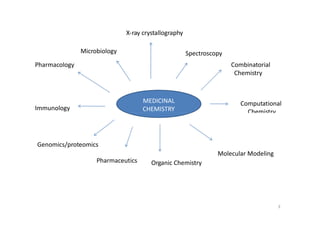
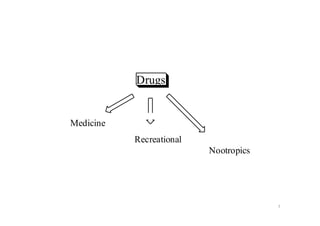
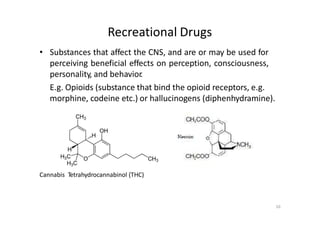
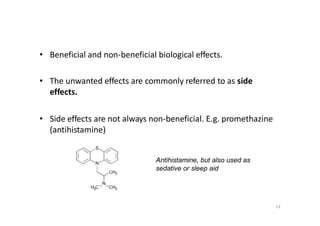
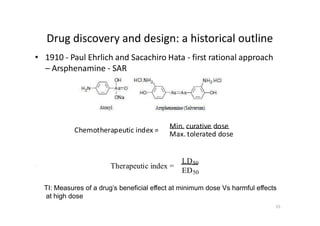
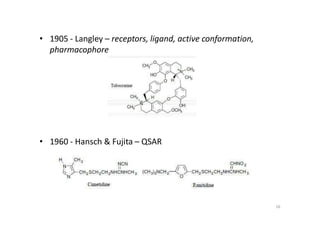
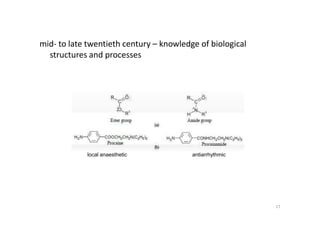
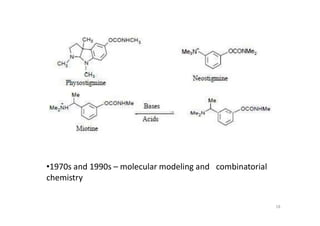
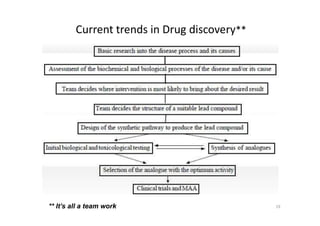
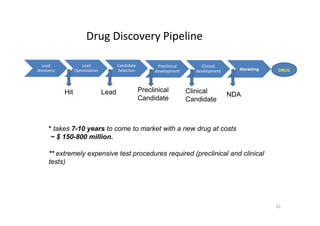
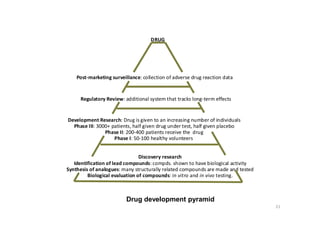
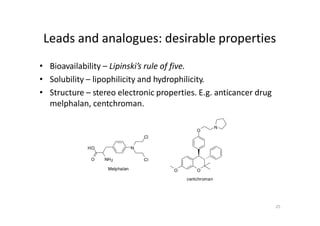
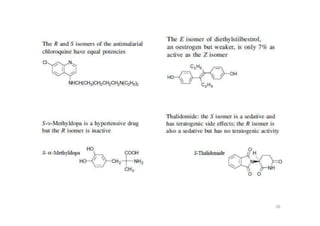
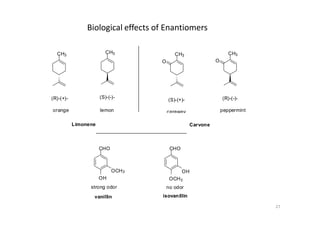
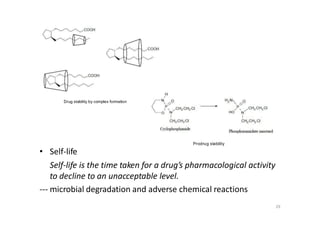
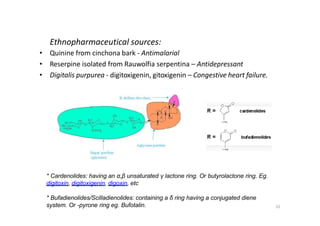
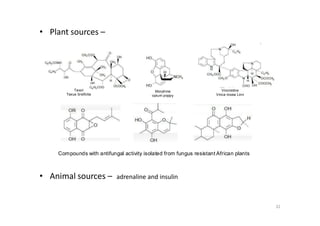
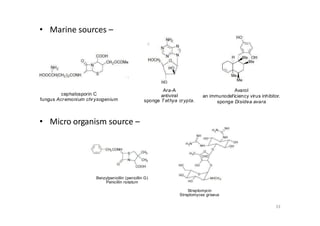
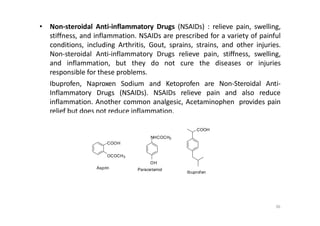
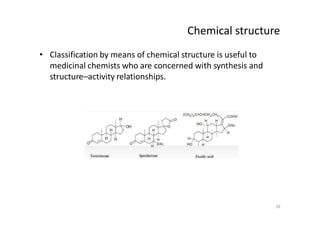
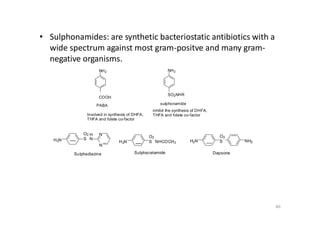
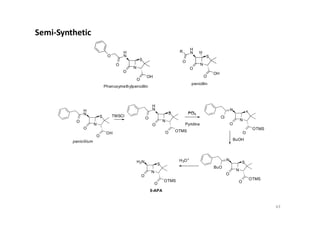
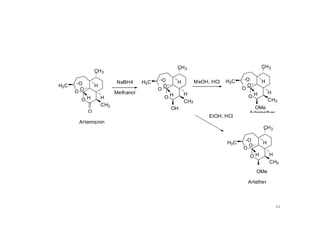
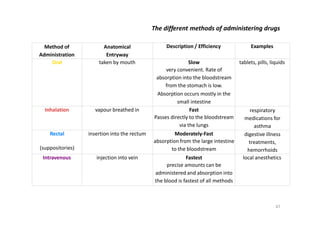
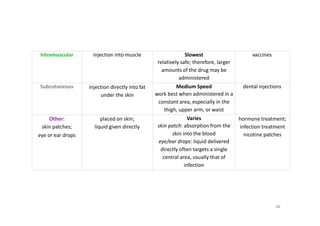
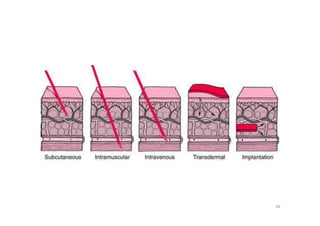
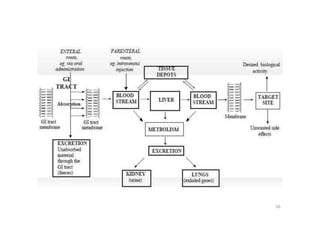
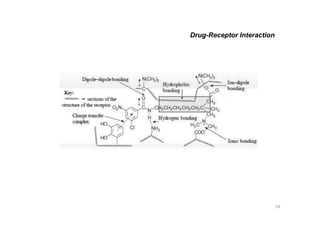
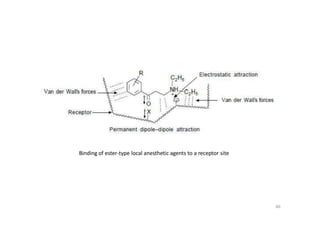
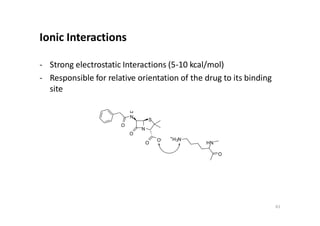
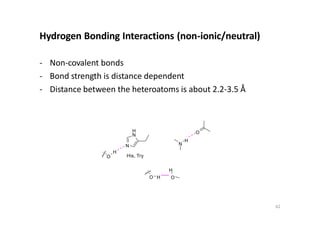
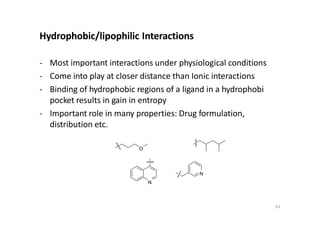
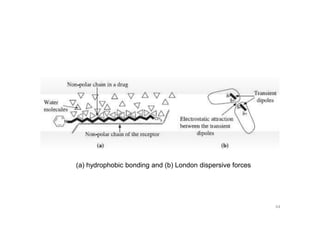
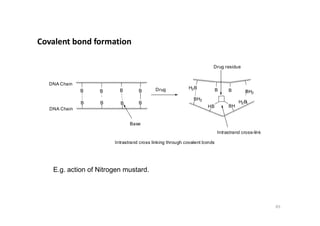
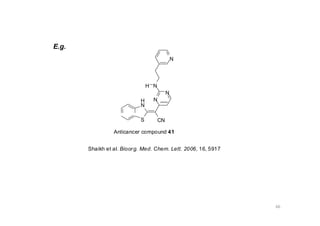
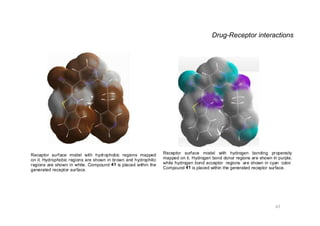
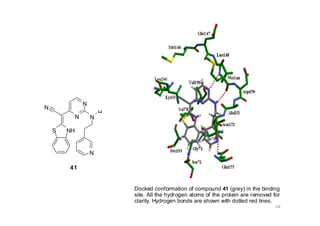
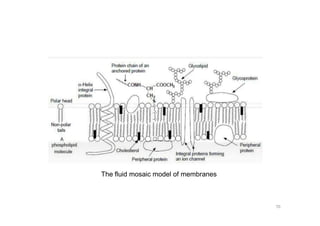
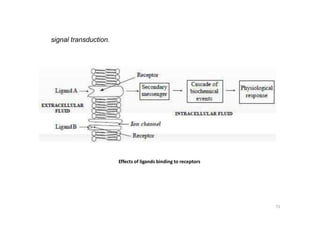
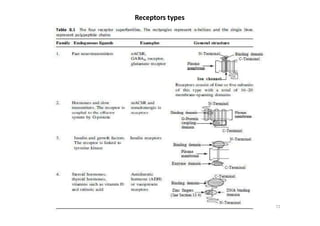
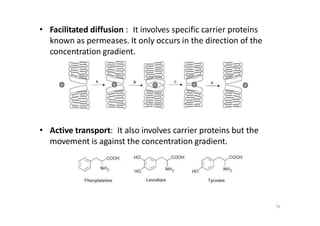
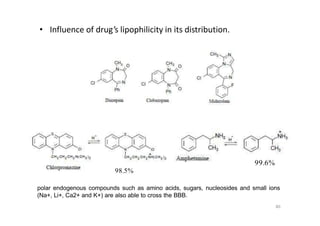
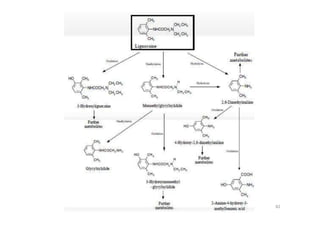
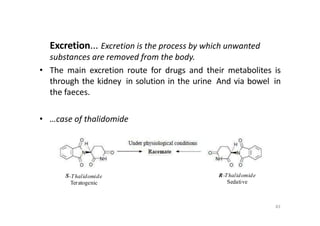
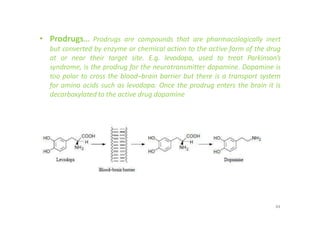
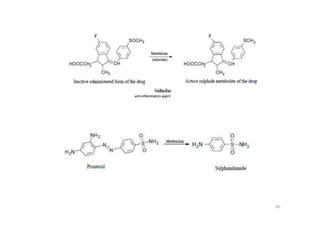
This document provides an overview of the field of medicinal chemistry. It defines medicinal chemistry as an interdisciplinary research area that incorporates different branches of chemistry and biology to research and develop new drugs. The roles of medicinal chemists include synthesizing and characterizing new compounds, determining biological effects, optimizing lead compounds, and studying pharmacokinetic properties. It also discusses the drug development process and clinical trials. Key aspects covered include drug classification, sources of drug leads, and factors that influence drug properties such as stability and bioavailability.