Mineralization of organic nitrogen
•
1 like•869 views
Mineralization of Organic Nitrogen- 1.Aminization 2.Ammonification 3.Nitrification
Report
Share
Report
Share
Download to read offline
Recommended
Transformation of Nitrogen, Phosphorous, Potassium and Sulphur
Transformation of Nitrogen, Phosphorous, Potassium and Sulphur 

Transformation of Nitrogen, Phosphorous, Potassium and Sulphur INDIRA GANDHI KRISHI VISWAVIDYALAYA, RAIPUR
The portion of a plant left in the field after harvest of the crop that is (straw, stalks, stems, leaves, roots) not used domestically or sold commercially”. The non – economical plant parts that are left in the field after harvest and remains that are generated from packing sheds or that are discarded during crop processing. Organic recycling has to play a key role in achieving sustainability in agricultural production. Multipurpose uses of crop residue include, but are not limited to, animal feeding, soil mulching, bio-manure, thatching of rural homes and fuel for domestic and industrial use. Thus, crop residues are of tremendous value to the farmers. Crop residue benefit the soil physically, chemically as well as biologically.
Crop Residue Management for Soil Health Enhancement

Crop Residue Management for Soil Health EnhancementVasantrao Nail Marathwada Krishi Vidyapeeth, Parbhani
Recommended
Transformation of Nitrogen, Phosphorous, Potassium and Sulphur
Transformation of Nitrogen, Phosphorous, Potassium and Sulphur 

Transformation of Nitrogen, Phosphorous, Potassium and Sulphur INDIRA GANDHI KRISHI VISWAVIDYALAYA, RAIPUR
The portion of a plant left in the field after harvest of the crop that is (straw, stalks, stems, leaves, roots) not used domestically or sold commercially”. The non – economical plant parts that are left in the field after harvest and remains that are generated from packing sheds or that are discarded during crop processing. Organic recycling has to play a key role in achieving sustainability in agricultural production. Multipurpose uses of crop residue include, but are not limited to, animal feeding, soil mulching, bio-manure, thatching of rural homes and fuel for domestic and industrial use. Thus, crop residues are of tremendous value to the farmers. Crop residue benefit the soil physically, chemically as well as biologically.
Crop Residue Management for Soil Health Enhancement

Crop Residue Management for Soil Health EnhancementVasantrao Nail Marathwada Krishi Vidyapeeth, Parbhani
More Related Content
What's hot
What's hot (20)
Potassium- Forms,Equilibrium in soils and its agricultural significance ,mech...

Potassium- Forms,Equilibrium in soils and its agricultural significance ,mech...
Essential nutrients-functions and deficiency symptoms, manures, fertilizers a...

Essential nutrients-functions and deficiency symptoms, manures, fertilizers a...
Survival and dispersal of important plant pathogen

Survival and dispersal of important plant pathogen
Similar to Mineralization of organic nitrogen
Similar to Mineralization of organic nitrogen (20)
Oxidation reduction reactions BY Muhammad Fahad Ansari 12IEEM14

Oxidation reduction reactions BY Muhammad Fahad Ansari 12IEEM14
Oxidation reduction reactions By MUHAMMAD FAHAD ANSARI 12 IEEM 14

Oxidation reduction reactions By MUHAMMAD FAHAD ANSARI 12 IEEM 14
.Nitrogen Cycle.ppt how the nitrogen are fixed in soil

.Nitrogen Cycle.ppt how the nitrogen are fixed in soil
Recently uploaded
Recently uploaded (20)
QUATER-1-PE-HEALTH-LC2- this is just a sample of unpacked lesson

QUATER-1-PE-HEALTH-LC2- this is just a sample of unpacked lesson
Introduction to TechSoup’s Digital Marketing Services and Use Cases

Introduction to TechSoup’s Digital Marketing Services and Use Cases
21st_Century_Skills_Framework_Final_Presentation_2.pptx

21st_Century_Skills_Framework_Final_Presentation_2.pptx
PANDITA RAMABAI- Indian political thought GENDER.pptx

PANDITA RAMABAI- Indian political thought GENDER.pptx
Interdisciplinary_Insights_Data_Collection_Methods.pptx

Interdisciplinary_Insights_Data_Collection_Methods.pptx
Unit 3 Emotional Intelligence and Spiritual Intelligence.pdf

Unit 3 Emotional Intelligence and Spiritual Intelligence.pdf
Beyond_Borders_Understanding_Anime_and_Manga_Fandom_A_Comprehensive_Audience_...

Beyond_Borders_Understanding_Anime_and_Manga_Fandom_A_Comprehensive_Audience_...
Mineralization of organic nitrogen
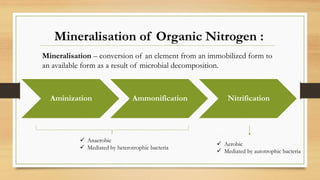
- 1. Aminization Ammonification Nitrification Anaerobic Mediated by heterotrophic bacteria Aerobic Mediated by autotrophic bacteria Mineralisation of Organic Nitrogen : Mineralisation – conversion of an element from an immobilized form to an available form as a result of microbial decomposition.
- 2. 1. Aminization : • Hydrolytic decomposition of protein and release of amines and amino acids by heterotrophs (bacteria like Bacillus, Pseudomonas) in absence of O2 . O.M.---------------> Polypeptides Polypeptides/Proteins --------- > R-NH2 +CO2 + Energy + other compounds. hydrolysis
- 3. 2. Ammonification : • The amines and amino acids so released are further utilised by other hetrotrophs by release of ammonical compounds in absence of O2 . R-NH2 + HOH --------> NH3 + R-OH + Energy 2NH3 + H2CO3 --------> (NH4)2CO3 2NH+ 4 + CO3 2- NH3 so released are subjected to several fates— (a) may be converted to Nitrites and Nitrates by nitrification. (b) may be absorbed directly by higher plants. (c) may be utilized by heterotrophs organisms in the further decom position of O.M.
- 4. (d) may be fixed in a biologically unavailable form (in subsoil about 40-50% and in top soils 6 %) in the lattice of certain expanding type of clay-minerals like momtmorillonite , illite and vermiculite. 3. Nitrification : • By autotrophs in presence of O2 i.e. biological oxidation. According to Winogradsky (1889), NH3 or NH+ 4 salt in the soil is oxidised into NO- 3 in two steps. (i) NH3 / NH4 + ------------------------> NO2 - [Nitrite] Nitrosomonas is Obligate parasite. (ii) NO2 - ---------------------> NO3 - [Nitrate] [O] Nitrosomonas Nitrobacter [O]
- 5. Opt. temperature for Nitrifying bacteria is 30-35° C and Opt. pH 6 .5-7.5. The activity of bacteria ceases below pH 5.0. Nitrobacteria = Nitrosomonas + Nitrobacter both. Reactions: 2 NH 4 + + 3O2 ---------------> 2NO2 -+ 2H2O + 4H+ + Energy . 2 NO 2 -+ O2 —---------> 2 NO3 - + energy. When NH 4 + - Fertilizers are added to soil, three important inferences are drawn by these nitrification equations – (i) Reactions require molecular oxygen means nitrification takes place most readily in well aerated soil. (ii) Reactions release H+ which results in acidification of soil. (iii) Microbial activity hence their rapidity and extent of transformation will be greatly influenced by soil environmental conditions such as moisture supply, temp etc.
- 6. Factors affecting Nitrification : (i) Supply of NH 4 + (ii) Population of nitrifying organisms (iii) Soil reactions (iv) Soil aeration (v) Soil moisture (vi) Soil temp. (30-35° C) (vii) C : N ratio - <20 favours mineralization (release of NH3 & NO3 - ) >30 immobilization 15-30 or 20-30 - both (viii) Fertility.
- 7. Thank You!