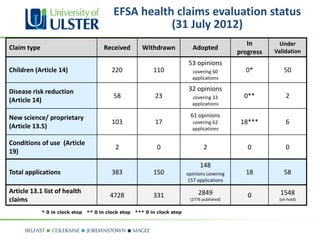
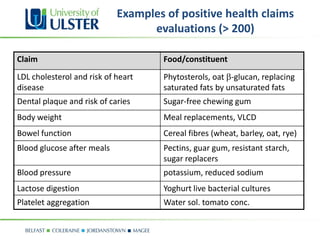
The document provides updates on nutrition labeling and claims regulation in Europe, focusing on the role of the European Food Safety Authority (EFSA) in assessing health claims based on scientific evidence. It outlines conditions under which nutrition and health claims may be authorized and emphasizes the importance of human dietary studies and causality in substantiation. Examples of health claims evaluations highlight key relationships between food constituents and health outcomes.