Mix2Vial
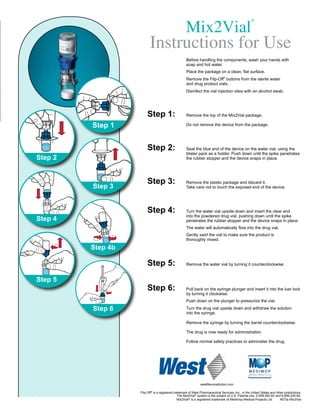
- 1. Mix2Vial ® Instructions for Use Before handling the components, wash your hands with soap and hot water. Place the package on a clean, flat surface. Remove the Flip-Off® buttons from the sterile water and drug product vials. Disinfect the vial injection sites with an alcohol swab. Step 1: Remove the top of the Mix2Vial package. Step 1 Do not remove the device from the package. Step 2: Seat the blue end of the device on the water vial, using the blister pack as a holder. Push down until the spike penetrates Step 2 the rubber stopper and the device snaps in place. Step 3: Remove the plastic package and discard it. Step 3 Take care not to touch the exposed end of the device. Step 4: Turn the water vial upside down and insert the clear end into the powdered drug vial, pushing down until the spike Step 4 penetrates the rubber stopper and the device snaps in place. The water will automatically flow into the drug vial. Gently swirl the vial to make sure the product is thoroughly mixed. Step 4b Step 5: Remove the water vial by turning it counterclockwise. Step 5 Step 6: Pull back on the syringe plunger and insert it into the luer lock by turning it clockwise. Push down on the plunger to pressurize the vial. Step 6 Turn the drug vial upside down and withdraw the solution into the syringe. Remove the syringe by turning the barrel counterclockwise. The drug is now ready for administration. Follow normal safety practices to administer the drug. westReconstitution.com Flip-Off® is a registered trademark of West Pharmaceutical Services, Inc., in the United States and other jurisdictions. The Mix2Vial® system is the subject of U.S. Patents nos. 6,558,365 B2 and 6,699,229 B2. Mix2Vial® is a registered trademark of Medimop Medical Projects Ltd. 4573a Mix2Vial
- 2. Drug Kitting Custom transfer and delivery solutions A drug distributed in kit form can facilitate clinical trials, differentiate the product in the market, create brand loyalty and can simplify and streamline its administration. Through its WestOneSource facility, West provides customers with a single source for kit design and assembly. The kit contains the materials needed to prepare and administer a drug in a single, convenient package, providing a ready-to-use solution for drug administration in clinical and home settings. Kit contents typically include: • The drug vial • Diluent in a vial or a pre-filled syringe • A syringe to administer the dose • A device to simplify reconstitution and transfer of the drug between vials and the syringe Innovations in • Alcohol swabs Safety and • Administration needle with optional safety device Administration Systems The kit can be fabricated using a cardboard or thermoformed plastic tray with com- West’s safety and administration partments to contain each of the components. systems enhance the delivery of drugs West offers kits that include its safety and administration systems: • MixJect® – a system for the safe, rapid preparation of lyophilized drugs through advanced reconstitution, with pre-attached administration needle. mixing and transfer technologies. • Mix2Vial – a needleless system for safe, rapid transfer and ® mixing between two vials. West’s systems are easy to use, help • Vial2Bag™ – a needleless system for the safe reconstitution and promote safety and compliance transfer of a drug between a vial or syringe and an IV bag. and do not require changes to • Vial Adapters – a cost-effective solution for the safe and rapid drug manufacturing processes. The transfer and reconstitution of drugs between vials and syringes. systems work with existing drug West can provide customers kits in quantities raging from samples to full commercial development. vials – there is no need for expensive re-registration or capital investment. To find out how West’s innovative systems can add value to your drug product or application, please contact your local West account representative. For direct access to a business development specialist, call 800-345-9800, option 8, in North America or +45 7561-6000 in Europe. You may also send email inquiries to SafetySystems@westpharma. com and visit our web site at westReconstitution.com. Trademarks and registered trademarks are the property of Medimop Medical Projects Ltd. 4573g Drug Kitting