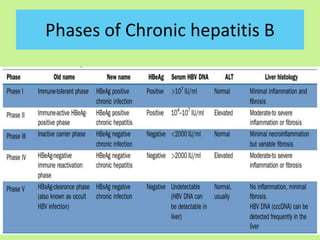
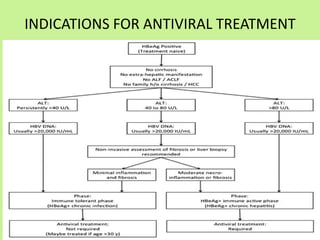
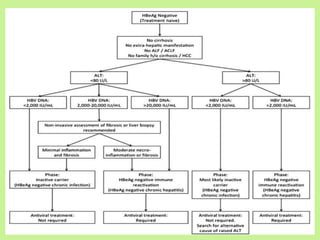
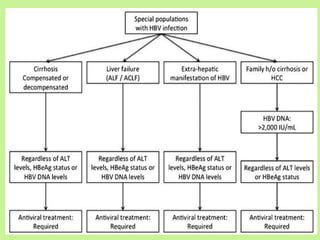
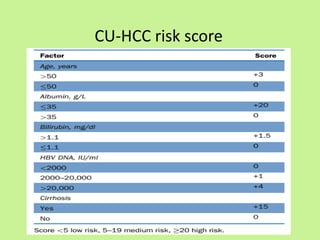
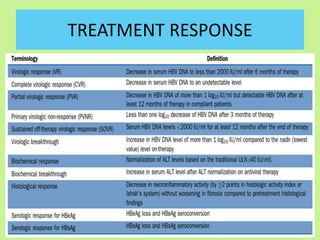
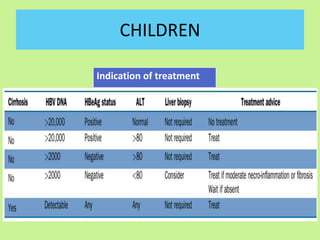
This document provides consensus statements from guidelines on the management of hepatitis B virus (HBV) infection in India. It covers epidemiology, vaccination, diagnosis, treatment goals and monitoring, antiviral treatment strategies, special populations including children and healthcare workers, and pregnancy considerations. The guidelines were developed through an expert panel discussion and aim to standardize management of HBV in India based on available evidence.