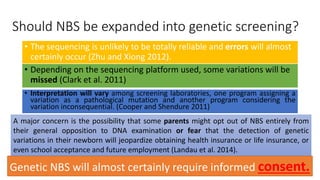
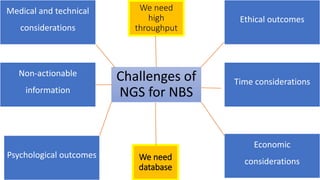
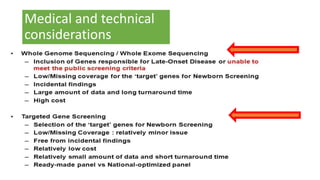
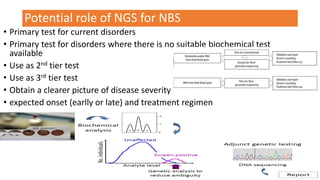
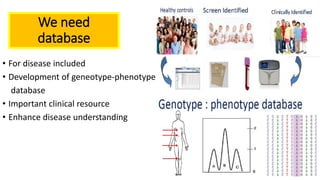
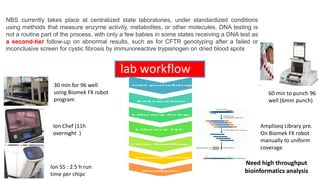
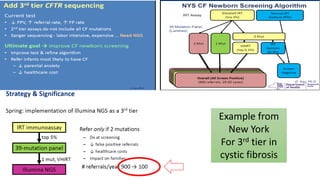
The document discusses the challenges of implementing next-generation sequencing (NGS) in newborn screening (NBS), emphasizing the ethical, psychological, and economic implications. Although NGS holds promise for expanding genetic screening, concerns regarding reliability, informed consent, and the potential for non-actionable information raise significant issues. Key challenges include high costs, the need for extensive bioinformatics support, and the ethical responsibility to ensure that screening results positively impact newborns without causing harm.