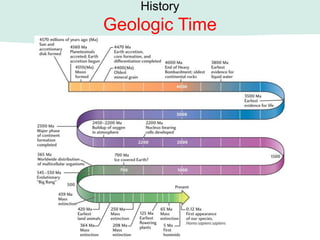
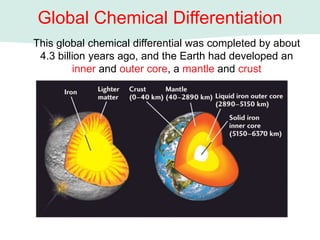
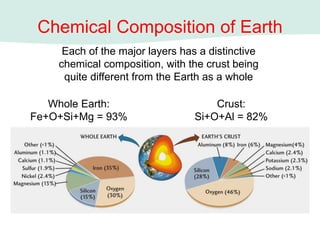
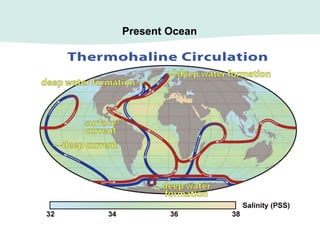
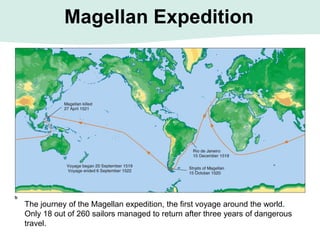
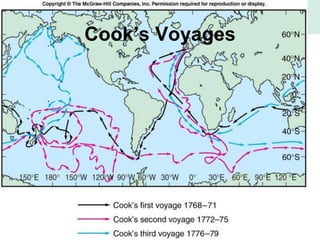
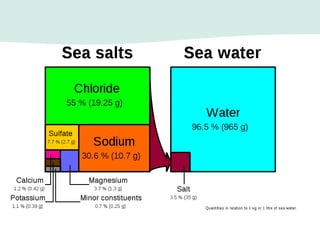
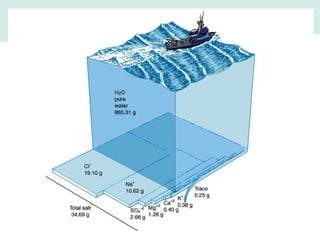
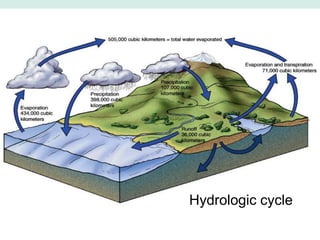
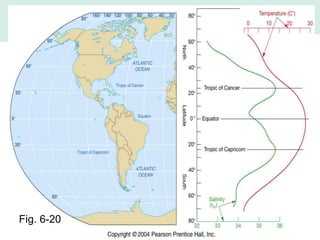
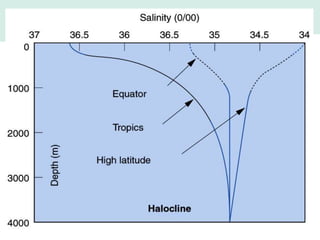
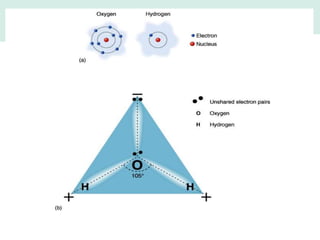
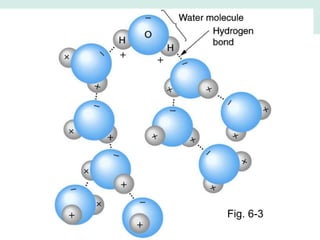
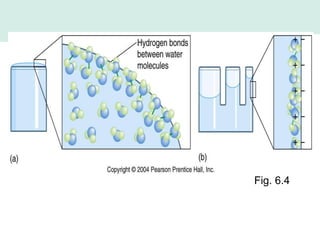
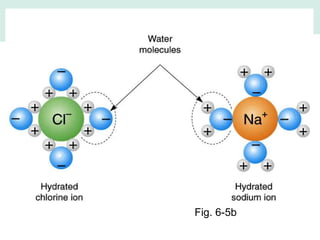
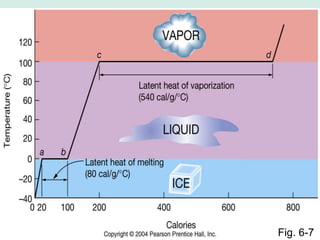
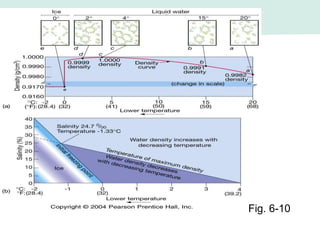
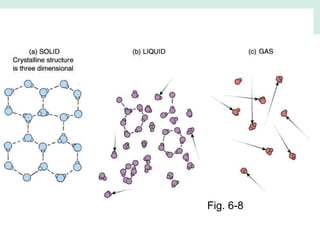
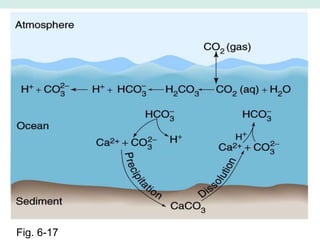
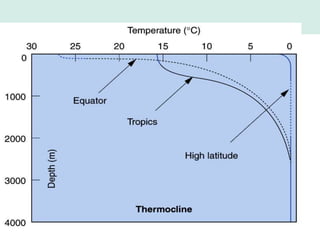
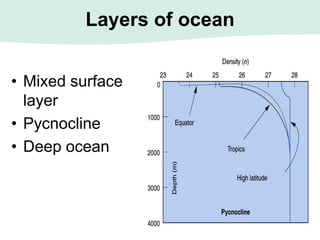
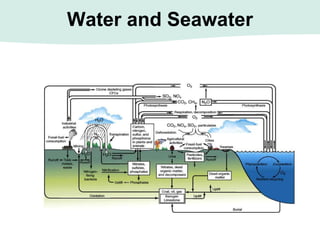
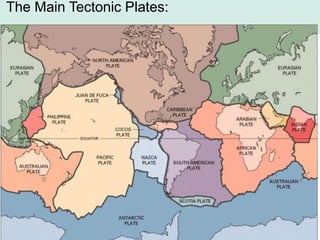
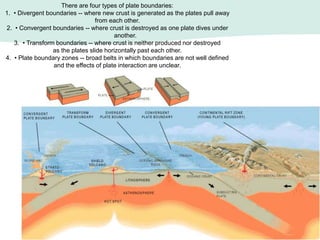
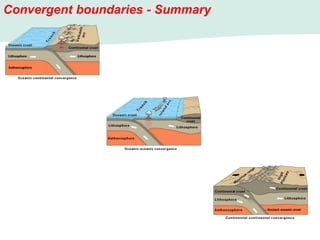
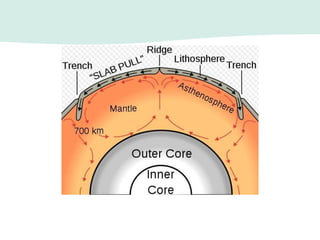
Oceanography is the study of the earth's oceans and their interconnected ecosystems, geological, chemical and physical processes. It includes the study of geological, chemical, physical, meteorological and biological aspects of the oceans. The development of the oceans and atmosphere has evolved over geologic time through processes like volcanic outgassing, asteroid impacts and plate tectonics. Modern oceanography developed through historic voyages of exploration and the rise of dedicated oceanographic research institutions.