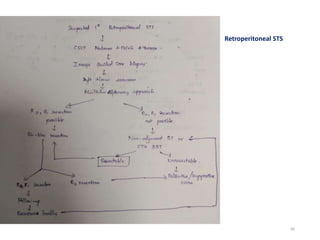
This document provides information on the management of soft tissue sarcoma, including epidemiology, pathology, etiology, clinical manifestations, diagnosis, staging, prognostic factors, and treatment options. It discusses that soft tissue sarcomas most commonly occur in the extremities, with the three most common histologic types being undifferentiated pleomorphic sarcomas, liposarcomas, and leiomyosarcomas. The mainstay of treatment is surgery, with the goal of wide excision and negative margins. For high grade tumors, radiation therapy is often recommended pre- or post-operatively to improve local control. Treatment requires a multidisciplinary approach and careful consideration of tumor factors, margins, and functional outcomes