This document discusses high-grade prostatic intraepithelial neoplasia (HGPIN). It notes that HGPIN is a precursor to some prostate cancers and an important finding on prostate biopsy. The document reviews the epidemiology of HGPIN, including its increased detection due to PSA screening and higher rates in African American and older men. A diagnosis of HGPIN alone has a slightly increased risk of finding prostate cancer on re-biopsy compared to an initial benign biopsy. Various chemoprevention strategies for prostate cancer are discussed, including vitamins, minerals, and hormonal manipulation, as HGPIN is a potential target for chemoprevention.

![1. Introduction
In 1969 prostatic intraepithelial neoplasia (PIN) was described by McNeal. [1], it is a
neoplastic propagation of prostatic epithelial cells that is confined to preceding prostatic ducts
or acini (glands). PIN was further considered and originally characterized intraductal
dysplasia by McNeal and Bostwick in 1986 [2]; the presently used term "prostatic
intraepithelial neoplasia" was later familiarized by Bostwick and Brawer in 1987, and
sanctioned by consensus at a 1989 conference [3, 4].
The era of prostate-specific antigen (PSA) screening has resulted in a rise in the number of
prostate biopsies being performed and as a result an increased detection of prostatic
intraepithelial neoplasia (PIN) on microscopic examination [5].
Major originator of prostate cancer is the high-grade prostatic intraepithelial neoplasia
(HGPIN) has shown that in many studies in current years. This review aims to simplify the
diagnostic terms used in pathology reports and the implications the terminology has upon
clinical management. Precisely, these article concentrations on HGPIN as well as analytical
terms using the word “atypical” in the prostate. It is important to identify and properly use the
term HGPIN to avoid misperception with other “atypical” components of the prostate, which
may vary with admiration to clinical consequence.
2. Epidemiology
By McNeal and Bostwick in 1986 described PIN, which was principally referred to as
“intraductal dysplasia,” was first defined to be a direct biological precursor to prostatic
adenocarcinoma [2]. The While in the introductory description of PIN the classification
comprised three different grades of dysplasia, at the present time, only HGPIN is described
by pathologists. This is characteristically the outcome of the deprived reproducibility among](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-2-320.jpg)
![pathologists of typical between low-grade PIN and benign prostate tissue [5, 6].
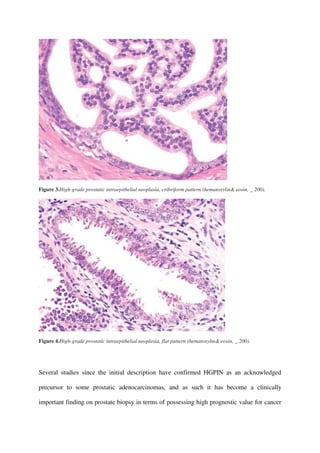
Figure 1.High-grade prostatic intraepithelial neoplasia, tufting pattern (hematoxylin& eosin, _ 400).
Figure 2.High-grade prostatic intraepithelial neoplasia, micropapillary pattern (hematoxylin& eosin, _ 400).](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-3-320.jpg)
![[7]. The proposed period frame to disease the progression of after HGPIN findings has been
reported to be between 29 and 36 months [8, 9]. In addition, with both prostate
adenocarcinoma and its multifocality has been accompanied a growth in the magnitude and
quantity of HGPIN foci [10-11]. HGPIN and carcinoma be inclined to preferentially include
the peripheral zone of the prostate [12, 13], and newly frequent biomarkers and molecular
changes such as TMPRSS2-ERG gene synthesis have been described in both entities (as
discussed further below) [14,15]. The general public incidence of HGPIN seems too similar
to that of prostate adenocarcinoma. Earlier autopsy studies discovered that HGPIN had a low
predominance in men during their third decade of life (7% in African Americans vs. 8% in
Caucasians) and progressively increased with advancing age (91% in African Americans vs.
67% in Caucasians) [16]. Sakr et al. [16,17] also experience a higher predominance of
HGPIN in African Americans than in age-matched Caucasian men, similar to prostate cancer.
However, the reported incidence of HGPIN in men participating in PSA broadcast after
needle biopsy varies extensively according to different series, ranging from 0 to 25% with a
mean incidence of 7.7% [7,18,19,20]. Once again, these varied findings may be explained by
the subjectivity of individual pathologists regarding what constitutes HGPIN and perhaps by
the technical aspects of tissue preparation [19]. The occurrence of HGPIN in Asian men is
similar to that in Western men. In a study of Korean men undergoing radical
cystoprostatectomy, 21% of the men who had no prostate cancer in the final pathology were
actually discovered to have HGPIN. As in studies of other races, HGPIN did not associate
with PSA, tumor volume, stage, grade, surgical margins, or lymphovascular incursion [21].
Han et al. [24] also found that both Western and Korean men have high rates of PIN
associated with prostate cancer in prostatectomy specimens. In a study from Singapore
consisting of 48 Chinese, 3 Malays, 2 Indians, and 3 other men with isolated HGPIN, 24% of
those who underwent repeat biopsies had prostate cancer. The authors also observed that](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-5-320.jpg)
![most cases of HGPIN affected only one core (79%), with 18% and 4% of cases affecting two
and three cores, respectively [22].
3. Summary
The diagnosis of HGPIN alone on needle core biopsy has only a slightly increased risk
(2324%) of finding prostate cancer on re-biopsy compared to an initial benign prostate biopsy
(20%). Therefore, an immediate re-biopsy within the first year may not be necessary for a
man with HGPIN without other clinical risk factors. Men with HGPIN should understand this
risk, be closely followed and be offered the chance to have re-biopsy when clinically
indicated. While the diagnosis of small atypical prostatic glands suspicious, but not
diagnostic of adenocarcinoma, also known as ASAP, requires an immediate re-biopsy to
establish a definitive diagnosis before radical treatment is initiated. The other “atypical”
prostate diagnosis should be clarified and not confused with above two entities. Furthermore,
failure to recognize HGPIN in prostate cancer research will lead to inaccurate conclusions,
which may impede the clinical diagnosis, treatment and prevention of prostate cancer.
4. Discussion
Over the last decade, the Chemoprevention Branch, Division of Cancer Prevention and
Control, National Cancer Institute, USA; have been developing drugs that will slow or stop
the progression to invasive cancer of precancerous (pre-invasive) lesions generally termed
‘intraepithelial dysplasia’ or ‘dysplasia'. After 40 short-term clinical trials are in progress
[23].Cancer prevention refers to the prevention or prolongation of the onset of carcinogenesis
by intervening with the agents to prevent, suppress, or reverse malignant transformation [24].
Why carcinoma prostate has been chosen for chemoprevention strategies? Carcinoma
prostate is considered to be ideal for chemoprevention because of the following reasons.
i.It has got a lingering course (long latency time).](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-6-320.jpg)
![ii.A high incidence rate.
iii.Availability of an effective marker like serum PSA.
iv.Possible availability of a genetic marker (P53)that can be used as an end point for prostate
cancer prevention.
v.Hormone dependency.
The study of prostate carcinogenesis and tumor progression is made difficult by the lack of
appropriate in vitro and in vivo models. High prevalence intra-epithelial neoplasia and latent
prostatic carcinoma, representing multiple steps in carcinogenesis to invasive carcinoma, are
relevant targets for cancer prevention. Webber et al. (2001) derived four tumorigenic cell
lines with progressive malignant characteristics. The MNU cell lines, in order of increasing
malignancy were; WPE1-NA22, WPE1NB14, WPE1-NB11, and WPE1-NB26. Development
of effective chemopreventive agents for human consumption requires conclusive evidence of
their efficacy in animal models that have relevance to human diseases. Transgenic
adenocarcinoma mouse prostate (TRAMP) is an excellent model of prostate cancer that
mimics progressive forms of human disease inasmuch as 100% of males develop
histologicalPINby8–12weeksofagethatprogresstoadenocarcinoma. In these animals, ornithine
decarboxylase (ODC) activity (> 3-fold) as well as protein expression (> 4-fold) was found to
be markedly higher in the dorsolateral prostate as compared with the nontransgenic
littermates, suggesting their suitability to determine the chemopreventive effect of alpha-
difluoromethylornithine (DFMO), an enzyme activated irreversible inhibitor of ODC, against
prostate cancer [25].
Prostatic intraepithelial neoplasia: A marker for high-risk groups and
a potential target for chemoprevention
The search for the precursor of prostatic adenocarcinoma has focused in recent years on two
histopathologic findings: high grade prostatic intraepithelial neoplasia (PIN) and atypical
adenomatous hyperplasia (AAH). Clinical studies suggest that PIN predates carcinoma by 10](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-7-320.jpg)
![years or more, with low grade PIN first appearing in men in their 30s. Unlike PIN, AAH is
weakly linked to carcinoma. It was stated that this family of cell lines with a common lineage
represents a unique and relevant model which mimics stages in prostatic intra-epithelial
neoplasia (PIN) and progression to invasive cancer, and can be used to study carcinogenesis,
progression, intervention, and chemoprevention [26]. Lopaczynski et al. (2001), in
preprostatectomy model observed that the IGF system appears to play an important role in
the development of prostate cancer by modulation of paracrine pathways, and also by
modulation of the concentrations of different stromal and epithelial IGFBP, which are
differentially expressed in the epithelium and the stroma. Nerve growth factor is capable of
stimulating a proliferative response via a high affinity Trk receptor present in normal and
malignant prostate epithelia, and alternatively can mediate apoptosis via the low affinity
p75NTR receptor that is progressively lost from the malignant prostate. As the role of each
stromal element involved in carcinogenesis becomes further defined, these elements offer
promising targets for new chemopreventive strategies [27]. There is a tremendous need for
exposure biomarkers, which need to function as intermediate end-points in cancer
chemoprevention studies. Imbalance between cell proliferation and cell apoptosis has been
considered a key factor in carcinogenesis. Prostatic intraepithelial neoplasia (PIN) is the most
likely precancerous lesion and represents the major target for chemoprevention of prostate
cancer [28]. High-grade prostatic intraepithelial neoplasia (HGPIN) has also been
investigated as an intermediate biomarker for prostate cancer. Endocrinetherapy changes the
morphology of PIN, hampering its identification by making it more closely resemble the
normal benign glands. Androgen deprivation therapy decreases the prevalence and extent of
PIN,suggesting that this form of treatment may play a role in chemoprevention [29, 30].
Cessation of endocrine therapy is likely to lead to renewed expansion of PIN, since PIN
continues to express androgen receptors and the cell cycle protein MIB-1under conditions of](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-8-320.jpg)
![low androgen levels. HGPIN have the advantage that it appears to be quite highly proximal to
the development of cancer and to be modifiable [31, 32]. Loss of expression of the pi-class
glutathione S-transferase enzyme GSTP1, which is associated with the hypermethylation of
dexoycytidine residues in the 5’-regulatory CG island region of the GSTP1 gene, is a near-
universal finding in human prostate cancer. Likewise high-grade PIN is completely devoid of
GSTP1. Hypermethylation of the 5’-regulatoryregion of the GSTP1 gene may serve as an
important molecular genetic biomarker for both prostate cancer and PIN [33].
Animal models in defining efficacy of chemoprevention agents
against prostate cancer
Animal models are crucial in preclinical efficacy testing of chemoprevention agents. The
most feasible, realistic, and potentially effective target for prostate cancer chemoprevention is
a progression of prostatic intraepithelial neoplasia (PIN) to histologic cancer and from
histologic to clinically manifest cancer. There are transgenic mouse models for prostate
cancer and models for a PIN, but these have not yet been fully developed and evaluated in
chemoprevention studies. Human prostate cancer xenografts in mice and Transplantable
Dunning rat prostate carcinomas can be used to assess tumor growth inhibition. PIN occurs
mostly in these two models, and metastases are frequent in some transgenic models and the
MNU-testosterone rate model [34]. The three most widely used carcinoma cell lines, DU-
145, PC-3, and LNCaP, developed between 1977 and 1980, have greatly contributed to our
present understanding of prostate cancer. These cell lines will further serve as useful models
for investigating the tumor progression, invasion, metastasis, new therapeutic strategies, drug
resistance and its reversal and chemoprevention[35].
Chemopreventive strategies
Chemopreventive agents (CPA) are classified as (i) Inhibitors of initiation, (ii) Anti-
promotional agents and (iii) Inhibitors of progression. The various bioactive compounds
which act as anti-oxidants and scavengers combine to the target tissues and protect the body](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-9-320.jpg)
![against the harmful effect of free radicals which would otherwise combine to these tissues
(Figure 1). A free radical is an atom or molecule that has one or more unpaired electrons its
consequent tendency to acquire an electron makes it highly reactive. Antioxidant is defined as
any compound, which breaks the free radical reaction chain. Different mechanisms have been
proposed for various kinds of chemopreventive agents. Recommended chemoprevention
strategies based on these mechanisms are (i) the development of better technology for early
diagnosis, (ii) the development of multiple agents that block intralesional proliferation at
steps along the signal pathway of mitotic signal transduction and along the signal pathway of
sythnesis of daughter cell components, (iii) the development of nontoxic anti-inflammatory
agents, antioxidants, antimutagens, and proapoptotics, (iv) the avoidance of ‘clonal escape’
through use of drug combinations, and (v) the use of computer-assisted quantitative image
analysis to assay modulation of surrogate end points in chemoprevention clinical trials [36].
The ideal chemopreventive agent should be nontoxic, efficacious, easily available and
inexpensive. CPA can be provided as a part of modified diet or synthetic derivatives (Pills).
The various possible chemopreventive agents and measures for prostate cancer are:
• Vitamins: A, D, C and E
• Minerals: Selenium
• Carotenoids: Lycopenes
• Dietary fat
• Hormonal manipulation: Flutamide, Dehydroepiandrosterone, 5alpha-reductase inhibitor
• Miscellaneous compounds: Nonsteroidal antiinflammatory drugs, Green tea.
1.Vitamin A
Fat soluble vitamin, which occurs in nature as retinal and dehydroretinol. Synthetically it is
derived from carotenoids (beta carotene). Carotenoids retinal and retinoic acid interact with
specific intracellular receptors and affect protein synthesis finally controlling cell chromatin,](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-10-320.jpg)
![cell growth and cell differentiation [37]. There are over six human retinoid receptors (RAR
[α, β and χ] and PXR [α, β and χ]) and all six belong to the steroid receptor superfamily.
Vitamin A and its analogues modulate the growth and differentiation of cancer cells
presumably by activating gene transcription via the nuclear retinoic acid receptor (RAR)
alpha, beta, and gamma and retinoid X receptor (RXR) alpha, beta, and gamma [38].
Fenretinide (N-4-hydroxyphenyl retinamide) (4HPR) a vitamin A analogues have been found
to be relatively nontoxic in preclinical experiments and early clinical trials. Experimental
studies have shown in Mouse prostate reconstitution model system, Fenretidine showed a
lowered incidence of tumor by 49%
Molecular Oxygen (toxic)
Water (4e-acquired)
Reduction [univalent reduction]
[Bivalent reduction]
Free radical (e- with high En-level) seavengers
-Superoxide radical superoxide dismutase
-hydrogen peroxide catalase/peroxidase
If left
Effect
-Hydroxyl redical (OH) -Cell membrane
-Singlet Oxygen -Cyt. Proteins](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-11-320.jpg)
![-DNA
-Initiations of carcinogenesis
Figure 1.Mitochondrial cytochrome oxidase system.
and tumor mass by 52% as compared to normal fed animals and in LN Cap cell line culture.
Fenretidine produced 82–95% suppression of cell growth, partial arrest in G1 phase of cell
cycle and marked increase in the number of apoptic cells [39].The chemopreventive effect of
retinoids is most likely exerted at the tumor promotion phase of carcinogenesis. Retinoids
block tumour promotion by inhibiting proliferation, inducing apoptosis, inducing
differentiation, or a combination of these actions [40]. It has been proposed that in prostate
cancer, urokinase-type plasminogen activator (u-PA) is the key enzyme which occupies a
place at the apex of the proteolytic cascade and initiates the degradative process needed for
ability to invade and metastasize. All-trans retinoic acid (RA) reduced the ability of u-PA-
mediated degradation of fibronectin and laminin [41]. Animal studies proved that prostate
carcinoma tissue contains 5–8 times less retinoic acid than normal prostate or BPH. The
lower retinoic acid content may contribute to cancer development or just be a marker of
cellular transformation, possibly explained by a more rapid degradation. However, the Alpha-
tocopherol Beta-carotene Cancer Prevention study in a placebo controlled, randomized trial
showed that beta carotene treatment resulted in increase in cancer at the lungs, prostate and
stomach. This effect was more evident in alcoholics, which might shift the dose response
curve. Another possibility could be that an excess administration may reduce the efficacy or
even promote tumors [24].
2. Vitamin D
Calcitriol (1,25-D3) is the active form of vit.D which is produced in the skin ultraviolet
radiation of 7-dehydrocholestol which is the active form of vit.D. Epidemiological studies
have shown that a lower 1,25-D3 is associated with increased incidence of carcinoma prostate](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-12-320.jpg)
![and an inverse relationship was observed between skin exposure and mortality rate for
carcinoma prostate [42, 43]. Animal studies have proved that vit. D3
inhibit cell proliferation, promote cell differentiation and selectively decreases level of type
IV collagen in carcinoma prostate tumor cells [44]. Human trials have shown that receptors
for vitamin D (1alpha, 25dihydroxyvitamin D3) exist in human prostate cancer cell lines like,
LN Cap, PC-3 and DU 145 [43]. The ubiquitous presence of vitamin D receptor and 24-
hydroxylaseactivity in human prostatic carcinoma cells suggests new alternatives for the
pharmacological treatment of advanced prostatic cancer and implies that chemoprevention
strategies could also make use of this endocrine axis [45]. It has been observed that vitamin D
and its analogoues induce cell cycle arrest and apoptosis in the premalignant adenoma cells
[46].
3. Vitamin E
It occurs in the nature as a mixture of several closely relative compounds called as
tocopherols. It is an important natural antioxodant and scavenger. Due to its lypophilic
character it accumulates in circulating lipoproteins, cellular membranes and fat deposits,
where it reacts very rapidly with molecular oxygen and free radicals. It acts as a scavenger
for these compounds, protecting unsaturated fatty acids from per oxidation reaction and
protects cellular respiration by stabilizing coenzyme-Q. Animal studies have shown that
dietary vit. E produces 30–60% inhibition of induced carcinogenesis due to its ability to
inhibit synthesis of nitrosamine compounds. In human alpha Tocopherol Bet-Carotene
Cancer Prevention Study (1995) showed that man receiving vit. E had a 34% lower incidence
of carcinoma prostate [47, 48]. Eichholzer et al. (1996) in their study observed that men with
low plasma levels of vit. E had an increased risk of carcinoma prostate [49].](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-13-320.jpg)
![4. Vitamin C
Vitamin C is a six carbon organic acid with structural similarity to glucose. It is a potent
reducing (antioxidant) in several hydroxylation reaction making it capable of reducing
compounds like molecular oxygen and nitrates (scavenger). It inhibit malignant
transformation, decreases chromosomal damage in the cell [50]. Diets mainly comprising of
fruits and vegetables rich in vitamin C are associated with low incidence of cancer of
oesophagus, stomach, colon, skin
and lung. A decreased risk of lower urinary tract cancer has been shown with increased
vitamin C intake in a study of patients matched for age, sex and ethnic groups to two
population based controls [51]. Animal studies have shown that when treated with vit. C both
tumor cell lines showed reduced viability for both DU145 (androgen independent) and LN
Cap (androgen dependent). Vitamin C induces these changes through production of hydrogen
peroxide, which in turn produces free radicals that damage the cells [52].
5. Selenium
It is an important component of metalloenzyme glutathione peroxidase, which destroys
peroxidase in cytosol. It acts as a synergistic anti-oxidant with vitamin E. Epidemiological
studies have shown that the risk of cancer for patients with low serum selenium levels
reported up to twice that of subjects with high levels [53, 54]. A significantly low risk of
developing prostate cancer has been observed in men receiving supplemental selenium.
6. Lycopene
Lycopene is carotenoids which are found in high levels in some fruits and vegetables (cooked
or raw tomatoes and watermelon). Lycopene acts as an antioxidant by preventing damage to
DNA by protecting 2-deoxy-guanosine against singlet oxygen damage. It suppresses insulin
like growth factor-1-stimulated cell proliferation. An important study showed that lycopene is
found in very high concentration in the prostate, adrenal and testes. Lycopene reduced levels
of serum protein thiol (an oxidative damage biomolecular marker) levels among prostate](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-14-320.jpg)
![cancer patients [55]. Prospective randomized controlled trials have shown lower prostate
cancer risk in men with elevated plasma lycopene levels. Research results presented at 90th
annual meeting of the American Association for Cancer Research (AACR) described the
different effects of lycopene like reduction in tumor size, lowering of S-PSA levels and
reduction in grade of PIN.
7. Dietary fat
A study by the American Cancer Society has shown that obesity increased the risk of
prostatic cancer [56]. Epidemiological studies done to compare different populations around
the world revealed the relation of dietary fat and risk of prostate cancer [57]. The Analysis
has revealed that the mortality rate of prostate cancer is correlated with the estimated intake
of dietary fat. Essential fatty acids influence cellular proliferation, tissue invasiveness,
metastatic spread of tumors and immune response as well as cell surface receptors [58]. Long
chain n-3 fatty acids have inhibitory effect and n-6 fatty acids have stimulatory effects.
Likewise, men with increased levels of cholesterol have more risk of carcinoma prostate.
8. Hormonal manipulation
Flutamide
High-grade prostatic intraepithelial neoplasia (HGPIN) is believed to be a precursor for
prostatic adenocarcinoma. The prevalence of prostatic intraepithelial neoplasia (PIN)
increases with advancing age. Autopsy studies suggest that PIN may precede the
development of prostatic adenocarcinoma by up to 10 years. Autopsy studies reveal that
HGPIN is found in association with cancer in 63% to 94% of malignant and 25% to 43% of
benign prostates [59]. As such, HGPIN is believed to be a marker of increased risk. This
provides a potential opportunity for chemoprevention. Flutamide is one agent with potential
activity and limited side effects that may act to prevent or delay the onset of prostatic
adenocarcinoma in men with HGPIN. A clinical trial is currently underway to assess the
efficacy of flutamide [60].](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-15-320.jpg)
![9.Dehydro epiandrosterone (DHEA)
In male Wistar-Unilever rats after experimentally induction of prostate adenocarcinomas, it
was seen that nontoxic doses of DHEA confer significant protection against prostate
carcinogenesis in rats. The efficacy of delayed administration of DHEA suggests that the
compound confers protection against later stages of prostate cancer induction and can
suppress the progression of existing preneoplastic lesions to invasive disease [61].
5 alpha-reductase inhibitor
This compound is again under scrutiny as it has been observed that in male ACI/Seg rats,
which spontaneously develop prostate cancer, the 5alpha-reductase inhibitor FK143 may, at
specific doses, reduce the incidence of spontaneously developing prostate cancer [62].
10.Miscellaneous compounds
Nonsteroidal anti-inflammatory drugs
Nonsteroidal anti-inflammatory drugs (NSAIDs) play potential role sin chemoprevention of
colon cancer and others by inhibiting prostaglandin synthesis. Western and northern blot
analyses demonstrated that flufenamic acid (FA) inhibited the androgen receptor (AR)
expression at mRNA and protein levels when used in LNCaP cells, an androgen-responsive
human prostate carcinoma cell line .The Suppressed AR expression may be the cause of FA-
mediated inhibition of the androgen inducible gene expression. FA and other similar NSAIDs
may be potential candidates for chemoprevention of human prostate cancer by modulating the
expression of AR [63].
Green tea
Ornithine decarboxylase (ODC), a rate-controlling enzyme in the polyamine biosynthetic
pathway, is over expressed in prostate cancer (PCA) and prostatic fluid in humans, intonation
of ODC could be effectual against prostate cancer. Variety of animal tumor models and in
some human epidemiological studies, Green tea polyphenols (GTPs) posses strong
chemopreventive properties. in anchorage independent growth assay of LNCaP cells where](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-16-320.jpg)
![pretreatment of the cells with GTP was found to result in a dose-dependent inhibition of
colony formation, the similar effects of GTPs were observed [64]. In another study, the major
constituent of green tea EGCG [(-)-epigallocatechin3-gallate], was examined to understand
mechanisms of action. Growth assays, cell cycle analysis, and western blots for the
retinoblastoma protein (pRB) were examined with the Effects of EGCG on the cell
population. EGCG inhibited growth In each cell type, with a decrease in efficacy as cells
progressed from normal to cancer [65].
7. Conclusion
Several dietary and nutritional factors involve in the studies of onset and progression of
prostate cancer. Hence, it is possible that bioactive compounds (anti-oxidants)can be a part of
chemopreventive strategies for prostate cancer, like vits. A, D, C, and E, mineral like
selenium and carotenoids like lycopene. Besides the dietary fat inflection the various other
compounds and procedures suggested to have a chemopreventive effect are nonsteroidal anti
inflammatory drugs, green tea and hormonal manipulation (Flutamide,
Dehydroepiandrosterone, 5alphareductase inhibitor). Ongoing studies may bring the required
evidence to support what is still only a hypothesis at present on nutrition and prostate cancer.
However utter recommendation will have to await the results of long term prospective
clinical trials.
8. REFERENCE
[1] McNeal JE. Origin and development of carcinoma in the prostate. Cancer 1969;23: 24-
34.
[2] McNeal JE and Bostwick DG. Intraductal dysplasia: A premalignant lesion of the
prostate. Hum Pathol 1986;17:64-71.](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-17-320.jpg)
![[3] Bostwick DG and Brawer MK. Prostatic intra-epithelial neoplasia and early invasion in
prostate cancer. Cancer 1987;59:788-794.
[4] Bostwick DG and Srigley J. Premalignant lesions. In: Pathology of the prostate, Bostwick
DG (Ed), Churchill-Livingston, New York 1990. p37.
[5] Epstein JI, Grignon DJ, Humphrey PA, McNeal JE, Sesterhenn IA, Troncoso P, et al.
Interobserver reproducibility in the diagnosis of prostatic intraepithelial neoplasia. Am J
Surg Pathol 1995;19:873-86.
[6] Allam CK, Bostwick DG, Hayes JA, Upton MP, Wade GG, Domanowski GF, et al.
Interobserver variability in the diagnosis of high-grade prostatic intraepithelial neoplasia and
adenocarcinoma. Mod Pathol 1996;9:742-51.
[7] Bostwick DG, Cheng L. Precursors of prostate cancer. Histopathology 2012;60:4-27.
[8] Guzzo TJ, Kutikov A, Canter DJ, Tomaszewski JE, Magerfleish L, VanArsdalen K, et al.
The clinical and pathological history of prostate cancer progression in men with a prior
history of high grade prostatic intraepithelial neoplasia. Can J Urol 2008;15: 4174-8.
[9]. Lefkowitz GK, Taneja SS, Brown J, Melamed J, Lepor H. Followup interval prostate
biopsy 3 years after diagnosis of high grade prostatic intraepithelial neoplasia is associated
with high likelihood of prostate cancer, independent of change in prostate specific antigen
levels. J Urol 2002;168:1415-8.
[10]. Antonelli A, Tardanico R, Giovanessi L, Pesenti N, Gatti L, Zambolin T, et al.
Predicting prostate cancer at rebiopsies in patients with high-grade prostatic intraepithelial
neoplasia: a study on 546 patients. Prostate Cancer Prostatic Dis 2011;14:173-6.
[11]. Kronz JD, Allan CH, Shaikh AA, Epstein JI. Predicting cancer following a diagnosis of
high-grade prostatic intraepithelial neoplasia on needle biopsy: data on men with more than
one follow-up biopsy. Am J Surg Pathol 2001;25:1079-85.](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-18-320.jpg)
![[12]. Bostwick DG. Prospective origins of prostate carcinoma. Prostatic intraepithelial
neoplasia and atypical adenomatous hyperplasia. Cancer 1996;78:330-6.
[13]. Häggman MJ, Macoska JA, Wojno KJ, Oesterling JE. The relationship between
prostatic intraepithelial neoplasia and prostate cancer: critical issues. J Urol 1997;158:12-22.
[14]. Cerveira N, Ribeiro FR, Peixoto A, Costa V, Henrique R, Jerónimo C, et al. TMPRSS2-
ERG gene fusion causing ERG overexpression precedes chromosome copy number changes
in prostate carcinomas and paired HGPIN lesions. Neoplasia 2006;8:826-32.
[15]. Perner S, Mosquera JM, Demichelis F, Hofer MD, Paris PL, Simko J, et al. TMPRSS2-
ERG fusion prostate cancer: an early molecular event associated with invasion. Am J Surg
Pathol 2007;31:882-8.
[16]. Sakr WA, Grignon DJ, Haas GP, Heilbrun LK, Pontes JE, Crissman JD. Age and racial
distribution of prostatic intraepithelial neoplasia. Eur Urol 1996;30:138-44.
[17]. Sakr WA, Grignon DJ, Haas GP. Pathology of premalignant lesions and carcinoma of
the prostate in African-American men. Semin Urol Oncol 1998;16:214-20.
[18]. Montironi R, Mazzucchelli R, Lopez-Beltran A, Cheng L, Scarpelli M. Mechanisms of
disease: high-grade prostatic intraepithelial neoplasia and other proposed preneoplastic
lesions in the prostate. Nat Clin Pract Urol 2007;4:321-32.
[19]. Epstein JI, Herawi M. Prostate needle biopsies containing prostatic intraepithelial
neoplasia or atypical foci suspicious for carcinoma: implications for patient care. J Urol
2006;175:820-34.
[20]. Zynger DL, Yang X. High-grade prostatic intraepithelial neoplasia of the prostate: the
precursor lesion of prostate cancer. Int J Clin Exp Pathol 2009;2:327-38.
[21]. Han KS, Jeong IG, Joung JY, Yang SO, Chung J, Seo HK, et al. Prevalence of high-
grade prostatic intraepithelial neoplasia in prostate gland of Korean men: comparisons
between radical prostatectomy and cystoprostatectomy. Urology 2007;70:1100-3.](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-19-320.jpg)
![[22]. Tan PH, Tan HW, Tan Y, Lim CN, Cheng C, Epstein JI, et al. Is high-grade prostatic
intraepithelial neoplasia on needle biopsy different in an Asian population: a
clinicopathologic study performed in Singapore. Urology 2006;68:800-3.
[23]. Boone CW, Kelloff GJ. Biomarker end-points in cancer chemoprevention trials. IARC
Sci Publ 1997; 142: 273–280.
[24]. Ashish M Kamat, Donald L Lamm. Chemoprevention of urological cancer. J Urol 1999;
161: 1748–1760.
[25]. Gupta S, Ahmad N, Marengo SR et al. Chemoprevention of prostate carcinogenesis by
alpha-difluoromethylornithine in TRAM mice. Cancer Res 2000; 60(18): 5125–5133.
[26]. Webber MM, Quader ST, Kelinman HK et al. Human cell lines as an in vitro/in vivo
model for prostate carcinogenesis and progression. Prostate 2001; 47(1): 1–13.
[27]. Lopaczynski W, Hruszkewycz AM, Lieberman R. Preprostatectomy: A clinical model
to study stromal-epithelial interactions. Urology 2001; 57 (4 Suppl 1): 194–199.
[28]. Xie W, Wong YC, Tsao SW. Correlation of increased apoptosis and proliferation with
development of prostatic intraepithelial neoplasia (PIN) in ventral prostate of the Noble rat.
Prostate 2000; 44(1): 31–39.
[29]. Bostwick DG. Prostatic intraepithelial neoplasia is a risk factor for cancer. Semin Urol
Oncol 1999; 17(4): 187–198.
[30]. Bostwick DG, Montironi R, Sesterhenn IA. Diagnosis of prostatic intraepithelial
neoplasia: Prostate Working Group/ consensus report. Scand J Urol Nephrol Suppl 2000;
205: 3–10.
[31]. van der Kwast TH. Intermediate biomarkers for chemoprevention of prostate cancer.
IARC Sci Publ 2001; 154: 199–205.
[32]. Marshall JR. High-grade prostatic intraepithelial neoplasia as an exposure biomarker for
prostate cancer chemoprevention research. IARC Sci Publ 2001; 154: 191–198.](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-20-320.jpg)
![[33]. Brooks JD, Weinstein M, Lin X et al. CG island methylation changes near the GSTP1
gene in prostatic intraepithelial neoplasia. Cancer Epidemiol Biomarkers Prev 1998; 7.
[34]. Bosland MC. Use of animal models in defining efficacy of chemoprevention agents
against prostate cancer. Eur Urol 1999; 35(5–6): 459–463.
[35]. Webber MM, Bello D, Quader S. Immortalized and tumorigenic adult human prostatic
epithelial cell lines: Characteristics and applications. Part 3. Oncogenes, suppressor genes,
and applications. Prostate 1997; 30(2): 136–142.
[36]. Boone CW, Bacus JW, Bacus JV et al. Properties of intraepithelial neoplasia relevant to
cancer chemoprevention and to the development of surrogate end points for clinical trials.
Proc Soc Exp Biol Med 1997; 216(2): 151–165.
[37]. Lokshin A, Zhang H, Mayotte J et al. Early effects of retinoic acid on proliferation,
differentiation and apoptosis in nonsmall cell lung cancer cell lines. Anticancer Res 1999;
19(6B): 5251–5254.
[38]. Sun SY, Yue P,Mao L et al. Identification of receptor-selective retinoids that are potent
inhibitors of the growth of human head and neck squamous cell carcinoma cells. Clin Cancer
Res 2000; 6(4): 1563–1573.
[39]. Slawin K, Kadmon D, Park SH et al. Dietary fenrethinide, a synthetic retinoid,
decreases the tumor incidence and tumor mass of ras+myc-induced carcinomas in the mouse
prostate reconstitution model system. Cancer Res 1993; 53: 4461.
[40]. Niles RM. Recent advances in the use of vitamin A (retinoids) in the prevention and
treatment of cancer. Nutrition 2000; 16(11–12): 1084–1089.
[41]. Webber MM, Waghray A. Urokinase-mediated extracellular matrix degradation by
human prostatic carcinoma cells and its inhibition by retinoic acid. Clin Cancer Res 1995;
1(7): 755– 761.](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-21-320.jpg)
![[42]. Schwartz GG, Hulka BS. Is vitamin D deficiency a risk factor for prostate cancer
(Hypothesis)? Anticancer Res 1990; 10: 1307.
[43]. Skowronski RJ, Peehl DM, Feldman D. Vitamin and prostate cancer: 1,25-
dihydrovitamin D3 receptors and actions in human prostate cancer lines. Endocrinology
1993; 132: 152.
[44]. Schwartz GG, Wang MH, Zang M et al. Alpha,25- Dihydroxyvitamin D (calciferol)
inhibits the invasiveness of human prostate cancer cells. Cancer Epidemiol Biomark Prev
1997; 6: 727.
[45]. Miller GJ, Stapleton GE, Hedlund TE, Moffat KA. Vitamin D receptor expression 24-
hydroxylase activity, and inhibition ofgrowth by 1alpha,25-dihydroxyvitamin D3 in seven
human prostatic carcinoma cell lines. Clin Cancer Res 1995; 1(9): 997–1003.
[46]. Diaz GD, Paraskeva C, Thomas MG et al. Apoptosis is induced by the active metabolite
of vitamin D3 and its analogue EB1089 in colorectal adenoma and carcinoma cells: Possible
implications for prevention and therapy. Cancer Res 2000; 60(8): 2304–2312.
[47]. Albanes D, Heinonen OP, Huttunen JK et al. Effects of alphatocopherol and beta-
carotene supplements on cancer incidence in the alpha-Tocopherol Bet-Carotene Cancer
Prevention. Am J Clin Nutr 1995; 62 (Suppl): 1427S.
[48]. The alpha-Tocopherol Bet-Carotene Cancer Prevention Study. The effect of vitamin E
and beta-carotene on the incidence of lung cancer and other cancers in male smokers. New
Engl J Med 1994; 330: 1029.
[49]. Eilchholzer M, Stahelin HB, Gey KF et al. Prediction of male cancer mortality by
plasma levels of interacting vitamins: 17- year follow-up of the prospective Basel study. Int J
Cancer1996; 66: 145.
[50]. Benedict WF, Jones PA. Inhibition of transformation and oncologic progression by
ascorbic acid: A possible role in chemoprevention. In: Arnot MS, van Eys J, Wang YM, eds,](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-22-320.jpg)
![Molecular Interrelations of Nutrition and Cancer. New York: Raven Press, 1982: 351.
[51]. Nomura AM, Klonel LN, Hankin JH, Youshizava CN. Dietary factors in cancer of the
lower urinary tract. Int J Cancer 1991; 48: 199.
[52] Marmag C, Menon M, Balaji KC et al. Effects of vitamin C on the prostate cancer cells
in vitro: Effect on cell number,viability, and DNA synthesis. Prostate 1997; 32: 188.
[53] Lew EA, Garfinkel. Variations in mortality by weight among 750,000 men and women.
J Chron Dis 1979; 32: 563.
[54] Willet WC, Polk BF, Morris JS et al. Prediagnostic serum selenium and risk of cancer.
Lancet 1983; 2: 130.
[55] Rose DP, Boyer AP, Wyner EI. International comparisons of mortality rates for cancer
of the breast, ovary, prostate and 214 colon, and per capita food consumption. Cancer 1986;
58:2363.
[56] Karmali RA. Eicosonoids in neoplasia. Prev Med 1987; 16: 493.
[57] Rose DP. Dietary fatty acids and prevention of hormone responsive cancer. Proc Soc
Exp Boil Med 1997; 216: 224.
[58] Sakr WA, Partin AW. Histological markers of risk and the role of high-grade prostatic
intraepithelial neoplasia. Urology 2001; 57 (4 Suppl 1): 115–120.
[59] Alberts SR, Blute ML. Chemoprevention for prostatic carcinoma: The role of flutamide
in patients with prostatic intraepithelial neoplasia. Urology 2001; 57 (4 Suppl 1): 188– 190.
[60] Rao KV, Johnson WD, Bosland MC et al. Chemoprevention of rat prostate
carcinogenesis by early and delayed administration of dehydroepiandrosterone. Cancer Res
1999; 59(13): 3084–3089.
[61] Homma Y, Kaneko M, Kondo Y et al. Inhibition of rat prostate carcinogenesis by a
5alpha-reductase inhibitor, FK143. J Natl Cancer Inst 1997; 89(11): 803–807.](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-23-320.jpg)
![[62] ZhuW, Smith A, Young CY. A nonsteroidal anti-inflammatory drug, flufenamic acid,
inhibits the expression of the androgen receptor in LNCaP cells. Endocrinology 1999;
140(11): 5451– 5454.
[63] Gupta S, Ahmad N, Mohan RR et al. Prostate cancer chemoprevention by green tea: In
vitro and in vivo inhibition of testosterone-mediated induction of ornithine decarboxylase.
Cancer Res 1999; 59(9): 2115–2120.
[64] Khafif A, Schantz SP, al-Rawi M et al. Green tea regulates cell cycle progression in oral
leukoplacia. Head Neck 1998; 20(6): 528–534.](https://image.slidesharecdn.com/highgradeprostaticintraepithelialneoplasia-181011061638/85/High-grade-prostatic-intraepithelial-neoplasia-24-320.jpg)