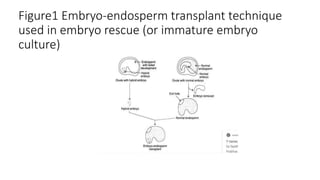
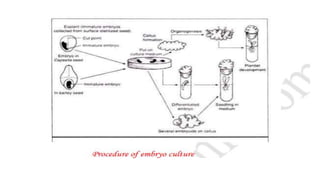
Embryo culture is a technique used to cultivate hybrid fertilization products, allowing for the rescue of immature embryos and the bypassing of seed dormancy. The method has two main types: mature embryo culture, which is easier and requires simple media, and immature embryo culture, which is more complex and requires careful dissection. This technique is significant for applications such as overcoming seed dormancy, shortening breeding cycles, and facilitating clonal propagation.