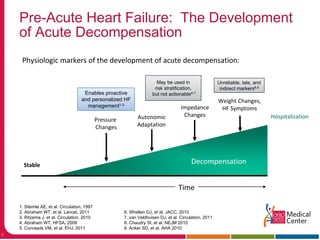
This document discusses management of acute heart failure, including pre-discharge and post-discharge care. It describes four clinical states of heart failure and emphasizes opportunities to prevent hospitalization during the pre-acute and post-acute phases. The CHAMPION trial found that managing heart failure guided by pulmonary artery pressure monitoring reduced heart failure hospitalizations compared to standard care. Post-discharge management strategies like nurse follow-up calls and physician visits in the first month were discussed to help prevent readmission.












![Ejection Fraction
Randomization
Group
Number of
Heart Failure
Hospitalizations
Annualized Rate of
Hospitalization for
Heart Failure
Hazard Ratio
(95% CI)
[p-value]
≥40%
Treatment Group
(n=62)
29 0.43 0.50
(0.35-0.70)
[p<0.0001]
Control Group
(n=57)
59 0.86
≥50%
Treatment Group
(n=35)
13 0.41 0.30
(0.18-0.48)
[p<0.0001]
Control Group
(n=31)
31 1.39
<40%
Treatment Group
(n=208)
153 0.67 0.74
(0.63-0.89)
[p=0.0010]
Control Group
(n=222)
220 0.90](https://image.slidesharecdn.com/pre-andpost-dischargemanagement-160202062621/85/Pre-and-post-discharge-management-14-320.jpg)

![Impact of PA Pressure Guided HF Management
on HF Hospitalizations in GDMT Population
0.39
0.69
HFrEF Patients on ACE/ARB and Beta Blocker Prior to
Implant
HF hospitalization rates
0
0.1
0.2
0.3
0.4
0.5
0.6
0.7
0.8
AnnualizedHFHospitalizationRate
PA Pressure Guided HF Management (Treatment Group)
Guideline Directed Standard of Care (Control Group)
43% Relative Risk Reduction
[HR 0.57, 95% CI 0.45-0.74, p<0.0001]
Number needed to treat: 3
Abraham et al. ACC 2015](https://image.slidesharecdn.com/pre-andpost-dischargemanagement-160202062621/85/Pre-and-post-discharge-management-16-320.jpg)
![Impact of PA Pressure Guided HF Management on
All-Cause Mortality in GDMT Population
57% Relative Risk Reduction
[HR 0.43, 95% CI 0.24-0.76, p=0.0026]
Number needed to treat: 7
Abraham et al. ACC 2015](https://image.slidesharecdn.com/pre-andpost-dischargemanagement-160202062621/85/Pre-and-post-discharge-management-17-320.jpg)





