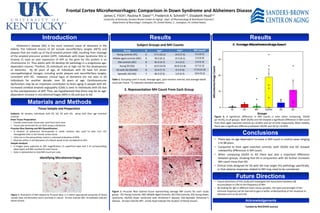
This study analyzed microhemorrhages (MH) in brain tissue samples from individuals with Down syndrome (DS), Alzheimer's disease (AD), and DS with AD, compared to age-matched controls. Brain tissue was stained to identify iron-rich MH near blood vessels. MH counts were significantly higher in samples from individuals with DS/AD compared to all other groups, as well as higher in AD samples compared to controls. The results suggest cerebrovascular dysfunction may contribute to brain aging in DS, and that DS combined with AD further increases MH counts more than AD alone.