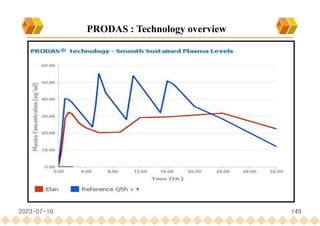
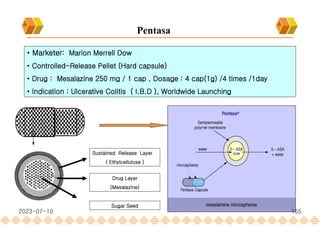
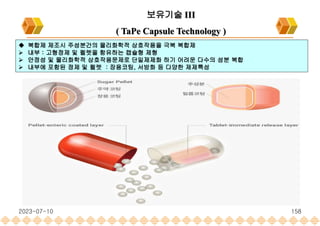
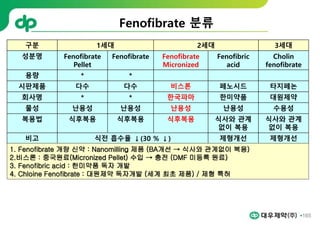
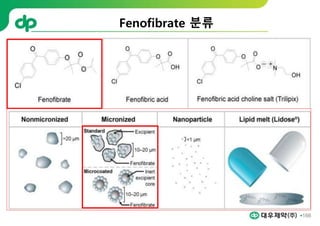
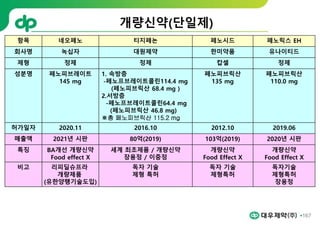
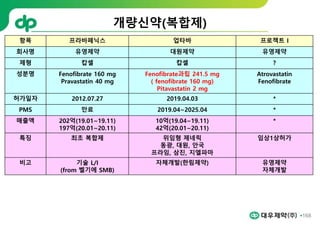
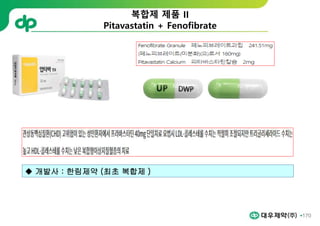
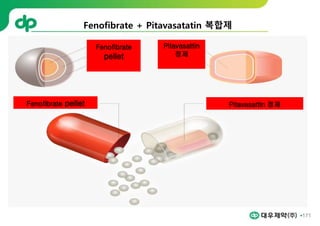
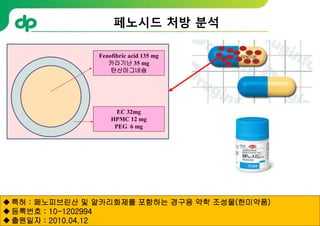
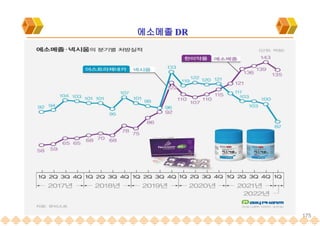
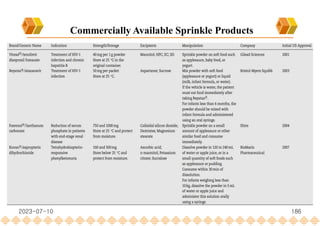
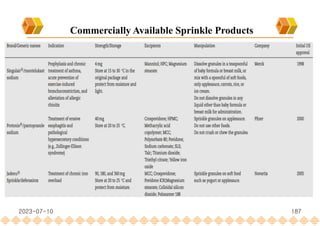
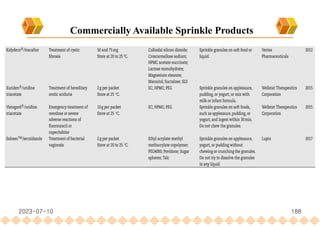
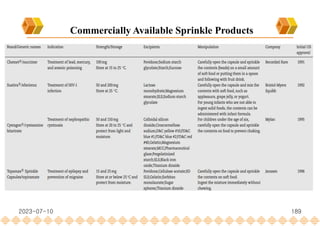
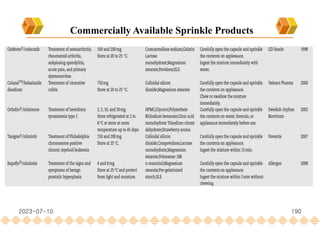
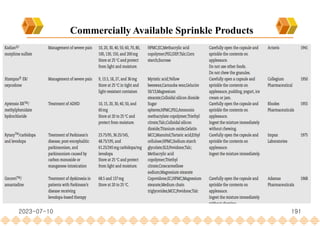
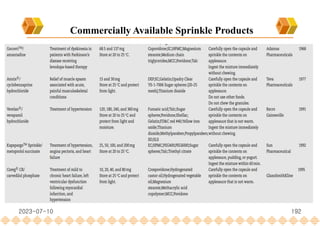
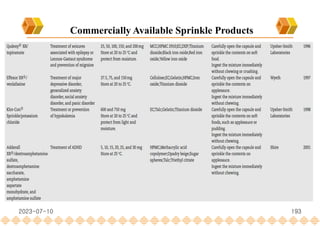
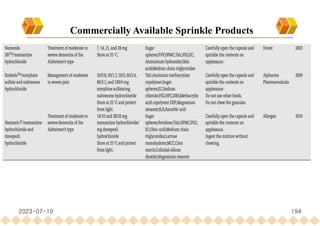
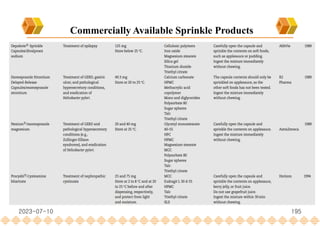
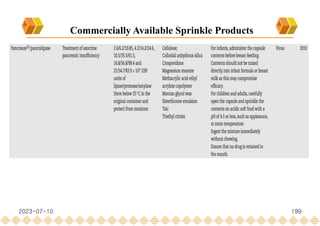
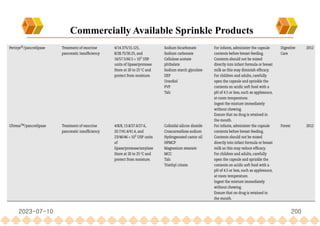
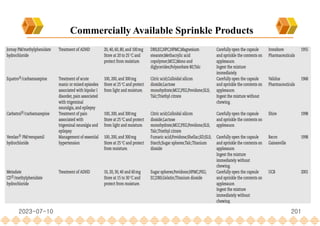
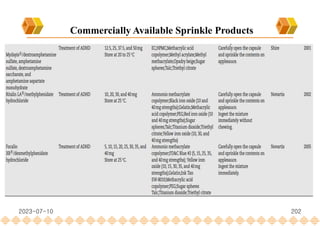
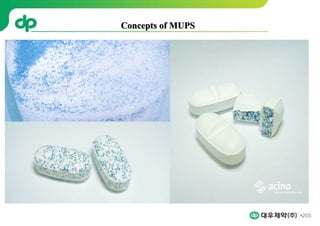
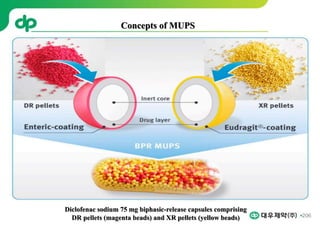
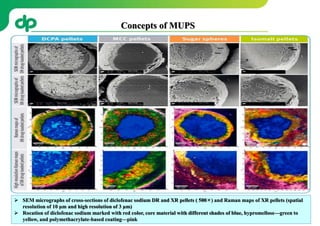
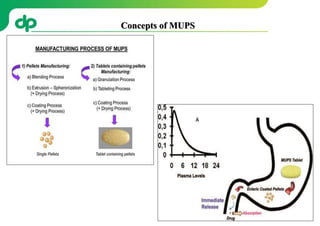
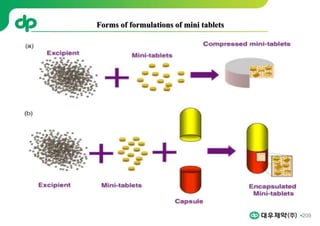
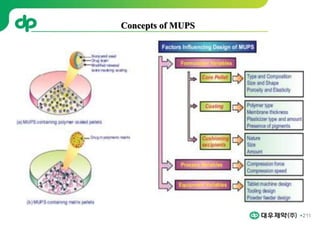
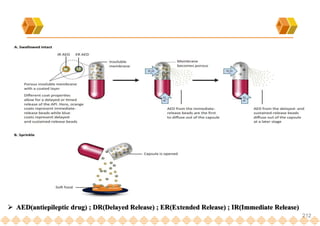
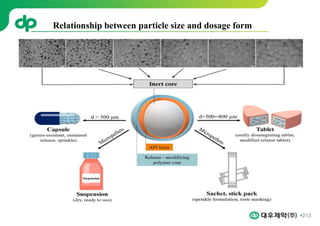
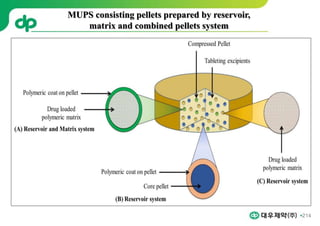
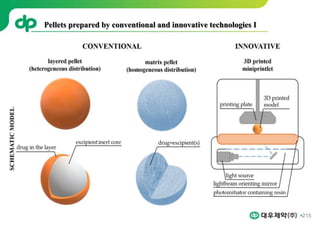
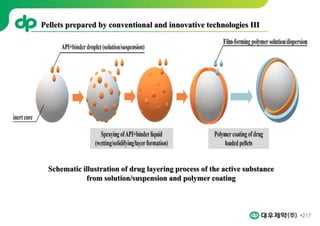
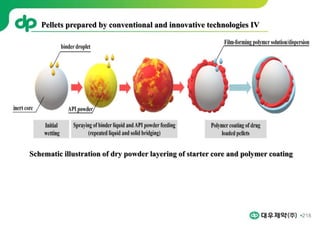
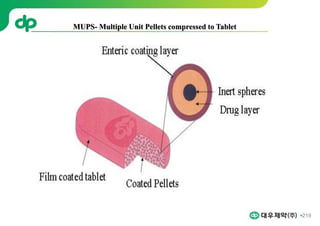
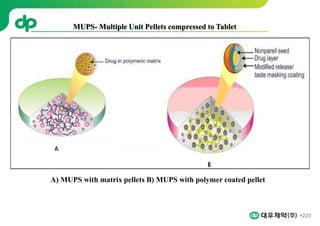
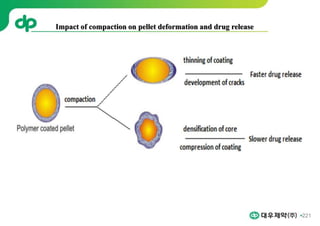
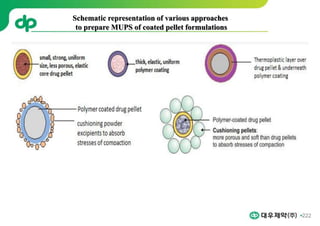
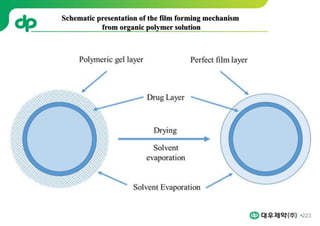
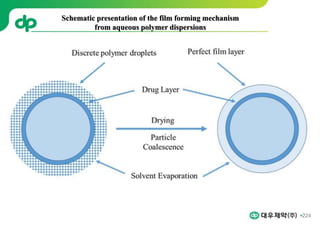
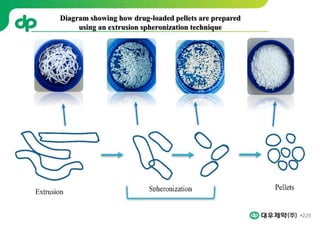
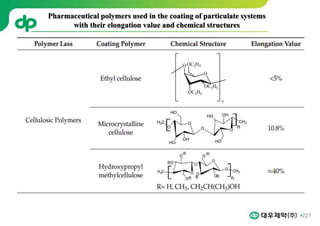
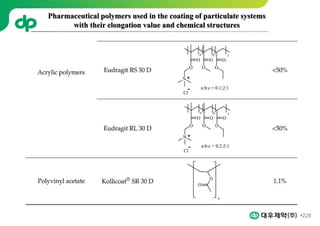
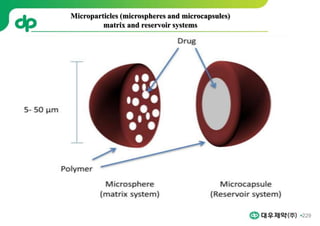
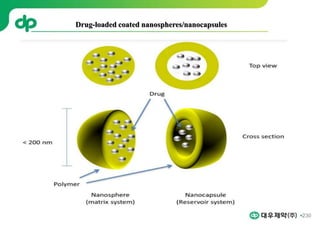
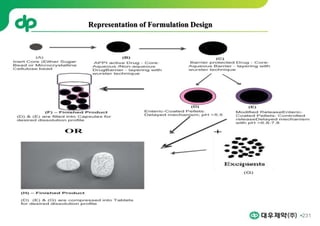
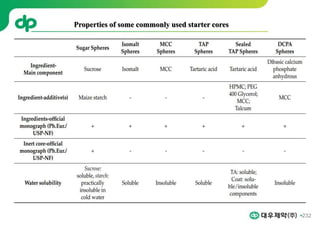
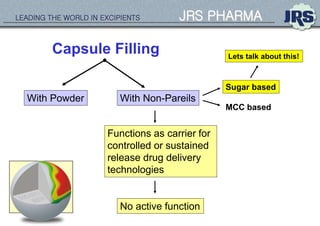
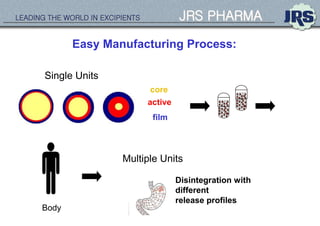
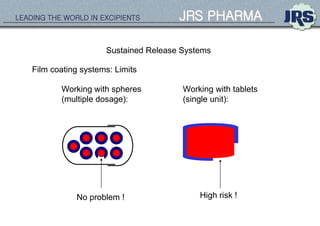
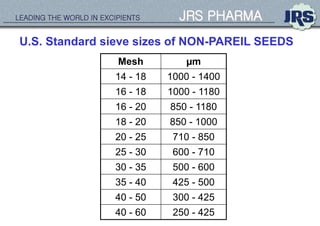
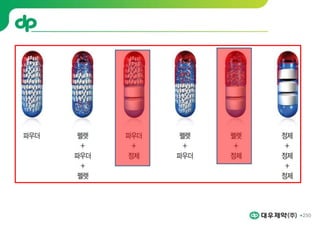
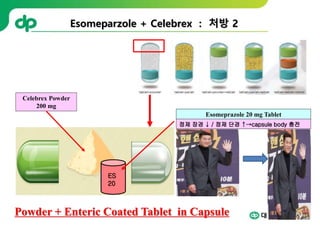
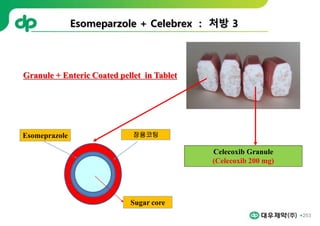
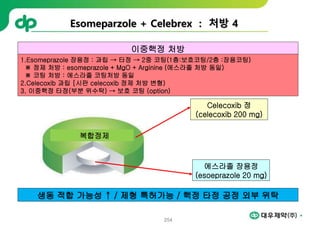
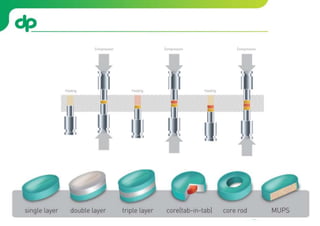
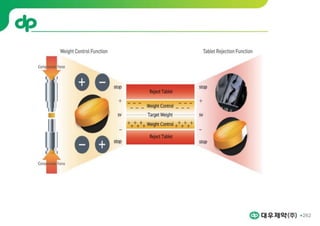
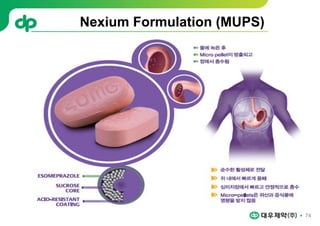
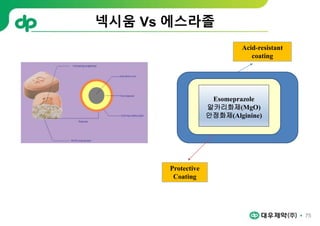
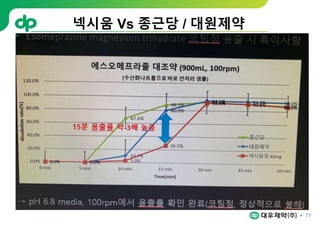
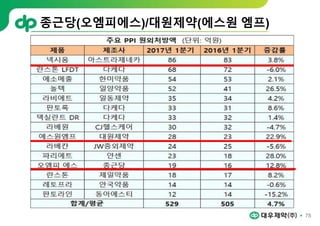
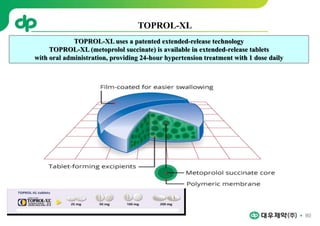
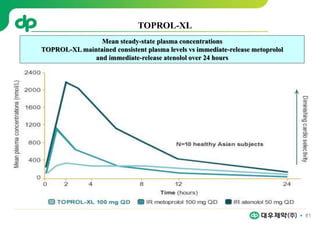
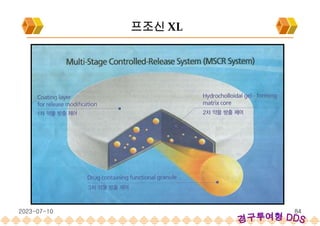
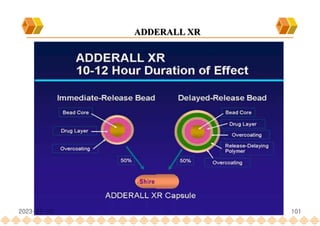
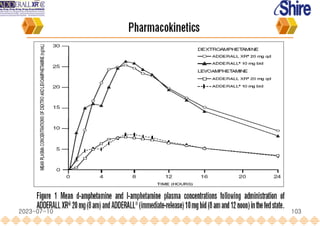
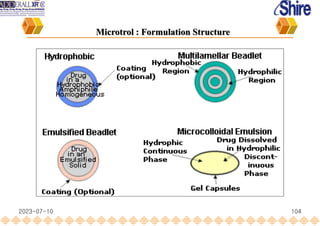
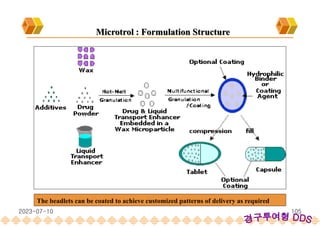
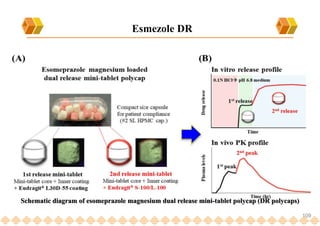
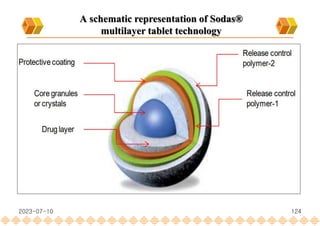
This document provides an overview of oral modified release multiple-unit particulate systems (MUPS). It discusses the introduction and advantages of MUPS, challenges in formulating MUPS, and how those challenges can be overcome. Some key challenges include maintaining the drug release profile after compaction into tablets and preventing damage to pellet coatings during compression. Factors that influence the challenges such as pellet properties, polymer coatings, extragranular materials, and processing are described. Extrusion and spheronization are introduced as methods for producing pellets or granules for use in MUPS.























































































![ SODAS continues to be an accepted and approved system by regulatory authorities with the
most recent regulatory approvals for a SODAS based system occurring since US in 2002 with
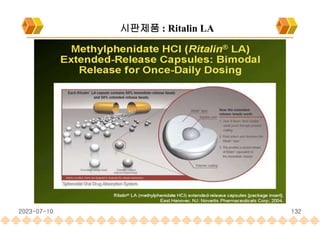
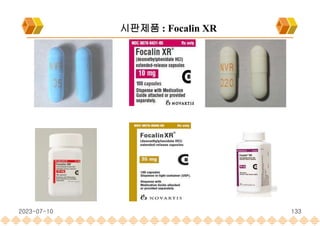
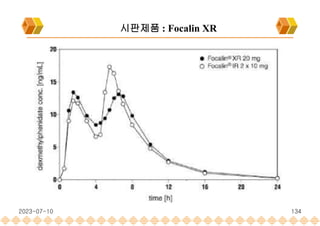
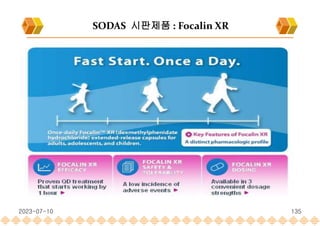
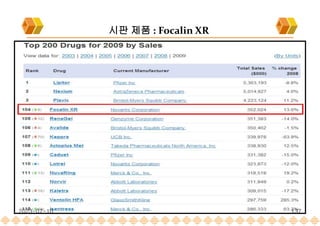
the launch of once daily oral dosage forms of Avinza®, Ritalin® LA and Focalin® XR
A number of other compounds are in late stage development utilizing Elan’s SODAS technology
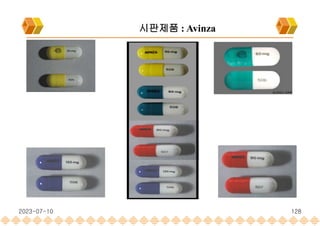
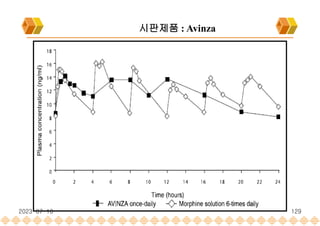
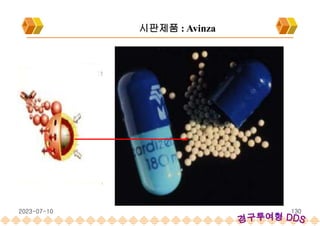
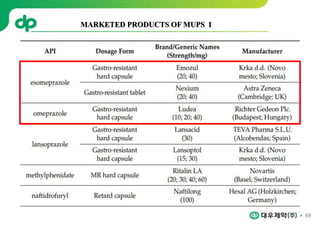
Avinza : Morphine Sulfate Extended release capsule[Norvatis]
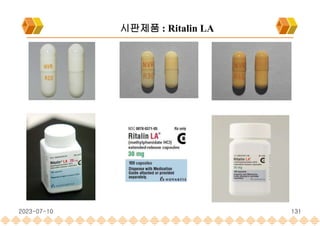
Ritalin LA : Methylphenidate HCl , Extended release capsule[Ligand]
Focalin XR : Dexmethylphenidate (Norvatis)
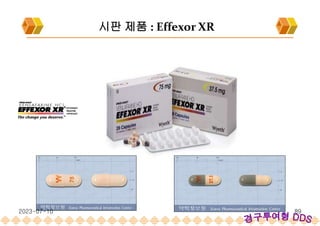
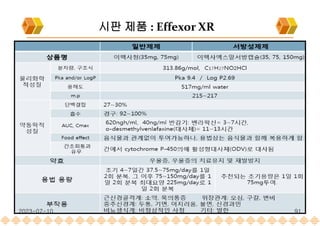
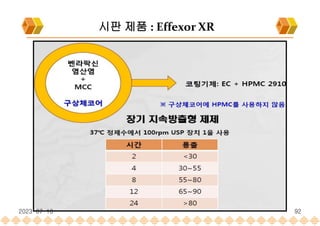
SODAS : 시판제품
2023-07-10 127](https://image.slidesharecdn.com/mupsver11-230710044036-6a68c3bc/85/mups-Ver-11-0-pptx-127-320.jpg)