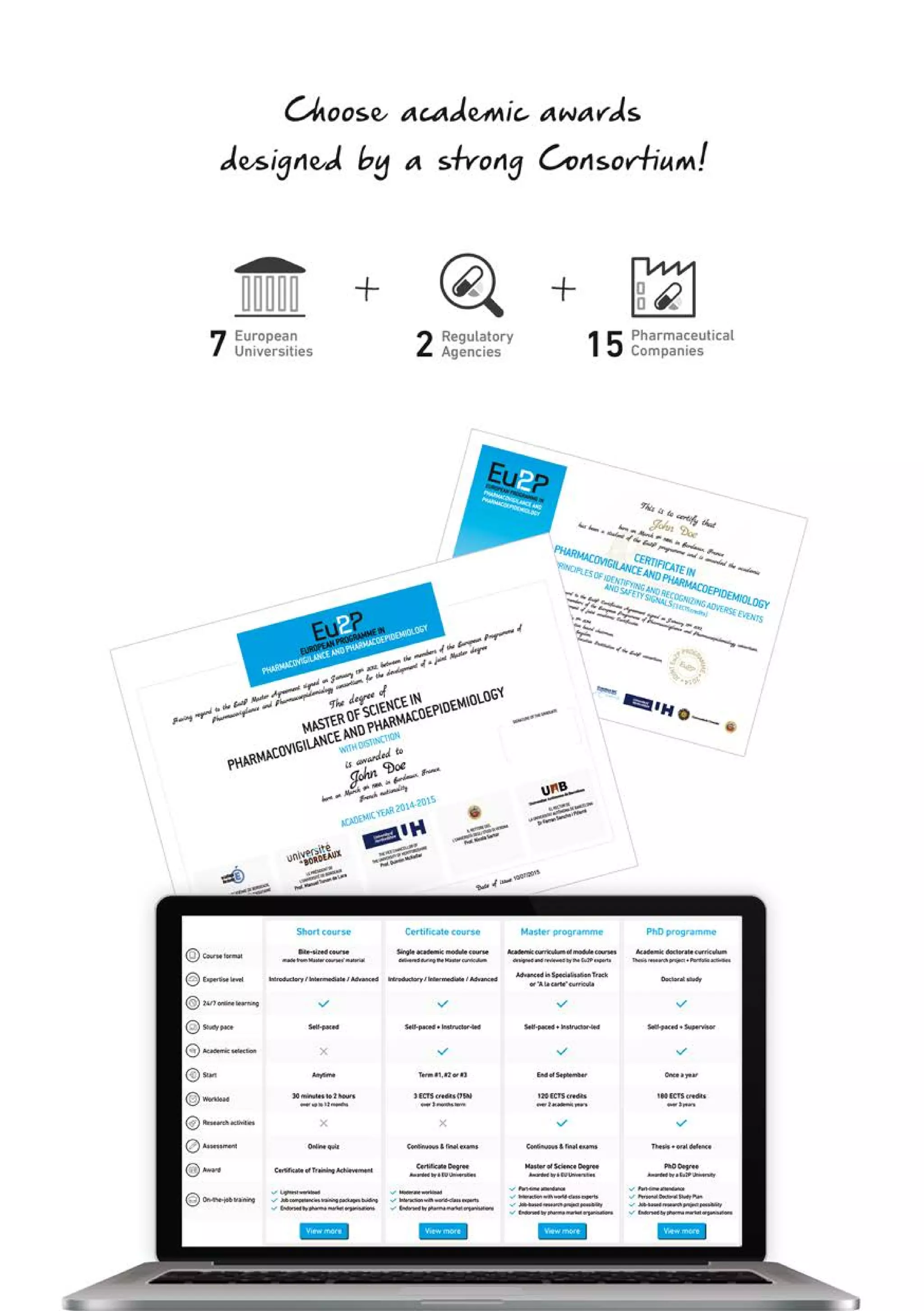
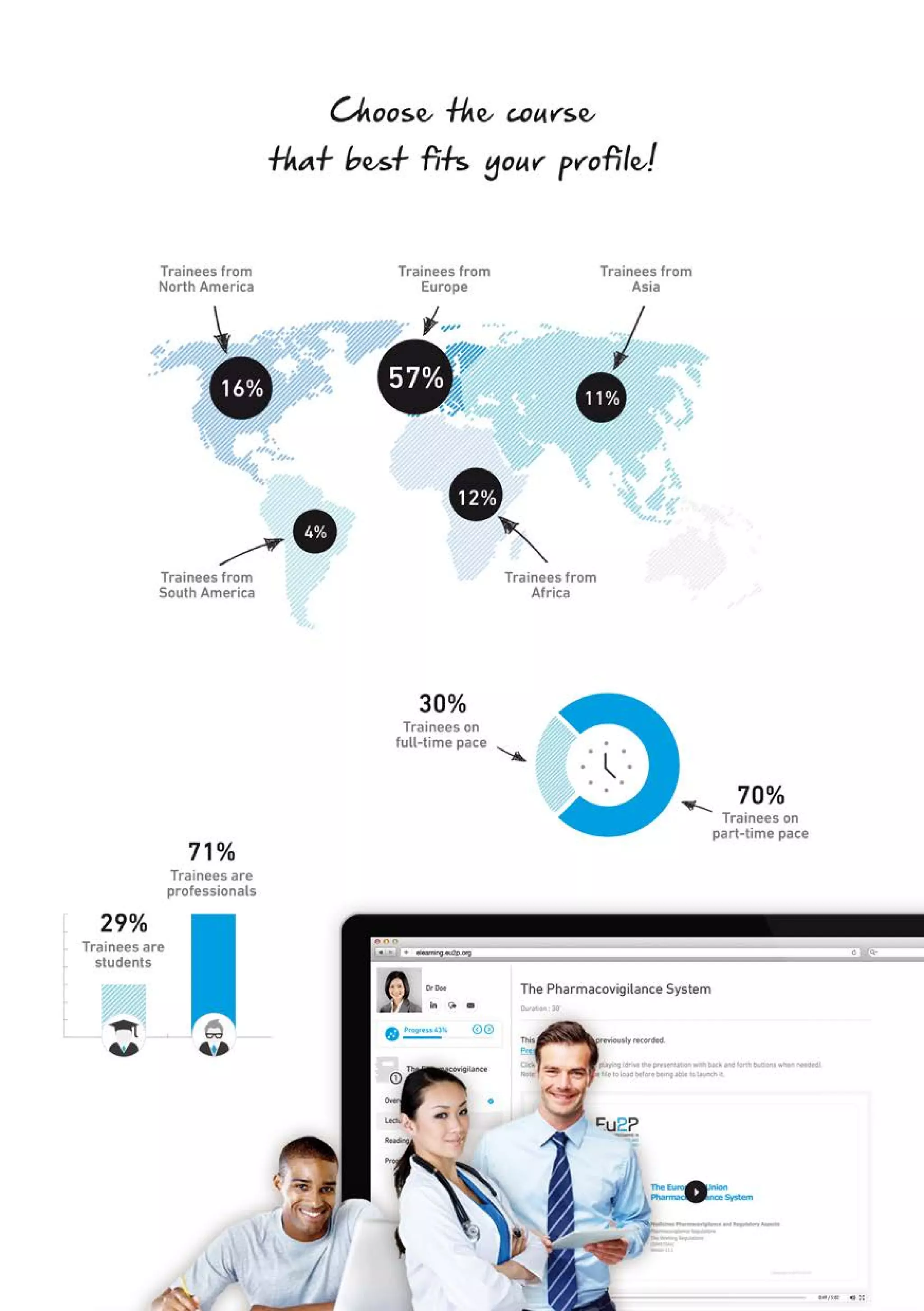
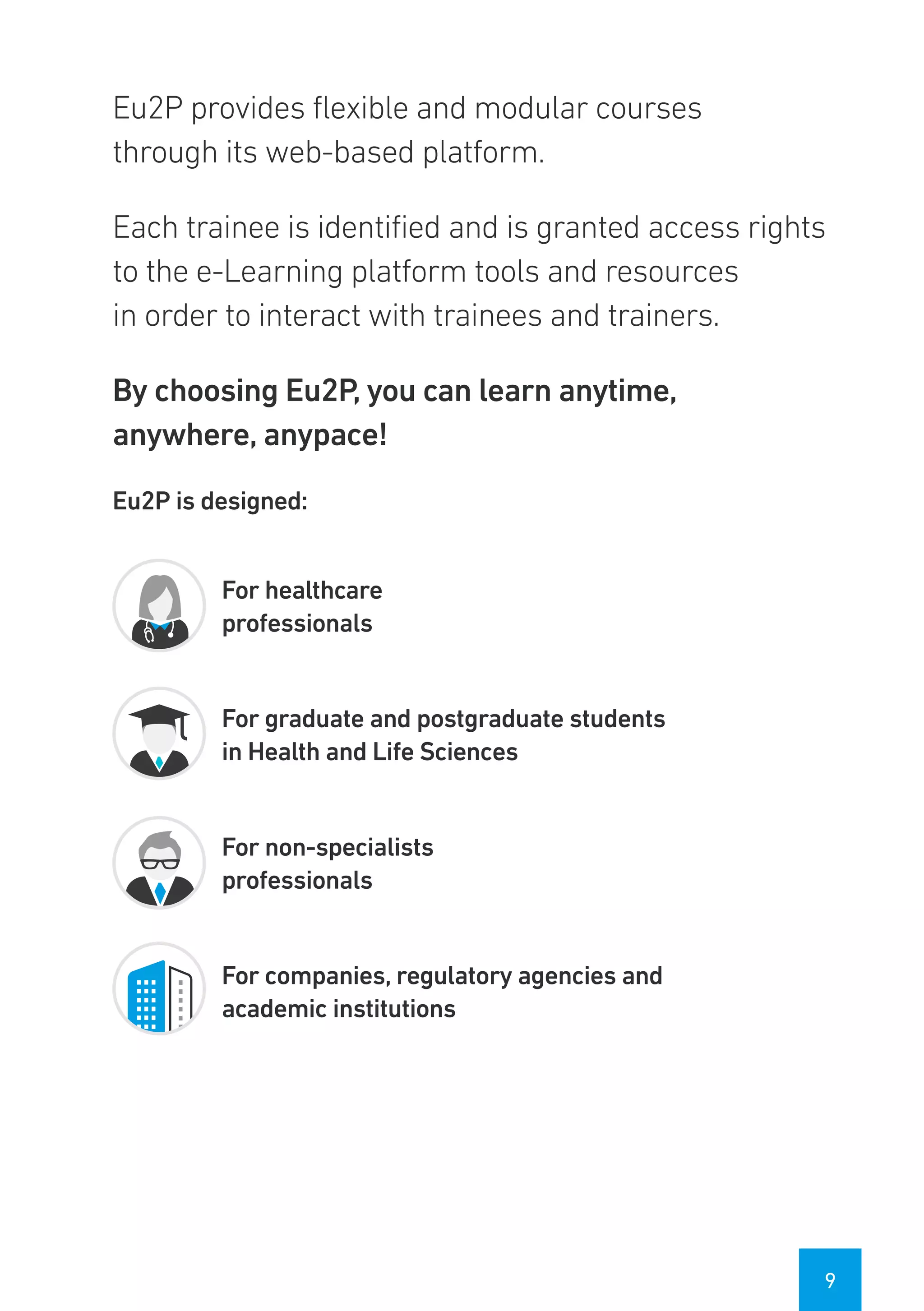
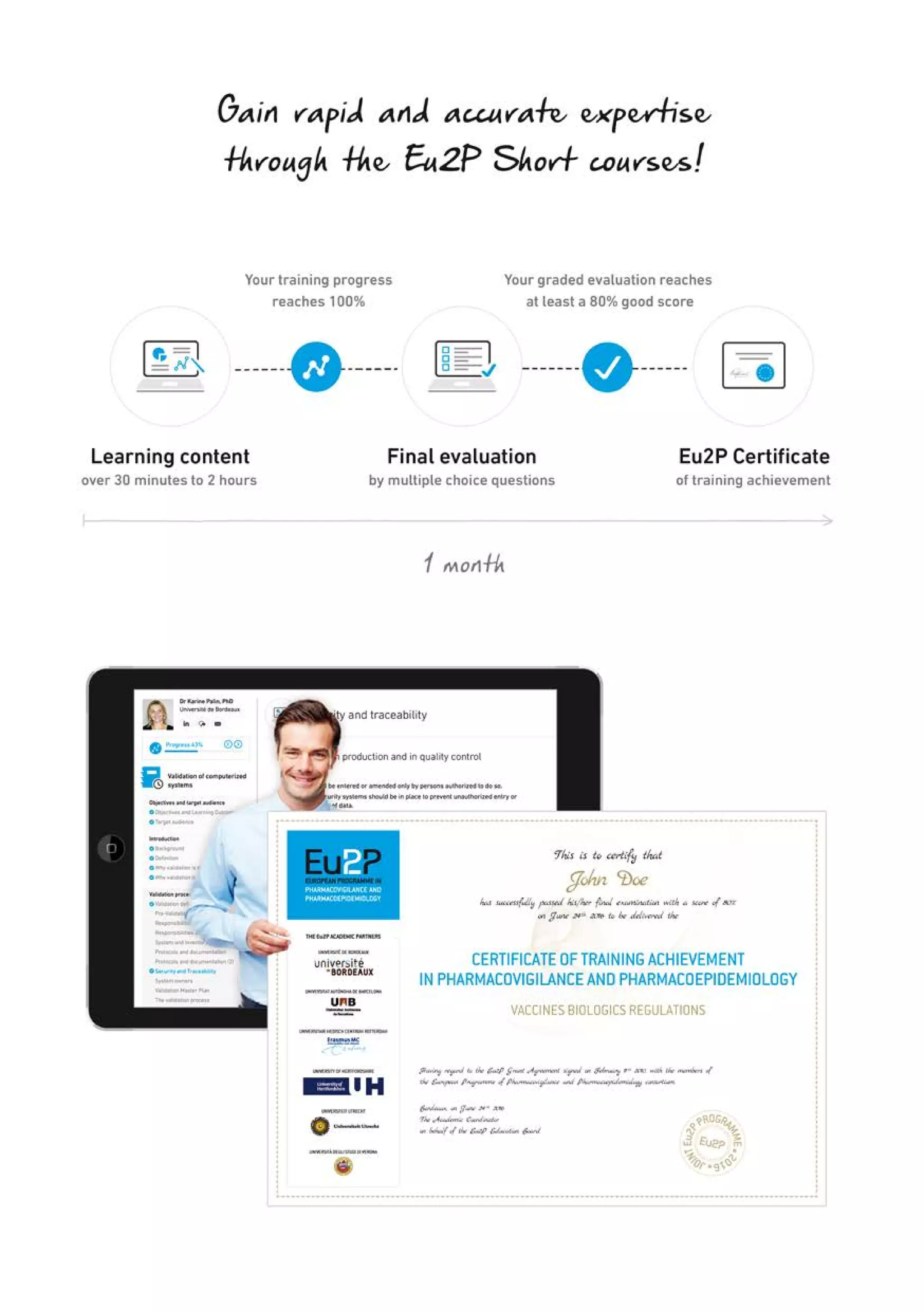
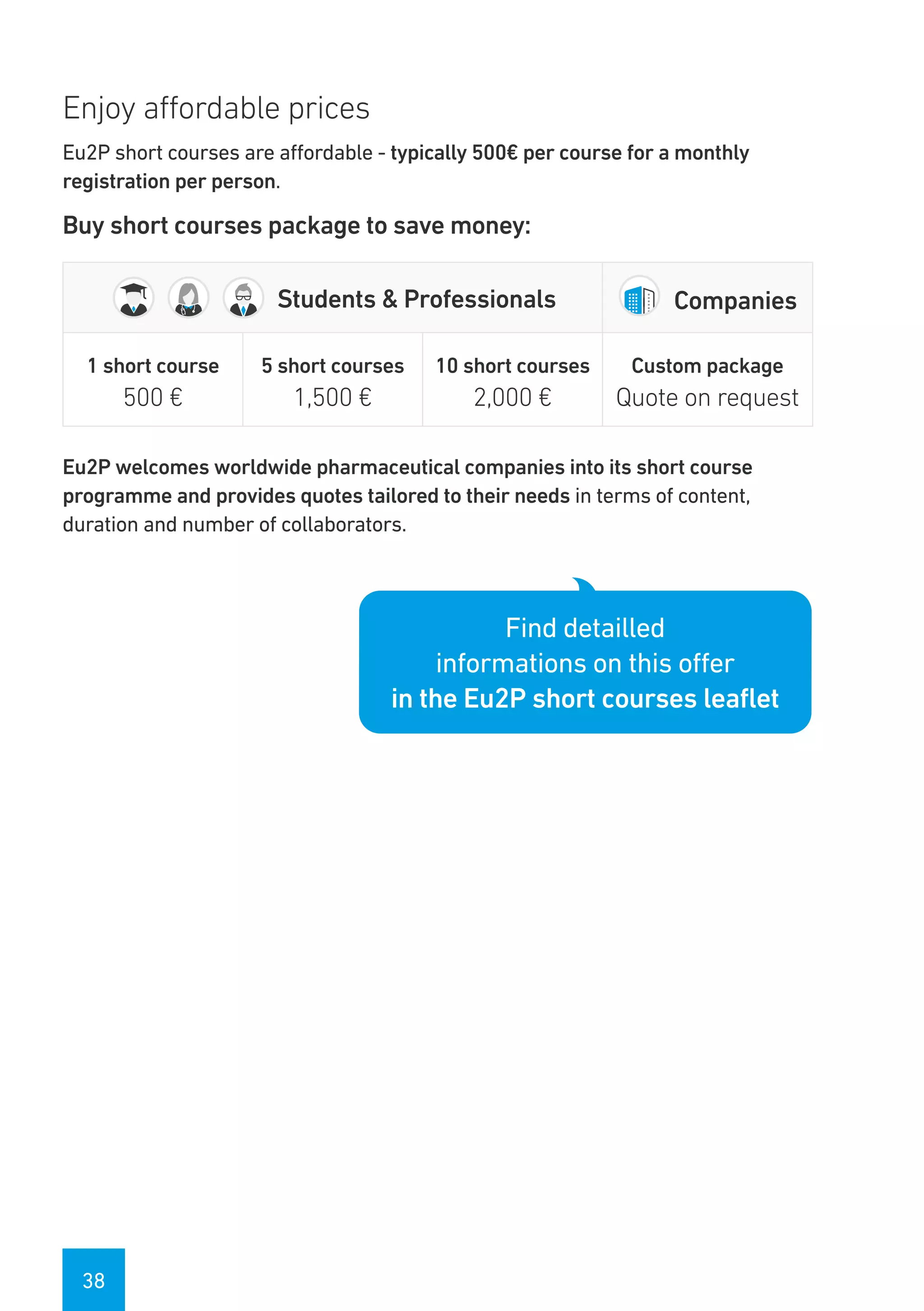
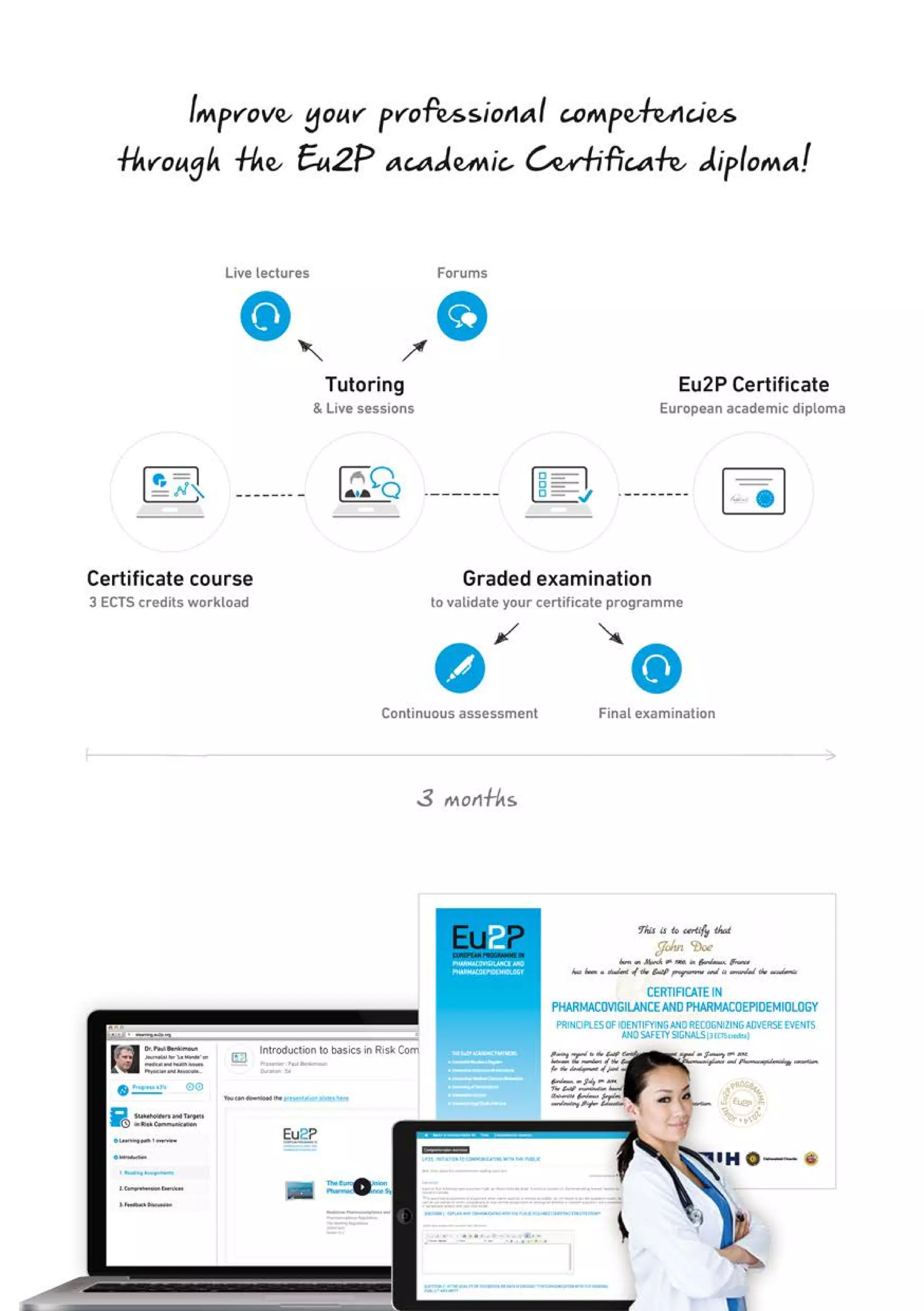
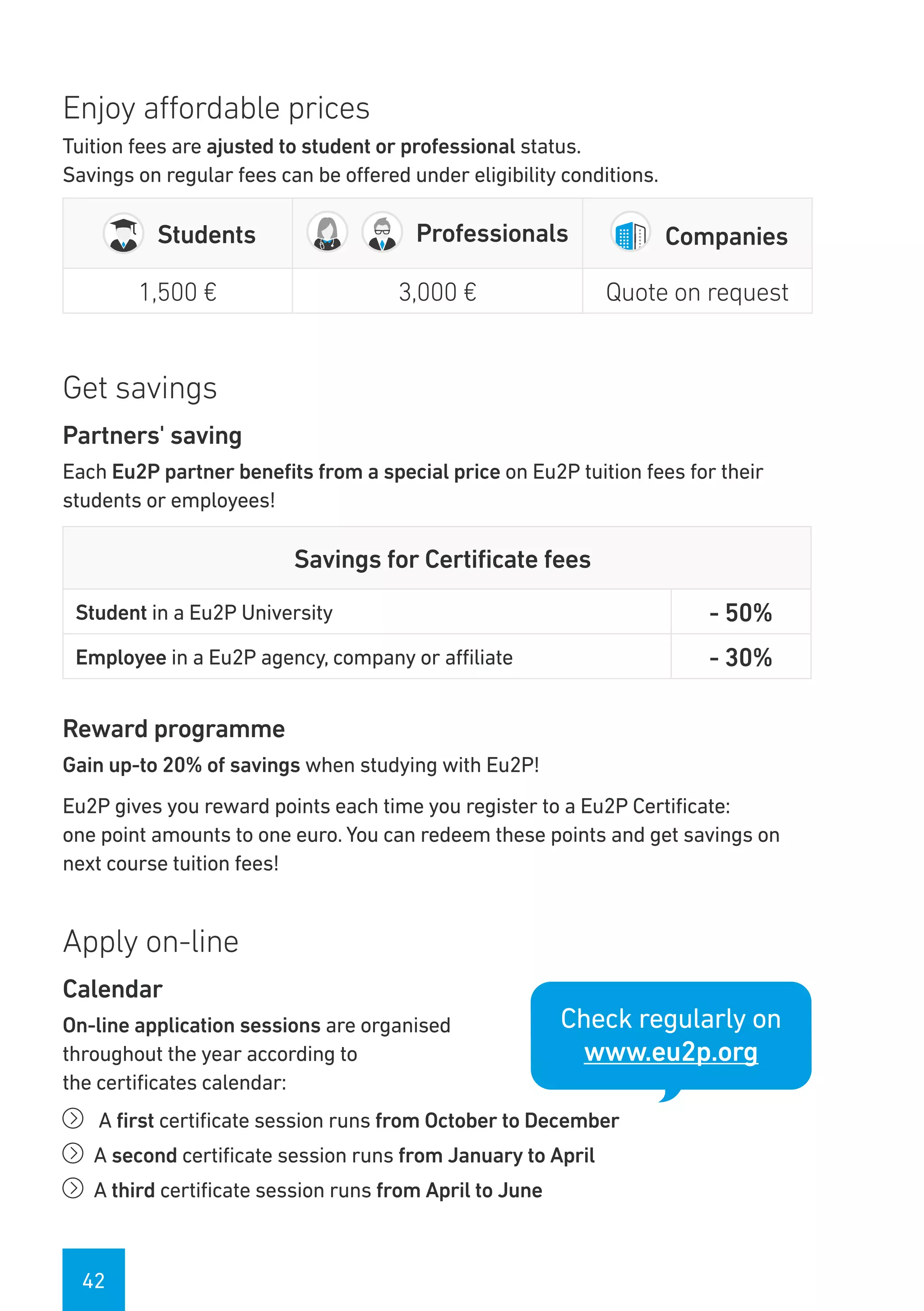
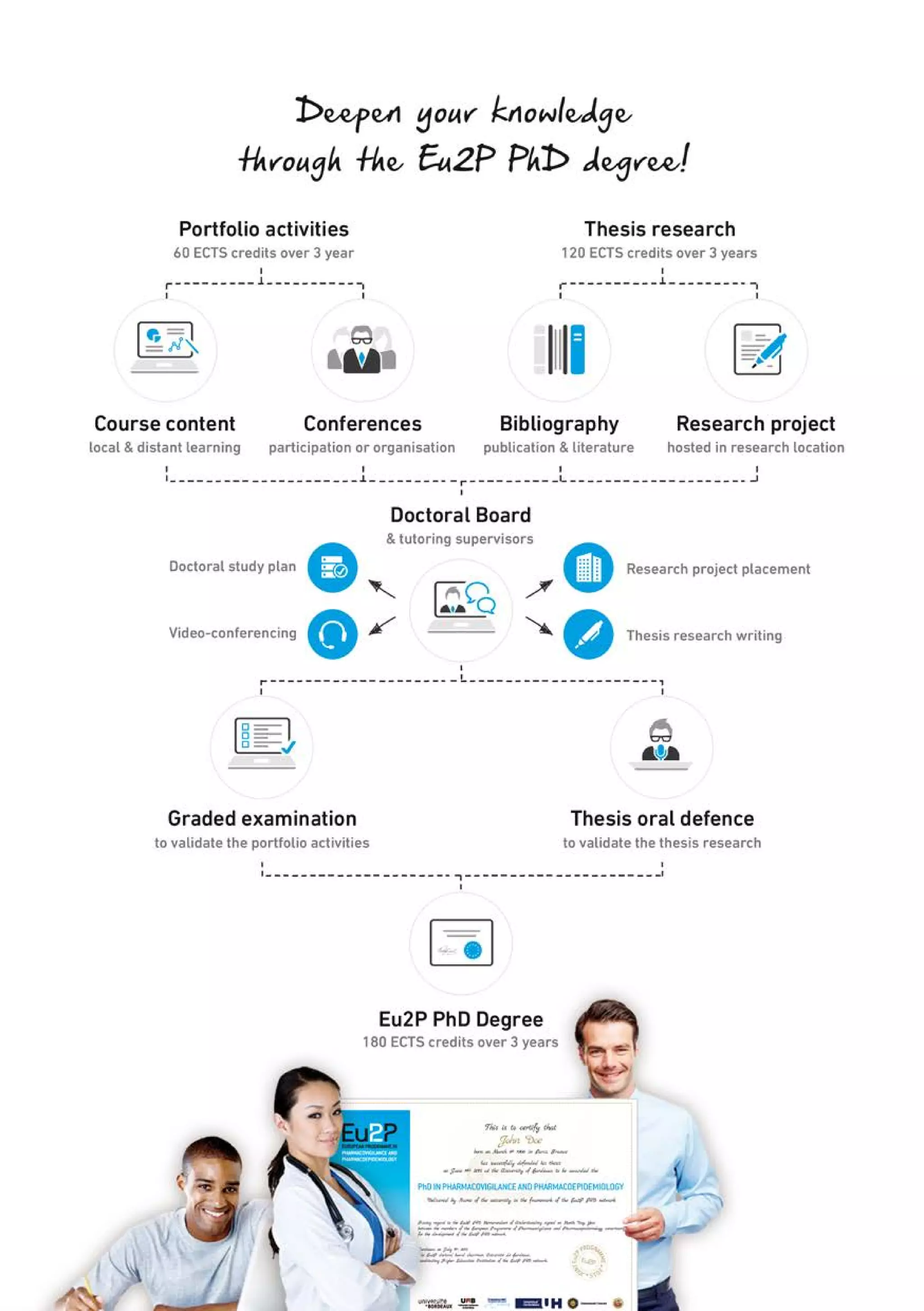
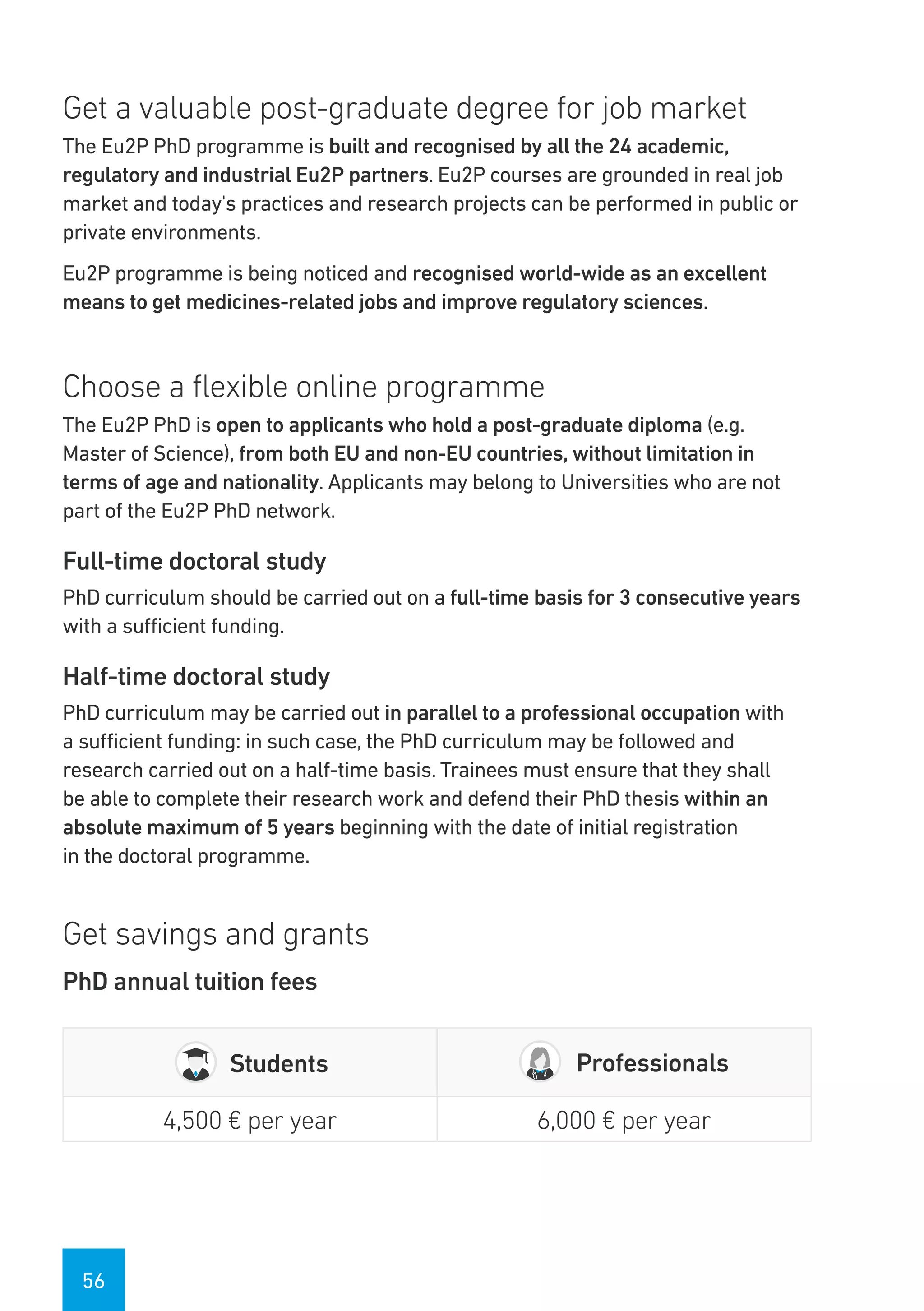
The Eu2P programme offers various training opportunities in pharmacovigilance and pharmacoepidemiology, including short courses, certificates, masters, and PhD programmes. The training is delivered through an online platform and is offered by a partnership between universities, pharmaceutical companies, and regulatory agencies. Courses cover topics such as basics of epidemiology and statistics, clinical research methods, pharmacovigilance regulations, risk identification and communication, and assessing the public health impact of medicines. The training is designed to improve professional competencies for healthcare professionals, students, and industry professionals.