Question Analysis Of Paper 3 Trial Chemistry from Different State
•Download as DOCX, PDF•
2 likes•2,107 views
Report
Share
Report
Share
Recommended
Question Analysis for Paper 3 Trial Chemistry 2011 from Different States

It provides a clear analysis of questions in Paper 3 Trial SPM 2011 from different state
Spinel type lithium cobalt oxide as a bifunctional electrocatalyst for oxygen...

Development of efficient, affordable electrocatalysts for the oxygen evolution reaction and
the oxygen reduction reaction is critical for rechargeable metal-air batteries. Here we present
lithium cobalt oxide, synthesized at 400 C (designated as LT-LiCoO2) that adopts a lithiated
spinel structure, as an inexpensive, efficient electrocatalyst for the oxygen evolution reaction.
The catalytic activity of LT-LiCoO2 is higher than that of both spinel cobalt oxide and layered
lithium cobalt oxide synthesized at 800 C (designated as HT-LiCoO2) for the oxygen evolution
reaction. Although LT-LiCoO2 exhibits poor activity for the oxygen reduction reaction,
the chemically delithiated LT-Li1xCoO2 samples exhibit a combination of high oxygen
reduction reaction and oxygen evolution reaction activities, making the spinel-type LTLi0,5CoO2
a potential bifunctional electrocatalyst for rechargeable metal-air batteries. The
high activities of these delithiated compositions are attributed to the Co4O4 cubane subunits
and a pinning of the Co3þ/4þ:3d energy with the top of the O2:2p band.
Research Paper Presentation by Ariful Islam

Department of Chemistry, Khulna University of Engineering and Technology (KUET), Khulna, Bangladesh
Recommended
Question Analysis for Paper 3 Trial Chemistry 2011 from Different States

It provides a clear analysis of questions in Paper 3 Trial SPM 2011 from different state
Spinel type lithium cobalt oxide as a bifunctional electrocatalyst for oxygen...

Development of efficient, affordable electrocatalysts for the oxygen evolution reaction and
the oxygen reduction reaction is critical for rechargeable metal-air batteries. Here we present
lithium cobalt oxide, synthesized at 400 C (designated as LT-LiCoO2) that adopts a lithiated
spinel structure, as an inexpensive, efficient electrocatalyst for the oxygen evolution reaction.
The catalytic activity of LT-LiCoO2 is higher than that of both spinel cobalt oxide and layered
lithium cobalt oxide synthesized at 800 C (designated as HT-LiCoO2) for the oxygen evolution
reaction. Although LT-LiCoO2 exhibits poor activity for the oxygen reduction reaction,
the chemically delithiated LT-Li1xCoO2 samples exhibit a combination of high oxygen
reduction reaction and oxygen evolution reaction activities, making the spinel-type LTLi0,5CoO2
a potential bifunctional electrocatalyst for rechargeable metal-air batteries. The
high activities of these delithiated compositions are attributed to the Co4O4 cubane subunits
and a pinning of the Co3þ/4þ:3d energy with the top of the O2:2p band.
Research Paper Presentation by Ariful Islam

Department of Chemistry, Khulna University of Engineering and Technology (KUET), Khulna, Bangladesh
Chemistry Test for FMDC

Chemistry Sample Question paper of Multiple Choice Questions (MCQs) Test for FMDC
INVESTIGATE THE PROPERTIES OF IONIC BOND AND COVALENT BOND THROUGH AN EXPERIMENT

CHEMISTRY FORM 4 KSSM
CHAPTER 5 : CHEMICAL BONDS (IONIC BOND AND COVALENT BOND)
EXPERIMENT 5.1 TO INVESTIGATE THE PROPERTIES OF IONIC BOND AND COVALENT BOND THROUGH EXPERIMENT
Surface reactions and performance of non-aqueous electrolytes with lithium me...

Six electrolyteswere investigated for lithium metal battery applications. The electrolyteswere composed
of combinations of four different salts (LiPF6, LiB(C2O4)2, LiI and LiN(SO2CF3)2) and three different solvents
(PC, DME, and 1,3-dioxolane). All six electrolytes had conductivities >3mScm−1 at temperatures
from −20 to 40 ◦C. Electrochemical impedance spectroscopy (EIS) and linear polarization, both at room
temperature and low temperature (−8 ◦C), provided congruent results. The LiI-based electrolyte had the
lowest film resistance, while 0.7M LiB(C2O4)2–PC:DME (1:1) had the highest impedance. The presence
of 1,3-dioxolane in electrolytes provided lower impedance with LiB(C2O4)2 but higher resistance with
LiPF6-based electrolytes. NMR analysis of electrolytes after thermal abuse indicate that LiN(SO2CF3)2-
based electrolytes are the most thermally stable. SEM analysis suggests that surface modification and
impedance changes are correlated.
© 2008 Elsevier B.V.
Journal of power_sources_volume_185_issue_2_2008
Oxide film growth on copper in neutral aqueous solutions

International peer-reviewed academic journals call for papers, http://www.iiste.org
Organometallic Reactions and Catalysis

Organometallic compounds undergo a rich variety of reactions (oxidative addition, reductive elimination, cyclometalization, migratory insertion, carbonylation, hydrometallation hydrate elimination, etc ) that can sometimes be combined into useful homogeneous catalytic cycles. In this presentation, I have discussed organometallic reactions of particular importance for synthetic and catalytic processes like the oxo process (hydroformylation), heck coupling reaction, Wilkinson’s Catalyst
(Hydrogenation) etc.
IRJET- Equilibrium Studies on Ternary Metal Complexes of Drug Ethambutol Hydr...

https://www.irjet.net/archives/V6/i8/IRJET-V6I8235.pdf
Synthesis, Characterization and antimicrobial activity of some novel sulfacet...

IOSR Journal of Applied Chemistry (IOSR-JAC) is a double blind peer reviewed International Journal that provides rapid publication (within a month) of articles in all areas of applied chemistry and its applications. The journal welcomes publications of high quality papers on theoretical developments and practical applications in Chemical Science. Original research papers, state-of-the-art reviews, and high quality technical notes are invited for publications.
Jawapan Module Perfect Score Chemistry SPM 2013

Jawapan Module Perfect Score Chemistry SPM 2013, untuk rujukan guru dan pelajar.
More Related Content
What's hot
Chemistry Test for FMDC

Chemistry Sample Question paper of Multiple Choice Questions (MCQs) Test for FMDC
INVESTIGATE THE PROPERTIES OF IONIC BOND AND COVALENT BOND THROUGH AN EXPERIMENT

CHEMISTRY FORM 4 KSSM
CHAPTER 5 : CHEMICAL BONDS (IONIC BOND AND COVALENT BOND)
EXPERIMENT 5.1 TO INVESTIGATE THE PROPERTIES OF IONIC BOND AND COVALENT BOND THROUGH EXPERIMENT
Surface reactions and performance of non-aqueous electrolytes with lithium me...

Six electrolyteswere investigated for lithium metal battery applications. The electrolyteswere composed
of combinations of four different salts (LiPF6, LiB(C2O4)2, LiI and LiN(SO2CF3)2) and three different solvents
(PC, DME, and 1,3-dioxolane). All six electrolytes had conductivities >3mScm−1 at temperatures
from −20 to 40 ◦C. Electrochemical impedance spectroscopy (EIS) and linear polarization, both at room
temperature and low temperature (−8 ◦C), provided congruent results. The LiI-based electrolyte had the
lowest film resistance, while 0.7M LiB(C2O4)2–PC:DME (1:1) had the highest impedance. The presence
of 1,3-dioxolane in electrolytes provided lower impedance with LiB(C2O4)2 but higher resistance with
LiPF6-based electrolytes. NMR analysis of electrolytes after thermal abuse indicate that LiN(SO2CF3)2-
based electrolytes are the most thermally stable. SEM analysis suggests that surface modification and
impedance changes are correlated.
© 2008 Elsevier B.V.
Journal of power_sources_volume_185_issue_2_2008
Oxide film growth on copper in neutral aqueous solutions

International peer-reviewed academic journals call for papers, http://www.iiste.org
Organometallic Reactions and Catalysis

Organometallic compounds undergo a rich variety of reactions (oxidative addition, reductive elimination, cyclometalization, migratory insertion, carbonylation, hydrometallation hydrate elimination, etc ) that can sometimes be combined into useful homogeneous catalytic cycles. In this presentation, I have discussed organometallic reactions of particular importance for synthetic and catalytic processes like the oxo process (hydroformylation), heck coupling reaction, Wilkinson’s Catalyst
(Hydrogenation) etc.
IRJET- Equilibrium Studies on Ternary Metal Complexes of Drug Ethambutol Hydr...

https://www.irjet.net/archives/V6/i8/IRJET-V6I8235.pdf
Synthesis, Characterization and antimicrobial activity of some novel sulfacet...

IOSR Journal of Applied Chemistry (IOSR-JAC) is a double blind peer reviewed International Journal that provides rapid publication (within a month) of articles in all areas of applied chemistry and its applications. The journal welcomes publications of high quality papers on theoretical developments and practical applications in Chemical Science. Original research papers, state-of-the-art reviews, and high quality technical notes are invited for publications.
What's hot (19)
INVESTIGATE THE PROPERTIES OF IONIC BOND AND COVALENT BOND THROUGH AN EXPERIMENT

INVESTIGATE THE PROPERTIES OF IONIC BOND AND COVALENT BOND THROUGH AN EXPERIMENT
Surface reactions and performance of non-aqueous electrolytes with lithium me...

Surface reactions and performance of non-aqueous electrolytes with lithium me...
8 ppt v2_chemistry_10_regularity of changes in acid-base properties of compou...

8 ppt v2_chemistry_10_regularity of changes in acid-base properties of compou...
Oxide film growth on copper in neutral aqueous solutions

Oxide film growth on copper in neutral aqueous solutions
IRJET- Equilibrium Studies on Ternary Metal Complexes of Drug Ethambutol Hydr...

IRJET- Equilibrium Studies on Ternary Metal Complexes of Drug Ethambutol Hydr...
“Kinetics and mechanism of sulphuric acid oxidation of glycolic (ga) by selen...

“Kinetics and mechanism of sulphuric acid oxidation of glycolic (ga) by selen...
Synthesis, Characterization and antimicrobial activity of some novel sulfacet...

Synthesis, Characterization and antimicrobial activity of some novel sulfacet...
Viewers also liked
Jawapan Module Perfect Score Chemistry SPM 2013

Jawapan Module Perfect Score Chemistry SPM 2013, untuk rujukan guru dan pelajar.
Golden Notes - Chemistry - Akshansh

MY BEST COLLECTION
My Notes contain the following:
+1 and +2 Chemistry Formulas,
Organic chemistry,
The best way to learn,
IIT Preparation,
BITSAT,
AIEEE
Find more study guides on:
www.akshansh.weebly.com
Perfect Score Chemistry SPM 2013

Module Perfect Score Chemistry SPM 2013, untuk rujukan guru dan pelajar.
Latihan kendiri Kimia Pertengahan Tahun Tingkatan 4

Latihan kendiri Pertengahan Tahun Tingkatan 4.
Untuk rujukan guru dan pelajar.
Viewers also liked (20)
Guidelines on answering paper 2 and paper 3 questions

Guidelines on answering paper 2 and paper 3 questions
Latihan kendiri Kimia Pertengahan Tahun Tingkatan 4

Latihan kendiri Kimia Pertengahan Tahun Tingkatan 4
Similar to Question Analysis Of Paper 3 Trial Chemistry from Different State
Madkour 1985-journal of-chemical_technology_and_biotechnology._chemical_techn...

Recommended Flowsheets for the Electrolytic Extraction of Lead and Zinc from Red Sea Polymetal Ore
The polymetal complex ore Umm-Gheig considered in Egypt as a rather rich source of lead and zinc is subjected to mineralogical, chemical, spectral, X-ray and differential thermal analyses. Hydrometallurgical treatments based on leaching, precipitation and electrodeposition of metal from the ore are established. The influences of current density, temperature and metal ion concentration on the Faradic current efficiency are discussed. Advantages and disadvantages of flow- sheets and various approaches depending on convenient baths for the electro- deposition of metals are investigated. The results of electron microscopic investiga- tion confirmed by metal value data given in the A.S.T.M. cards coincide well with those given by chemical analysis.
Synthesis characterisation_and_evaluation_of_ir_o2_based_binary_metal_oxide_...

Synthesis characterisation_and_evaluation_of_ir_o2_based_binary_metal_oxide_electrocatalysts_for_oxygen_evolution_reaction
Oxide film growth on copper in neutral aqueous solutions

International peer-reviewed academic journals call for papers, http://www.iiste.org
The Cause of Metallic Corrosion in Crude Oils

Metallic corrosion is a severe interrogation regarding most of industrial applications of metals such as the industry of crude oil refining because of the foremost role of metals. The term of corrosion is usually explained as the formation of the metal oxide, sulfide, hydroxide or any compound related with a carboxylic group on the surface due to the impact of the surrounded conditions. The content of sulfur, acidity, mercaptans and salt content are the well ascertained corrosive properties of crude oils also the chemical composition of metals acquit oneself the fundamental preface for the corrosion. The surveillance of the comparison of corrosive capabilities of Murban and Das Blend crude oils in front of seven different types of ferrous metals that used in Sri Lankan refining industry was the dominant intention of this research. The chemical composition of used metals were determined by XRF detector while testing the contents of sulfur, acidities, contents of mercaptans and salt contents with respect to both crude oils simultaneously. Batches of well cleaned and equal sized metal pieces were immersed in both crude oils separately and the corrosion rate of each type of metal piece was determined after 15, 30 and 45 days from the immersion by weight loss method under the observations through the optical microscope. The ferrous and copper concentrations of each crude oil sample were tested while testing the variation of the hardness of each metal as a confirmation stage. According to results there were found relatively higher amount of sulfur, mercaptans, acids and lower amount of salt in Das Blend than Murban although relatively higher corrosion rates of most of metals in the Murban than the Das Blend. The slight reduction of hardness in each metal piece was found while observing significant concentration of ferrous and copper from some of crude oil samples.
THE CAUSE OF METALLIC CORROSION IN CRUDE OILS

Metallic corrosion is a severe interrogation regarding most of industrial applications of metals such as the industry of crude oil refining because of the foremost role of metals. The term of corrosion is usually explained as the formation of the metal oxide, sulfide, hydroxide or any compound related with a carboxylic group on the surface due to the impact of the surrounded conditions. The content of sulfur, acidity, mercaptans and salt content are the well ascertained corrosive properties of crude oils also the chemical composition of metals acquit oneself the fundamental preface for the corrosion. The surveillance of the comparison of corrosive capabilities of Murban and Das Blend crude oils in front of seven different types of ferrous metals that used in Sri Lankan refining industry was the dominant intention of this research. The chemical composition of used metals were determined by XRF detector while testing the contents of sulfur, acidities, contents of mercaptans and salt contents with respect to both crude oils simultaneously. Batches of well cleaned and equal sized metal pieces were immersed in both crude oils separately and the corrosion rate of each type of metal piece was determined after 15, 30 and 45 days from the immersion by weight loss method under the observations through the optical microscope. The ferrous and copper concentrations of each crude oil sample were tested while testing the variation of the hardness of each metal as a confirmation stage. According to results there were found relatively higher amount of sulfur, mercaptans, acids and lower amount of salt in Das Blend than Murban although relatively higher corrosion rates of most of metals in the Murban than the Das Blend. The slight reduction of hardness in each metal piece was found while observing significant concentration of ferrous and copper from some of crude oil samples.
Role of the_morphology_and_surface_planes_on_the_catalytic_activity_of_spinel...

Role of the_morphology_and_surface_planes_on_the_catalytic_activity_of_spinel_li_mn1.5ni0.5o4_for_oxygen_evolution_reaction
Role of the morphology and surface planes on the catalytic activity of spinel...

The electrocatalytic activity of the spinel oxide LiMn1.5Ni0.5O4 with
different morphologies (cubic, spherical, octahedral, and truncated octahedral) has been
investigated for the oxygen evolution reaction (OER) in alkaline solutions that is of
interest for metal−air batteries. The OER activity increases in the order truncated
octahedral < cubic < spherical < octahedral, despite a larger surface area (2.9 m2 g−1) for
the spherical sample compared to nearly similar surface areas (0.3−0.7 m2 g−1) for the
other three samples. The high activity of the octahedral sample is attributed to the
regular octahedral shape with low-energy {111} surface planes, whereas the lowest
activity of the truncated octahedral sample is attributed to the high-energy {001} surface
planes. The octahedral sample also exhibits the lowest Tafel slope of 70 mV dec−1 with
the highest durability whereas the truncated octahedral sample exhibits the highest Tafel
slope of 120 mV dec−1 with durability similar to the cubic and spherical samples. The
study demonstrates that the catalytic activities of oxide catalysts could be tuned and
optimized by controlling the surface morphologies/planes via novel synthesis approaches.
Effects of Precipitation pH Values on the Electrochemical Properties of β-Nic...

IOSR Journal of Applied Chemistry (IOSR-JAC) is a double blind peer reviewed International Journal that provides rapid publication (within a month) of articles in all areas of applied chemistry and its applications. The journal welcomes publications of high quality papers on theoretical developments and practical applications in Chemical Science. Original research papers, state-of-the-art reviews, and high quality technical notes are invited for publications.
Comparative study on structure, corrosion and hardness of Zn-Ni alloy

Comparative study on structure, corrosion and hardness of Zn-Ni alloyUniversidade Federal de Campina Grande
caracterização da liga Zn-NiSimilar to Question Analysis Of Paper 3 Trial Chemistry from Different State (20)
Madkour-1985-Journal_of_Chemical_Technology_and_Biotechnology._Chemical_Techn...

Madkour-1985-Journal_of_Chemical_Technology_and_Biotechnology._Chemical_Techn...
Madkour 1985-journal of-chemical_technology_and_biotechnology._chemical_techn...

Madkour 1985-journal of-chemical_technology_and_biotechnology._chemical_techn...
Synthesis characterisation_and_evaluation_of_ir_o2_based_binary_metal_oxide_...

Synthesis characterisation_and_evaluation_of_ir_o2_based_binary_metal_oxide_...
Oxide film growth on copper in neutral aqueous solutions

Oxide film growth on copper in neutral aqueous solutions
Synthesis, characterisation and evaluation of ir o2 based binary metal oxide ...

Synthesis, characterisation and evaluation of ir o2 based binary metal oxide ...
Synthesis, characterisation and evaluation of ir o2 based binary metal oxide ...

Synthesis, characterisation and evaluation of ir o2 based binary metal oxide ...
Synthesis, characterisation and evaluation of ir o2 based binary metal oxide ...

Synthesis, characterisation and evaluation of ir o2 based binary metal oxide ...
Role of the_morphology_and_surface_planes_on_the_catalytic_activity_of_spinel...

Role of the_morphology_and_surface_planes_on_the_catalytic_activity_of_spinel...
Role of the morphology and surface planes on the catalytic activity of spinel...

Role of the morphology and surface planes on the catalytic activity of spinel...
Effects of Precipitation pH Values on the Electrochemical Properties of β-Nic...

Effects of Precipitation pH Values on the Electrochemical Properties of β-Nic...
A new chelating sorbent for metal ion extraction under high

A new chelating sorbent for metal ion extraction under high
Comparative study on structure, corrosion and hardness of Zn-Ni alloy

Comparative study on structure, corrosion and hardness of Zn-Ni alloy
More from sweemoi khor
Chemistry SPM 2012 Paper 1 Question Analysis

Question Analysis for Paper 1 SPM 2012 according to topics
Operational Definition of Reactivity of Alkali Metals with oxygen

How to define operationally for the reactivity of alkali metals towards oxygen
Heat of combustion of Various Alcohols

Definition of heat of combustion, method to determine heat of combustion and compare heat of combustion between various alcohols
Interpreting energy level diagram

interpret energy level diagram for exothermic & endothermic reaction
More from sweemoi khor (20)
Operational Definition of Reactivity of Alkali Metals with oxygen

Operational Definition of Reactivity of Alkali Metals with oxygen
Predict the Product of Electrolysis Of Aqueous Solution Flowchart

Predict the Product of Electrolysis Of Aqueous Solution Flowchart
Recently uploaded
678020731-Sumas-y-Restas-Para-Colorear.pdf

KKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKKK
Polish students' mobility in the Czech Republic

Polish students mobility to the Czech Republic within eTwinning project "Medieval adventures with Marco Polo"
Chapter 3 - Islamic Banking Products and Services.pptx

Chapter 3 - Islamic Banking Products and Services.pptxMohd Adib Abd Muin, Senior Lecturer at Universiti Utara Malaysia
This slide is prepared for master's students (MIFB & MIBS) UUM. May it be useful to all.Digital Tools and AI for Teaching Learning and Research

This Presentation in details discusses on Digital Tools and AI for Teaching Learning and Research
special B.ed 2nd year old paper_20240531.pdf

Instagram:-
https://instagram.com/special_education_needs_01?igshid=YmMyMTA2M2Y=
WhatsApp:-
https://chat.whatsapp.com/JVakNIYlSV94x7bwunO3Dc
YouTube:-
https://youtube.com/@special_education_needs
Teligram :- https://t.me/special_education_needs
Slide Shere :-
https://www.slideshare.net/shabnambano20?utm_campaign=profiletracking&utm_medium=sssite&utm_source=ssslideview
The Roman Empire A Historical Colossus.pdf

The Roman Empire, a vast and enduring power, stands as one of history's most remarkable civilizations, leaving an indelible imprint on the world. It emerged from the Roman Republic, transitioning into an imperial powerhouse under the leadership of Augustus Caesar in 27 BCE. This transformation marked the beginning of an era defined by unprecedented territorial expansion, architectural marvels, and profound cultural influence.
The empire's roots lie in the city of Rome, founded, according to legend, by Romulus in 753 BCE. Over centuries, Rome evolved from a small settlement to a formidable republic, characterized by a complex political system with elected officials and checks on power. However, internal strife, class conflicts, and military ambitions paved the way for the end of the Republic. Julius Caesar’s dictatorship and subsequent assassination in 44 BCE created a power vacuum, leading to a civil war. Octavian, later Augustus, emerged victorious, heralding the Roman Empire’s birth.
Under Augustus, the empire experienced the Pax Romana, a 200-year period of relative peace and stability. Augustus reformed the military, established efficient administrative systems, and initiated grand construction projects. The empire's borders expanded, encompassing territories from Britain to Egypt and from Spain to the Euphrates. Roman legions, renowned for their discipline and engineering prowess, secured and maintained these vast territories, building roads, fortifications, and cities that facilitated control and integration.
The Roman Empire’s society was hierarchical, with a rigid class system. At the top were the patricians, wealthy elites who held significant political power. Below them were the plebeians, free citizens with limited political influence, and the vast numbers of slaves who formed the backbone of the economy. The family unit was central, governed by the paterfamilias, the male head who held absolute authority.
Culturally, the Romans were eclectic, absorbing and adapting elements from the civilizations they encountered, particularly the Greeks. Roman art, literature, and philosophy reflected this synthesis, creating a rich cultural tapestry. Latin, the Roman language, became the lingua franca of the Western world, influencing numerous modern languages.
Roman architecture and engineering achievements were monumental. They perfected the arch, vault, and dome, constructing enduring structures like the Colosseum, Pantheon, and aqueducts. These engineering marvels not only showcased Roman ingenuity but also served practical purposes, from public entertainment to water supply.
Welcome to TechSoup New Member Orientation and Q&A (May 2024).pdf

In this webinar you will learn how your organization can access TechSoup's wide variety of product discount and donation programs. From hardware to software, we'll give you a tour of the tools available to help your nonprofit with productivity, collaboration, financial management, donor tracking, security, and more.
Biological Screening of Herbal Drugs in detailed.

Biological screening of herbal drugs: Introduction and Need for
Phyto-Pharmacological Screening, New Strategies for evaluating
Natural Products, In vitro evaluation techniques for Antioxidants, Antimicrobial and Anticancer drugs. In vivo evaluation techniques
for Anti-inflammatory, Antiulcer, Anticancer, Wound healing, Antidiabetic, Hepatoprotective, Cardio protective, Diuretics and
Antifertility, Toxicity studies as per OECD guidelines
Honest Reviews of Tim Han LMA Course Program.pptx

Personal development courses are widely available today, with each one promising life-changing outcomes. Tim Han’s Life Mastery Achievers (LMA) Course has drawn a lot of interest. In addition to offering my frank assessment of Success Insider’s LMA Course, this piece examines the course’s effects via a variety of Tim Han LMA course reviews and Success Insider comments.
TESDA TM1 REVIEWER FOR NATIONAL ASSESSMENT WRITTEN AND ORAL QUESTIONS WITH A...

TESDA TM1 REVIEWER FOR NATIONAL ASSESSMENT WRITTEN AND ORAL QUESTIONS WITH ANSWERS.
Home assignment II on Spectroscopy 2024 Answers.pdf

Answers to Home assignment on UV-Visible spectroscopy: Calculation of wavelength of UV-Visible absorption
Model Attribute Check Company Auto Property

In Odoo, the multi-company feature allows you to manage multiple companies within a single Odoo database instance. Each company can have its own configurations while still sharing common resources such as products, customers, and suppliers.
The Challenger.pdf DNHS Official Publication

Read| The latest issue of The Challenger is here! We are thrilled to announce that our school paper has qualified for the NATIONAL SCHOOLS PRESS CONFERENCE (NSPC) 2024. Thank you for your unwavering support and trust. Dive into the stories that made us stand out!
BÀI TẬP BỔ TRỢ TIẾNG ANH GLOBAL SUCCESS LỚP 3 - CẢ NĂM (CÓ FILE NGHE VÀ ĐÁP Á...

BÀI TẬP BỔ TRỢ TIẾNG ANH GLOBAL SUCCESS LỚP 3 - CẢ NĂM (CÓ FILE NGHE VÀ ĐÁP Á...Nguyen Thanh Tu Collection
https://app.box.com/s/hqnndn05v4q5a4k4jd597rkdbda0fniiThe approach at University of Liverpool.pptx

How libraries can support authors with open access requirements for UKRI funded books
Wednesday 22 May 2024, 14:00-15:00.
Recently uploaded (20)
Chapter 3 - Islamic Banking Products and Services.pptx

Chapter 3 - Islamic Banking Products and Services.pptx
Digital Tools and AI for Teaching Learning and Research

Digital Tools and AI for Teaching Learning and Research
Welcome to TechSoup New Member Orientation and Q&A (May 2024).pdf

Welcome to TechSoup New Member Orientation and Q&A (May 2024).pdf
TESDA TM1 REVIEWER FOR NATIONAL ASSESSMENT WRITTEN AND ORAL QUESTIONS WITH A...

TESDA TM1 REVIEWER FOR NATIONAL ASSESSMENT WRITTEN AND ORAL QUESTIONS WITH A...
Home assignment II on Spectroscopy 2024 Answers.pdf

Home assignment II on Spectroscopy 2024 Answers.pdf
BÀI TẬP BỔ TRỢ TIẾNG ANH GLOBAL SUCCESS LỚP 3 - CẢ NĂM (CÓ FILE NGHE VÀ ĐÁP Á...

BÀI TẬP BỔ TRỢ TIẾNG ANH GLOBAL SUCCESS LỚP 3 - CẢ NĂM (CÓ FILE NGHE VÀ ĐÁP Á...
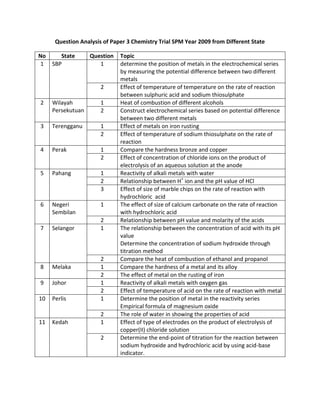
Question Analysis Of Paper 3 Trial Chemistry from Different State
- 1. Question Analysis of Paper 3 Chemistry Trial SPM Year 2009 from Different State NoStateQuestionTopic1SBP1determine the position of metals in the electrochemical series by measuring the potential difference between two different metals2Effect of temperature of temperature on the rate of reaction between sulphuric acid and sodium thiosulphate2Wilayah Persekutuan1Heat of combustion of different alcohols2Construct electrochemical series based on potential difference between two different metals3Terengganu1Effect of metals on iron rusting2Effect of temperature of sodium thiosulphate on the rate of reaction4Perak1Compare the hardness bronze and copper 2Effect of concentration of chloride ions on the product of electrolysis of an aqueous solution at the anode5Pahang1Reactivity of alkali metals with water2Relationship between H+ ion and the pH value of HCl3Effect of size of marble chips on the rate of reaction with hydrochloric acid6Negeri Sembilan1The effect of size of calcium carbonate on the rate of reaction with hydrochloric acid2Relationship between pH value and molarity of the acids7Selangor1The relationship between the concentration of acid with its pH valueDetermine the concentration of sodium hydroxide through titration method2Compare the heat of combustion of ethanol and propanol8Melaka1Compare the hardness of a metal and its alloy2The effect of metal on the rusting of iron9Johor1Reactivity of alkali metals with oxygen gas2Effect of temperature of acid on the rate of reaction with metal10Perlis1Determine the position of metal in the reactivity seriesEmpirical formula of magnesium oxide2The role of water in showing the properties of acid11Kedah1Effect of type of electrodes on the product of electrolysis of copper(II) chloride solution2Determine the end-point of titration for the reaction between sodium hydroxide and hydrochloric acid by using acid-base indicator. 12Kelantan1Reactivity of alkali metals with oxygenpH value of different alkalis2Effect of different metal on the rusting of iron13Sabah1To determine the position of metal in the electrochemical series based on the potential difference between two different metals2Compare the ability of chlorine, bromine and iodine as oxidizing agent in displacement of halogen reaction.14Penang15Sarawak chem2u.blogspot.com