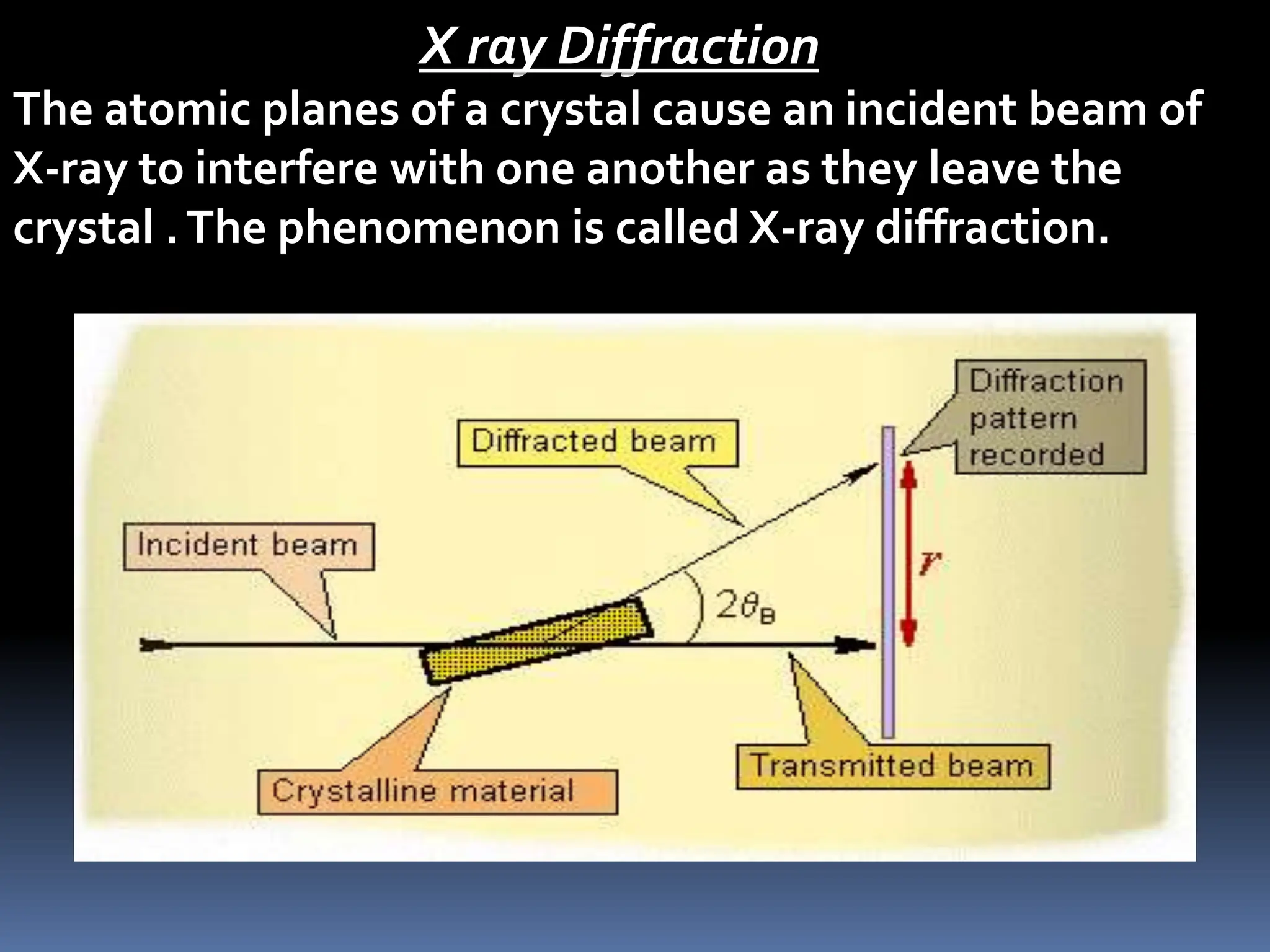
The document discusses X-ray diffraction and the structure factor in determining crystal structures. It provides background on how X-rays were first used by Max Von Laue and William Bragg to diffract off crystal planes. Bragg's law relates the wavelength of X-rays to the diffraction pattern. The structure factor describes the amplitude and phase of diffracted waves from crystal lattice planes and is essential for determining 3D electron density and crystal structures. Methods for X-ray diffraction including Laue, rotating crystal, and powder are described. Expressions for calculating the structure factor of simple cubic, body-centered cubic, and face-centered cubic unit cells are also provided.










![I = 𝐹 2 = F*F
= [ 𝑖 𝑓𝑗cos 2𝜋( 𝑢𝑗ℎ′ + 𝑣𝑗𝑘′ + 𝑤𝑗𝑙′)]2 + [ 𝑖 𝑓𝑗cos 2𝜋( 𝑢𝑗ℎ′ + 𝑣𝑗𝑘′ + 𝑤𝑗𝑙′)]2
where F* is the complex conjugate of F .It is to be noted
that ,whereas the space lattice of a crystal structure may
be determined from the position of diffraction lines in the
X-ray diffraction pattern , the determination of basis is
much more complicated and requires the knowledge of
intensity of various diffraction lines .Thus the concept of
structure factor carries a special importance in the context
of structure determination .](https://image.slidesharecdn.com/presentationatomicandlaser-240405083551-58ffadb7/75/jNVU-Presentation-on-atomic-and-laser-ppsx-12-2048.jpg)

![Since both the atoms are identical ,equation (5) gives the value of F
as
F(h’ k’ l’) = f 𝑗 𝑒𝑖2𝜋(𝑢𝑗ℎ′+𝑣𝑗𝑘′+𝑤𝑗𝑙′)
= f[1+𝑒𝑖𝜋(ℎ′+𝑘′+𝑙′
] ……(7)
the expression within the square brackets represents the structure
factor for bcc crystal .Here it has been assumed that only one of the
eight corner of the cube is occupied and has the coordinate (0,0,0)
.The validity of this assumption can be verified by considering all
the corner position and using the fact that the contribution of each
corner atom is 1
8 .This yields the same structure factor as included
in equation (7)
we also find from equation (7) that the structure factor
becomes zero for odd values of (h’ + k’+ l’) ,since 𝑒𝑖𝜋𝑛 equals -1 if n is
odd .For even values of (h’ + k’+ l’) , F(h’ k’ l’) equals 2f and , from
equation (6) the intensity becomes proportional to 4𝑓2 .](https://image.slidesharecdn.com/presentationatomicandlaser-240405083551-58ffadb7/75/jNVU-Presentation-on-atomic-and-laser-ppsx-14-2048.jpg)
![Face Centered Cubic Crystal : An fcc unit cell has four identical atoms.
One of these atoms is contributed by corners and may arbitrary be
assigned coordinates (0,0,0) ,whereas the other three are
contributed by face centers and have the coordinates (0,0,0) ;
(1
2 , 0, 1
2 ) ; (1
2 , 1
2,0) and (0, 1
2, 1
2) .
F(h’ k’ l’) = f[1+𝑒𝑖𝜋(ℎ′+𝑙′)
+ 𝑒𝑖𝜋(ℎ′+𝑘′)
+ 𝑒𝑖𝜋(𝑘′+𝑙′)
]
Where the expression within the square brackets is the structure
factor for fcc the crystals . It is obvious that the structure factor is
non –zero only if h ,k ,l are all even or all odd and has a value equal
to 4 .Thus the amplitude becomes 4f and the intensity becomes
proportional to 16 f2 .](https://image.slidesharecdn.com/presentationatomicandlaser-240405083551-58ffadb7/75/jNVU-Presentation-on-atomic-and-laser-ppsx-15-2048.jpg)
