Lymphoma diffuse large cells.docx
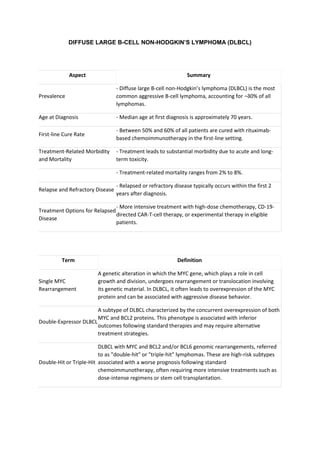
- 1. DIFFUSE LARGE B-CELL NON-HODGKIN’S LYMPHOMA (DLBCL) Aspect Summary Prevalence - Diffuse large B-cell non-Hodgkin’s lymphoma (DLBCL) is the most common aggressive B-cell lymphoma, accounting for ∼ 30% of all lymphomas. Age at Diagnosis - Median age at first diagnosis is approximately 70 years. First-line Cure Rate - Between 50% and 60% of all patients are cured with rituximab- based chemoimmunotherapy in the first-line setting. Treatment-Related Morbidity and Mortality - Treatment leads to substantial morbidity due to acute and long- term toxicity. - Treatment-related mortality ranges from 2% to 8%. Relapse and Refractory Disease - Relapsed or refractory disease typically occurs within the first 2 years after diagnosis. Treatment Options for Relapsed Disease - More intensive treatment with high-dose chemotherapy, CD-19- directed CAR-T-cell therapy, or experimental therapy in eligible patients. Term Definition Single MYC Rearrangement A genetic alteration in which the MYC gene, which plays a role in cell growth and division, undergoes rearrangement or translocation involving its genetic material. In DLBCL, it often leads to overexpression of the MYC protein and can be associated with aggressive disease behavior. Double-Expressor DLBCL A subtype of DLBCL characterized by the concurrent overexpression of both MYC and BCL2 proteins. This phenotype is associated with inferior outcomes following standard therapies and may require alternative treatment strategies. Double-Hit or Triple-Hit DLBCL with MYC and BCL2 and/or BCL6 genomic rearrangements, referred to as "double-hit" or "triple-hit" lymphomas. These are high-risk subtypes associated with a worse prognosis following standard chemoimmunotherapy, often requiring more intensive treatments such as dose-intense regimens or stem cell transplantation.
- 2. Parameter General Index Age Adjusted Index Age > 60 years 1 n.a. PS (Performance Status) 1 1 Stage 3-4 1 1 LDH elevated 1 1 Extra nodal >1 site non considered 5 No. of Risk Factors IPI Group 0 - 1 1 = low 2 2 = low intermediate 3 3 = high intermediate 4 - 5 4 = high Treatment Drugs Included Response Rate Progression- Free Survival Overall Survival First Line R-CHOP Rituximab, Cyclophosphamide, Doxorubicin, Vincristine, Prednisone ~70% ~3 years ~60% at 5 years DA-EPOCH-R Rituximab, Etoposide, Prednisone, Vincristine, Cyclophosphamide, Doxorubicin ~80% ~3 years ~60% at 5 years
- 3. Polatuzumab vedotin + R-CHP Rituximab, Cyclophosphamide, Doxorubicin, Vincristine, Prednisone, Polatuzumab vedotin ~85% Not yet established Not yet established Acalabrutinib + R-CHOP or R- DHAP Rituximab, Cyclophosphamide, Doxorubicin, Vincristine, Prednisone (or High-Dose Cytarabine) + Acalabrutinib Not yet established Not yet established Not yet established Tafasitamab + Lenalidomide + R-CHP Rituximab, Cyclophosphamide, Doxorubicin, Vincristine, Prednisone, Tafasitamab, Lenalidomide Not yet established Not yet established Not yet established Second Line ICE Ifosfamide, Carboplatin, Etoposide ~60% ~2 years ~25-30% at 5 years R-ICE Rituximab, Ifosfamide, Carboplatin, Etoposide ~70% ~2 years ~30-40% at 5 years GEMOX Gemcitabine, Oxaliplatin ~50% ~1.5 years ~35-40% at 5 years Third Line
- 4. DHAP or ESHAP Cytarabine, Cisplatin, Dexamethasone (or Etoposide, Methylprednisolone, Cisplatin) ~50% ~1 year ~25-30% at 5 years R-DHAP or R- ESHAP Rituximab, Cytarabine, Cisplatin, Dexamethasone (or Etoposide, Methylprednisolone, Cisplatin) ~60% ~1.5 years ~30-40% at 5 years GDP Gemcitabine, Dexamethasone, Cisplatin ~50% ~1 year ~25-30% at 5 years Stage of Disease Treatment Limited stage (stage I or II) Combined modality therapy consisting of 3-4 cycles of R- CHOP chemotherapy followed by involved-field radiation therapy (IFRT) Advanced stage (stage III or IV) R-CHOP chemotherapy for 6-8 cycles Advanced stage (stage III or IV) with high tumor burden or poor prognostic factors R-CHOP chemotherapy followed by high-dose chemotherapy and autologous stem cell transplant (ASCT) Relapsed or refractory disease Salvage chemotherapy followed by high-dose chemotherapy and autologous stem cell transplant (ASCT), or CAR T-cell therapy
- 5. Second-line treatment for patients ineligible for ASCT Polatuzumab vedotin in combination with bendamustine and rituximab (BRP) Maintenance therapy Lenalidomide or rituximab for up to 2 years after initial therapy Pharmaceutical Name Commercial Name Manufacturer Dose and Route of Administration Polatuzumab vedotin Polivy Genentech/Roche 1.8 mg/kg intravenous Acalabrutinib Calquence AstraZeneca 100 mg twice daily oral Tafasitamab Monjuvi MorphoSys/Incyte 12 mg/kg intravenous over approximately 30 minutes on days 1, 8, 15, and 22 of each cycle in combination with lenalidomide Lisocabtagene maraleucel Breyanzi Bristol Myers Squibb/Juno Therapeutics A single infusion of 0.6 to 6.0 × 10^8 CAR-positive viable T cells in 68 to 92 mL via intravenous infusion Umbralisib Ukoniq TG Therapeutics 800 mg orally once daily Drugs Indications Year of Approval FDA Approval EMA Approval Clinical Trials Results
- 6. Polatuzumab vedotin In combination with chemotherapy 2019 Yes Yes GO29365 (NCT02257567) Improved median OS vs R-CHOP (12.4 vs 4.7 months) Acalabrutinib In combination with chemotherapy or alone 2020 Yes Yes ACE-LY-308 (NCT02953814) Improved ORR and PFS vs placebo Tafasitamab In combination with lenalidomide 2020 Yes Yes L-MIND (NCT02399085) Improved ORR and CR vs lenalidomide alone Lisocabtagene maraleucel (Liso-cel) Relapsed or refractory DLBCL 2021 Yes Pending TRANSCEND (NCT02631044) ORR of 73% and CR of 53% Umbralisib In combination with ublituximab 2021 Yes Pending UNITY-NHL (NCT02793583) Improved ORR and CR vs ublituximab alone Zanubrutinib In combination with rituximab 2021 Yes Pending SEQUOIA (NCT03332017) Improved PFS and ORR vs R-CHOP Drugs Advantages Disadvantages Adverse Events (percentage) Polatuzumab vedotin Improved progression-free survival and overall survival when added to chemotherapy compared to chemotherapy alone Increased risk of infection, liver damage, and nerve damage; infusion reactions (31% had grade 3 or higher) Infections (48%), neutropenia (46%), anemia (32%), thrombocytopenia (21%), peripheral neuropathy (16%)
- 7. Acalabrutinib Oral administration and favorable safety profile compared to traditional chemotherapy Increased risk of bleeding and atrial fibrillation; possible infections and skin cancers Headache (41%), fatigue (36%), diarrhea (31%), bruising (30%), nausea (28%), anemia (27%), upper respiratory tract infection (26%), cough (24%), dizziness (23%), pyrexia (20%), pneumonia (20%), arthralgia (19%), peripheral edema (19%), rash (19%), neutropenia (16%), muscle spasms (16%), dyspnea (15%), hypertension (15%), vomiting (14%), constipation (13%), back pain (13%), urinary tract infection (12%), abdominal pain (12%), decreased appetite (11%) Tafasitamab Improved progression-free survival when added to lenalidomide compared to lenalidomide alone Increased risk of infection and infusion reactions; possible liver damage Infusion-related reactions (74%), neutropenia (38%), fatigue (32%), upper respiratory tract infection (30%), anemia (28%), diarrhea (26%), cough (22%), pyrexia (22%), thrombocytopenia (21%), nausea (20%), headache (19%), peripheral edema (19%), abdominal pain (19%), decreased appetite (17%), pneumonia (17%), rash (15%), dizziness (15%), bronchitis (15%), sinusitis (15%) Lisocabtagene maraleucel (Liso-cel) High response rate in patients with relapsed or refractory DLBCL Increased risk of cytokine release syndrome (CRS) and neurotoxicity; possible infections and secondary malignancies CRS (42%), neutropenia (39%), anemia (30%), fatigue (30%), fever (29%), decreased appetite (28%), hypotension (27%), pneumonia (26%), nausea (25%), headache (23%), diarrhea (23%), cough (22%), vomiting (20%), leukopenia (20%), thrombocytopenia (20%), hypoxia (19%), encephalopathy (16%), tachycardia (16%), chills (15%), infusion-related reactions (15%), dizziness (14%), hypoxemia (14%), myalgia (13%), constipation (13%), rash (13%), peripheral edema (12%), abdominal pain (11%), dyspnea (11%), hypertension (11%), pruritus (11%) Umbralisib Oral administration and potential for improved progression-free Increased risk of infections and liver damage; possible Diarrhea (53%), fatigue (49%), nausea (34%), neutropenia (31%), anemia (28%), cough (26%), pyrexia (26%), upper respiratory tract
- 8. survival when added to ublituximab compared to ublituximab alone bleeding and skin cancers infection (23%), pneumonia (22%), headache (21%), rash (21%), abdominal pain (20%), decreased appetite (19%), vomiting (19%), peripheral edema (19%), thrombocytopenia Polatuzumab vedotin: Polatuzumab vedotin was approved by the FDA in 2019 for the treatment of relapsed or refractory DLBCL in combination with bendamustine and rituximab (BRP), following at least two prior therapies. In the updated table, polatuzumab vedotin is listed as a second-line treatment option for patients who are ineligible for ASCT. Acalabrutinib: Acalabrutinib is a Bruton's tyrosine kinase (BTK) inhibitor that was approved by the FDA in 2020 for the treatment of adult patients with relapsed or refractory DLBCL who have received at least two prior lines of systemic therapy. However, it should be noted that acalabrutinib is currently not listed in the NCCN guidelines as a standard treatment option for DLBCL. Tafasitamab: Tafasitamab is a CD19-directed antibody that was approved by the FDA in 2020 for the treatment of relapsed or refractory DLBCL in combination with lenalidomide. In the updated table, tafasitamab is not listed as a standard treatment option, but could be considered in certain cases as salvage therapy. Lisocabtagene maraleucel: Lisocabtagene maraleucel (liso-cel) is a chimeric antigen receptor (CAR) T- cell therapy that targets CD19. It was approved by the FDA in 2021 for the treatment of relapsed or refractory DLBCL after at least two prior therapies. In the updated table, liso-cel is listed as a potential option for patients with relapsed or refractory disease. Umbralisib: Umbralisib is a phosphoinositide 3-kinase (PI3K) delta inhibitor that was granted accelerated FDA approval in 2021 for the treatment of relapsed or refractory marginal zone lymphoma and follicular lymphoma. It has not yet been approved for the treatment of DLBCL.
- 13. Table 1: Diagnosis, Staging, and Initial Work-up Aspect Summary Initial Work-up - Includes complete physical examination, blood count, renal and liver function, and more. - Contrast-enhanced CT scan of neck, thorax, and abdomen is often performed at first presentation. - Bone-marrow biopsy with t(14;18) analysis is conducted in all patients to exclude lymphoma infiltration.
- 14. Aspect Summary Risk Stratification - IPI (International Prognostic Index) used for risk assessment at initial diagnosis. - CNS-IPI for estimating individual prognosis and CNS involvement risk. - Consider cerebral MRI and cerebrospinal fluid analysis for high CNS-IPI or neurological symptoms. - Additional risk factors discussed but not conclusive for CNS involvement. Comorbidity Assessment - Echocardiography, electrocardiogram, brain natriuretic peptide for anthracycline-based chemotherapy. - HIV, hepatitis B and C serology checked before treatment. Psychological Support - Psychological support offered to patients and families at initial diagnosis. Table 2: Histological Assessment at First Diagnosis Aspect Summary Histological Assessment - Histological assessment is the cornerstone of first diagnosis. - Standard immunohistochemistry panel used for B- and T-cell markers and cell of origin analysis. - MYC, BCL2, and BCL6 FISH analysis conducted as per WHO classification. - Assessment of human herpesvirus-8 status in relevant pleural or peritoneal effusion cases. Table 3: First-line Treatment of Low-risk Patients Aspect Summary Low-risk Patients- Rituximab-based chemoimmunotherapy for patients with an IPI below 2. - Equivalence of four vs. six cycles of R-CHOP chemotherapy in limited disease. - Abbreviated treatment and interim PET for selected patients with low-risk and limited-stage disease. - Consider consolidation radiotherapy or prolonging R-CHOP for positive interim PET cases.
- 15. Aspect Summary - Repeated biopsy for patients with progressive disease based on CT and PET after three cycles of R-CHOP. Table 4: First-line Treatment of Higher-risk Patients Aspect Summary Higher-risk Patients - Polatuzumab vedotin preferred in patients with IPI >2. - Use of anti-nausea medications and filgrastim to manage chemotherapy side effects. - Liposomal anthracyclines in patients with cardiovascular risk factors and age >60 years. - Clinical examination and CT/PET scans during treatment to assess response. - Consider early PET scans for patients with signs of early progression. Table 5: Survivorship and Follow-up After First-line Treatment Aspect Summary Survivorship and Follow- up - Psychological support and psychotropic drugs offered for anxiety. - Symptomatic treatment for polyneuropathy, dose reductions, and rehabilitation clinics recommended. - Avoid regular CT scans in asymptomatic patients due to lack of efficacy. - Recommended follow-up includes blood count, renal and liver function, LDH, and clinical examination. - Use chest X-ray and ultrasonography for selected cases. Table 6: Relapsed or Refractory Disease Aspect Summary Relapsed or Refractory Disease - Conduct a new biopsy to confirm malignancy and the former diagnosis. - CAR-T-cell therapy preferred in the second-line setting for eligible patients.
- 16. Aspect Summary - Alternative therapeutic antibodies, such as polatuzumab vedotin and tafasitamab with lenalidomide, used. - Epcoritamab and glofitamab considered for heavily pretreated patients. - Individualized treatment decisions for patients not recruitable to clinical trials. Table: Evolving Treatment Approaches for Diffuse Large B-cell Lymphoma (DLBCL) Aspect Summary CAR T-Cell Therapy - Three FDA-approved options for relapsed/refractory DLBCL patients who had at least two prior lines of therapy. - Recent approvals for second-line therapy (axicabtagene ciloleucel and lisocabtagene maraleucel) based on ZUMA-7 and TRANSFORM studies. TRANSFORM Study - TRANSFORM study compared liso-cel to standard-of-care (high-dose chemotherapy and autologous stem cell transplantation). - Liso-cel group showed significantly improved median event-free survival, progression-free survival, and complete response rate. - Low rates of grade 3 cytokine release syndrome (1%) and neurologic events (4%). Glofitamab - A promising CD20/CD3 bispecific monoclonal antibody for relapsed/refractory DLBCL patients. - Phase 2 trial results showed significant complete and objective response rates, with low-grade cytokine release syndrome. Bispecific Antibodies - Advantages include off-the-shelf access and favorable toxicity profiles. - Longer follow-up needed to determine response durability beyond 1 year. Future Directions - Questions about sequencing with CAR T-cell therapy and combining bispecific antibodies with other regimens. - Ongoing studies of bispecific antibodies in combination with chemoimmunotherapy and other treatments.
- 17. Table: Axi-Cel Therapy for Relapsed/Refractory Large B-cell Lymphoma Aspect Summary Response Rates - In a multicenter phase 2 trial, 82% of patients with refractory large B-cell lymphoma had an objective response, and 54% had a complete response with axi-cel therapy. Comparison to Existing Therapies - Axi-cel response rates compare favorably to SCHOLAR-1 study results for existing therapies (objective response rate of 26% and complete response rate of 7%). Duration of Response - Responses were ongoing in 42% of patients, including 40% with a complete response, with the emergence of a plateau in response duration at 6 months. Monitoring for Improved Response - Consider monitoring patients without a complete response at the first assessment for an opportunity for improvement, as consolidation with allogeneic stem-cell transplantation has high treatment-related mortality. Overall Survival - Median overall survival not reached; 18-month overall survival rate of 52%. Ongoing durable remissions observed at 24 months. Consistency Across Covariates - Responses consistent across key covariates, including CD19 status and T-cell phenotypes. Need for Prospective Data - Prospective data needed to understand the influence of disease biology (e.g., double- and triple-hit lymphomas) on CAR T-cell therapy outcomes. Centralized Manufacturing and Coordination - Confirmed feasibility and reliability of centralized manufacturing and coordination of leukapheresis procedures. Feasibility in Medical Facilities - Axi-cel safely administered even in centers with no prior CAR T- cell therapy experience. Management of Cytokine Release Syndrome - Effective implementation of algorithms for cytokine release syndrome management; incidence decreased over time. Safety Compared to Allogeneic Stem-Cell Transplant - Favorable 3% rate of death during treatment compared to allogeneic stem-cell transplantation rates. Correlation of CAR T-Cell Levels with Response - CAR T-cell levels during the first 28 days correlated with objective response, supporting the potential for higher levels to augment efficacy.
- 18. Aspect Summary Biomarker Analysis - Serum biomarker analysis confirmed associations with the cytokine release syndrome and neurologic events, aiding in understanding adverse events. Use of Immunosuppressive Agents - Use of tocilizumab or glucocorticoids did not appear to affect overall response. Future Optimization and Combinations - Ongoing exploration of CAR construct and manufacturing optimization and combination strategies with immunomodulatory agents. Table 1: Current First-Line Treatment of DLBCL Treatment Patient Population Key Findings R-CHOP Elderly patients (60-80 years) Improved complete response rates (76% vs 63%) compared to CHOP. Improved EFS and OS without increased toxicity. 10-year PFS: 36.5% with R-CHOP vs 20% with CHOP alone. 10-year OS: 43.5% with R-CHOP vs 27.6% with CHOP alone. [16,17] R-CHOP Young patients (18- 60 years) Increased complete response rates, improved EFS, and OS. 6- year EFS: 74.3% with R-CHOP+rituximab vs 55.8% without. 6- year OS: 90.1% with R-CHOP+rituximab vs 80.0% without. [18,19] R-CHOP Elderly patients (≥60 years) No significant difference in outcomes between six and eight cycles of R-CHOP. [20] R-CHOP Standard First-Line Therapy Six cycles of R-CHOP established as standard first-line therapy for DLBCL. Table 2: Beyond Standard R-CHOP Therapy Treatment Patient Population Key Findings R-ACVBP Young patients (18-59 years) Significantly improved EFS and OS but logistically complex and with high risk of hematological toxicities. Limited to patients with aa-IPI scores of 1. [21] DA-EPOCH-R Not specified No demonstrated significant benefit over R-CHOP. [22] High-dose chemotherapy (HDC) Not specified No demonstrated significant benefit over R-CHOP. [23,24,25]
- 19. Treatment Patient Population Key Findings Novel agents with R- CHOP Not specified None showed significant success despite promising early signals of efficacy. [26-31] Lenalidomide Elderly patients responding to R-CHOP Maintenance therapy with lenalidomide prolonged PFS but no OS benefit. Controversial due to toxicities. [32] Polatuzumab vedotin (pola-R-CHP) Intermediate to high-risk DLBCL (IPI score ≥ 2) Improved PFS over R-CHOP. No significant OS difference. [37,38] Polatuzumab vedotin (pola-R-CHP) Intermediate to high-risk DLBCL (IPI score ≥ 2) Less subsequent treatment required with pola-R- CHP. [37,38] Table 3: Low Risk Patients with Limited-Stage Disease Treatment Patient Population Key Findings R-CHOP Limited stage (I or II) with aa-IPI of 0 and non-bulky disease (< 7.5 cm) Highly favorable outcomes with 6- year PFS approximating 90% and OS at 95%. [18,19] CHOP + Radiation Limited stage disease Non-inferior to eight cycles of CHOP alone but inadequate by modern standards. [43,44] Four cycles of R-CHOP + Two additional rituximab doses Young patients (18-60 years) with limited stage and non-bulky disease (< 7.5 cm), good ECOG performance status (0-1), and normal serum LDH levels Non-inferior to standard six cycles of R-CHOP. [46] PET Response-Adapted Approach Limited stage (non-bulky < 10 cm) and aa-IPI of 0 Four cycles of R-CHOP in case of negative interim PET resulted in excellent survival. [47] PET Response-Adapted Approach Limited stage (aa-IPI scores of 0) A negative interim PET allowed patients to receive four cycles, while a positive interim PET led to six cycles. Non-inferiority demonstrated. [48] Table 1: Current First-Line Treatment of DLBCL
- 20. Treatment Category Treatment Regimen Patient Criteria Outcomes Standard-Risk DLBCL R-CHOP chemoimmunotherapy All standard-risk DLBCL patients 6 cycles, 21-day cycle Intermediate/High- Risk Pola-R-CHP IPI score of 2–5, cost not a limiting factor 6 cycles, 21-day cycle Low Risk (Limited- Stage) Abbreviated R-CHOP Limited stage (I or II) with aa-IPI of 0, non- bulky 4 cycles, 21-day cycle Abbreviated R-CHOP (FLYER Criteria) Limited stage, non- bulky, good ECOG, normal LDH 4 cycles R-CHOP followed by 2 cycles rituximab PET-Guided Approach For selected "low- risk" DLBCL cases 3-4 cycles R-CHOP followed by consideration of consolidation radiation therapy, guided by PET imaging Table 2: High-Risk Genetics in DLBCL High-Risk Genetics Treatment Regimen Patient Criteria Notes Single MYC Rearrangement R-CHOP chemoimmunotherapy All patients with single MYC rearrangements 6 cycles, 21-day cycle Double-Expressor DLBCL R-CHOP chemoimmunotherapy All patients with double- expressor DLBCL 6 cycles, 21-day cycle Double-Hit or Triple-Hit DA-EPOCH-R or R-CHOP Patients with double-hit or triple-hit genetics 6 cycles, 21-day cycle for both DA-EPOCH-R and R-CHOP Consideration of consolidation therapies Table 3: Special Considerations Patient Group Treatment Regimen Patient Criteria Notes Elderly and Frail R-miniCHOP or other regimens Patients aged over 80 years or frail patients Consideration of alternative regimens Disease Site- Specific Various Based on primary site of the disease Tailored treatment approaches for specific disease sites
- 21. Patient Group Treatment Regimen Patient Criteria Notes DLBCL in HIV/AIDS DA-EPOCH-R or Abbreviated EPOCH- RR Patients with HIV- associated DLBCL Consideration of response- adapted strategies Table 4: Role of CNS Prophylaxis in DLBCL CNS Prophylaxis Strategy Patient Criteria Preferred Method Notes HD-MTX High CNS-IPI scores, high- risk sites Over IT prophylaxis Intercalated or at the end of induction Intrathecal (IT) MTX Variable based on guidelines Less preferred May not significantly reduce CNS relapse Table 1: Treatment Options for Relapsed or Refractory DLBCL Patient Eligibility Treatment Regimen Notes Transplant Eligible R-ICE, R-DHAP, R-GDP, Pola-R-ICE High-dose chemotherapy followed by ASCT is a curative option Transplant Ineligible R-GDP, R-GEMOX, Pola-BR, Pola-R-ICE Options for patients ineligible for ASCT or CAR-T therapy CAR-T Therapy Eligible CD19-directed CAR-T therapies Axicabtagene ciloleucel, tisagenlecleucel, lisocabtagene maraleucel for eligible patients CAR-T Therapy Ineligible Pola-BR, Tafasitamab + Lenalidomide, Loncastuximab tesirine, Selinexor Options for patients not eligible for ASCT or CAR-T therapy Table 2: Use of Next-Generation Sequencing in DLBCL Molecular Classification System Clinical Implications Current Clinical Practice Molecular Classification Systems Identification of unique genomic subtypes Subtyping DLBCL based on molecular classification systems is not yet a standard practice in current clinical treatment decisions. C1 to C5, BN2, A53, EZB, ST2, MCD, N1 Genetic clusters with distinct signatures These classifications are based on whole exome and deep targeted sequencing and may guide future clinical trials.
- 22. Molecular Classification System Clinical Implications Current Clinical Practice LymphGen Probabilistic Classifier Assign patients to genetic subtypes Currently, routine genomic profiling's value in frontline or relapsed treatment selection is unclear. Heterogeneity of DLBCL Potential rational explanation for targeted therapy failures Challenges, costs, and implications on treatment selection need further study.