Embed presentation
Download to read offline


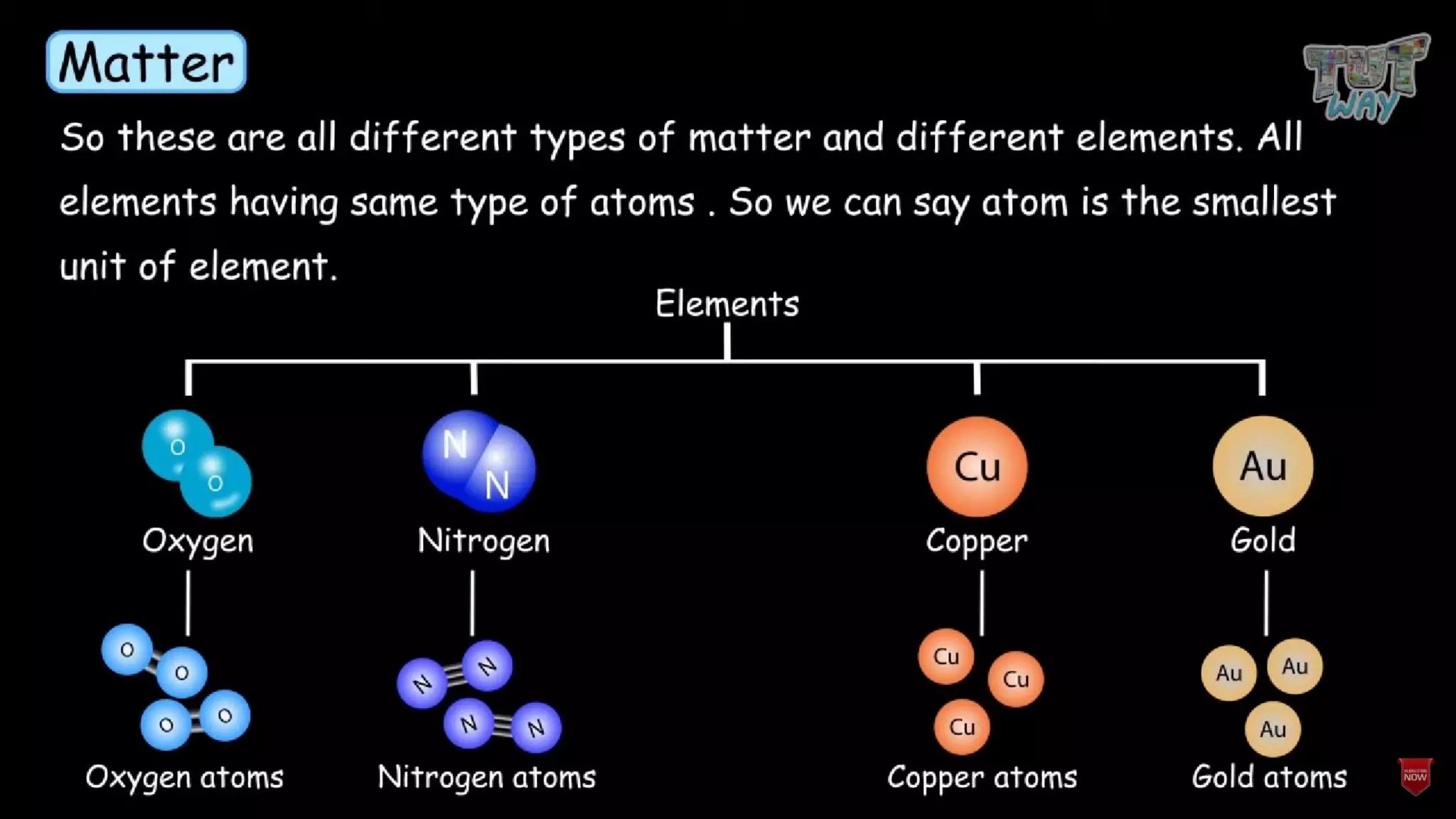




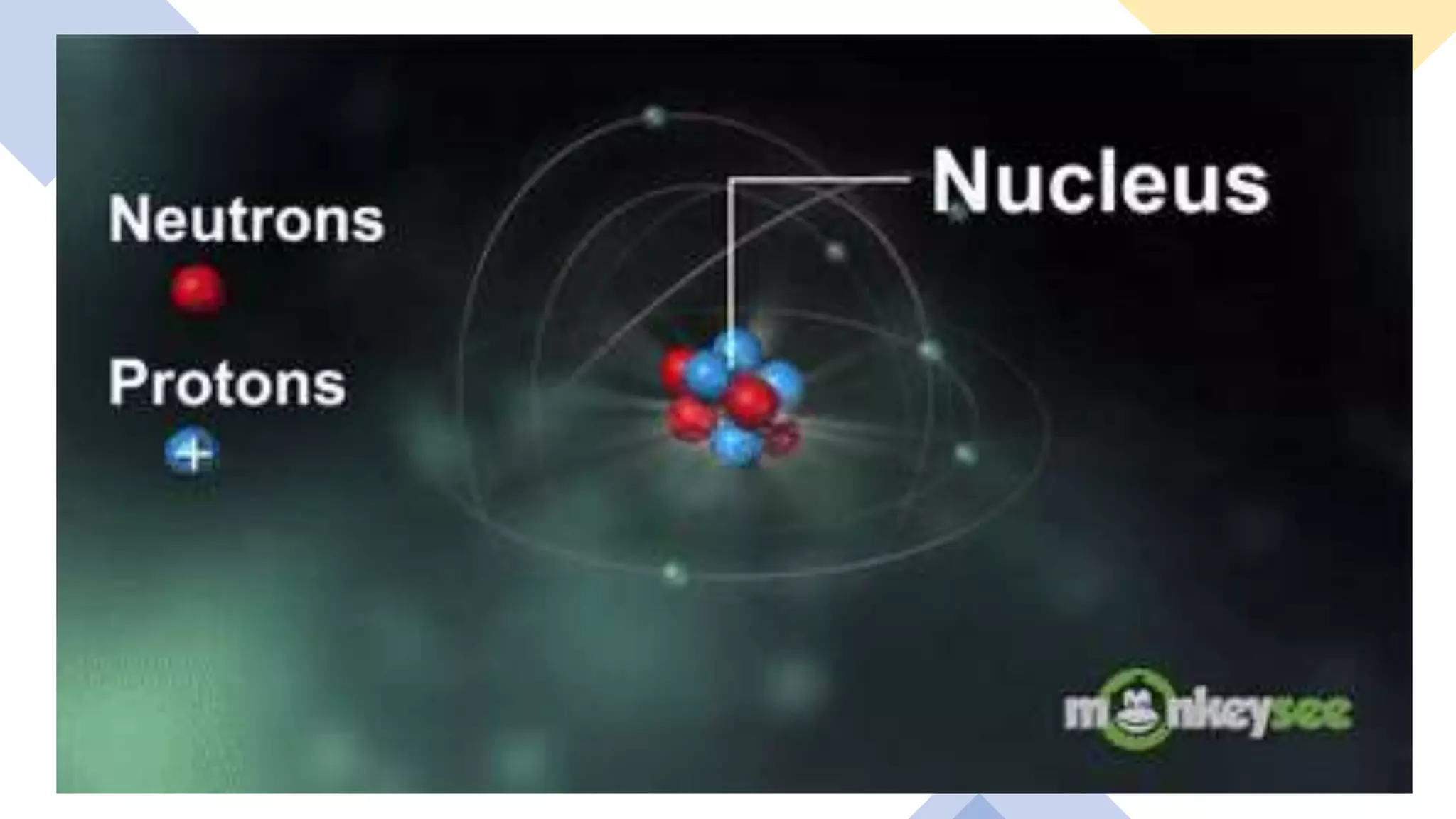
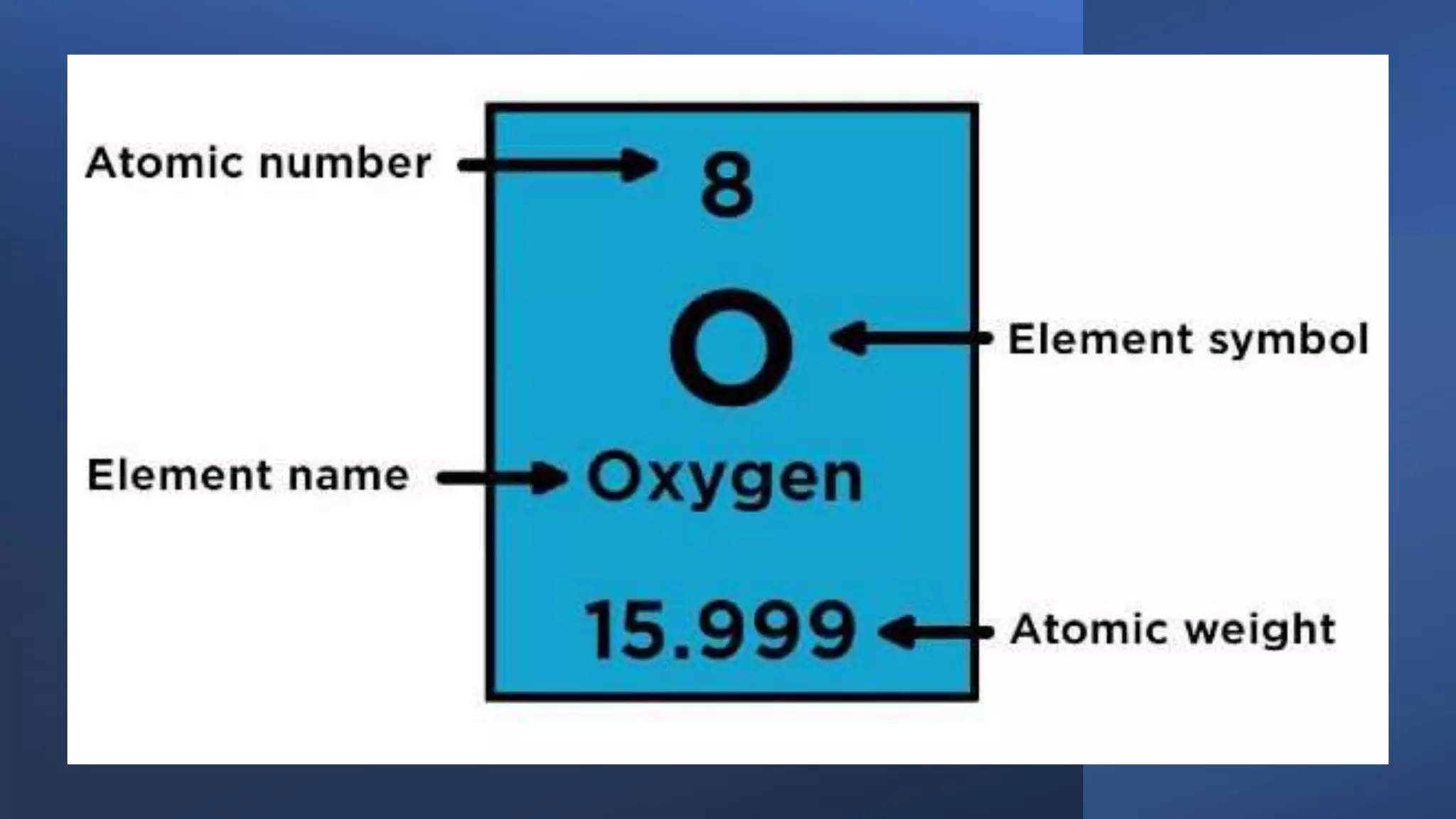
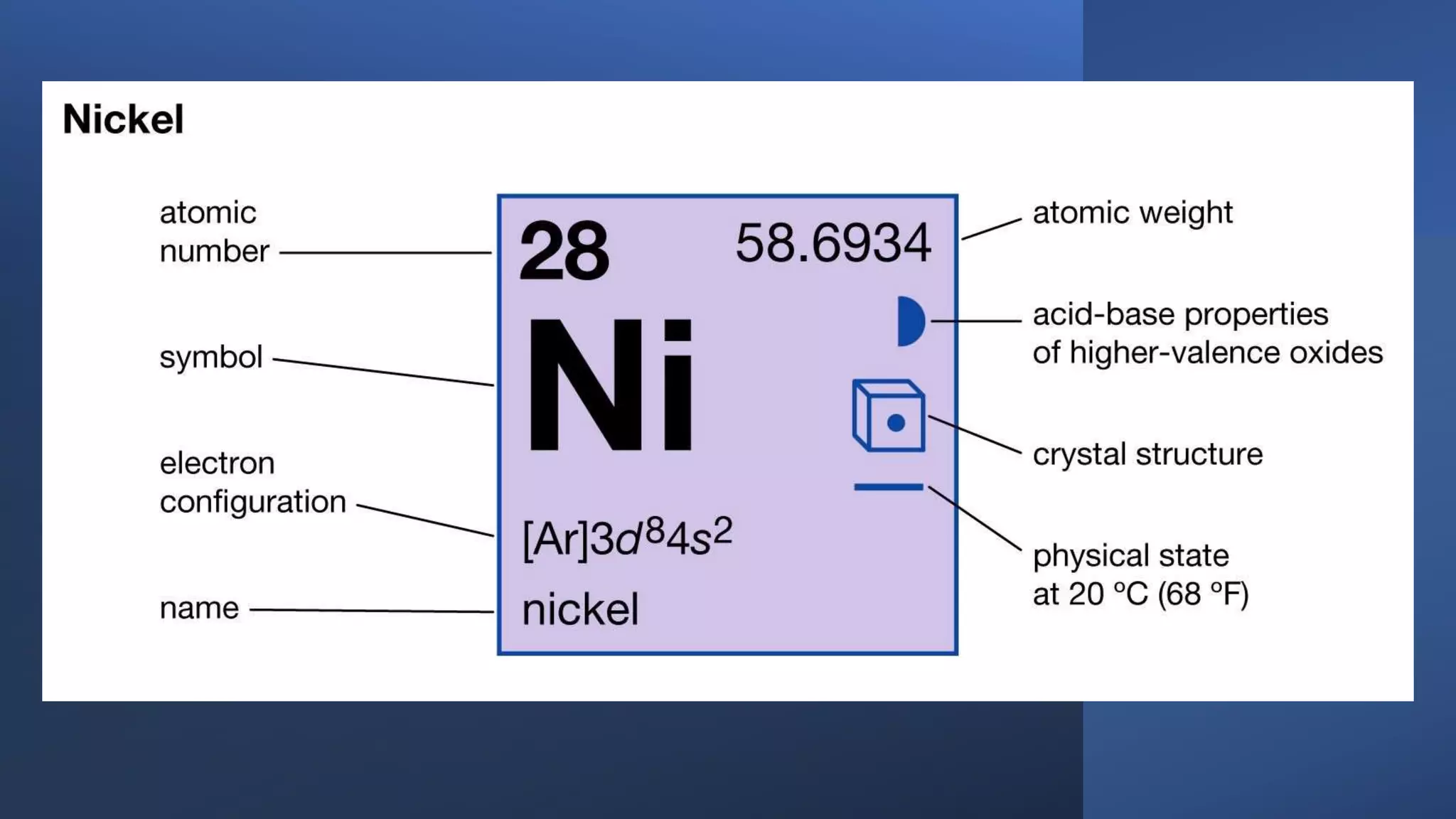


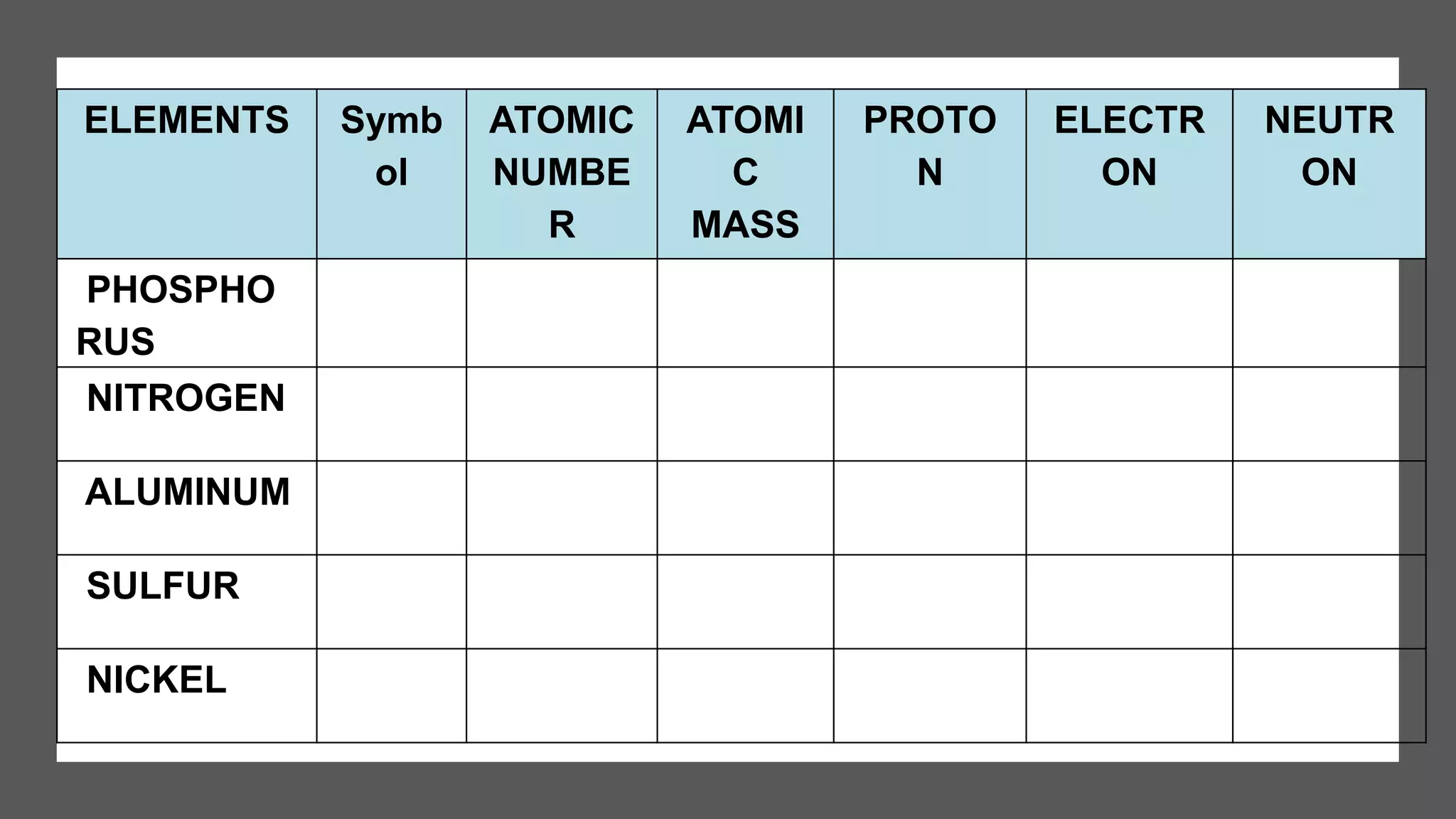
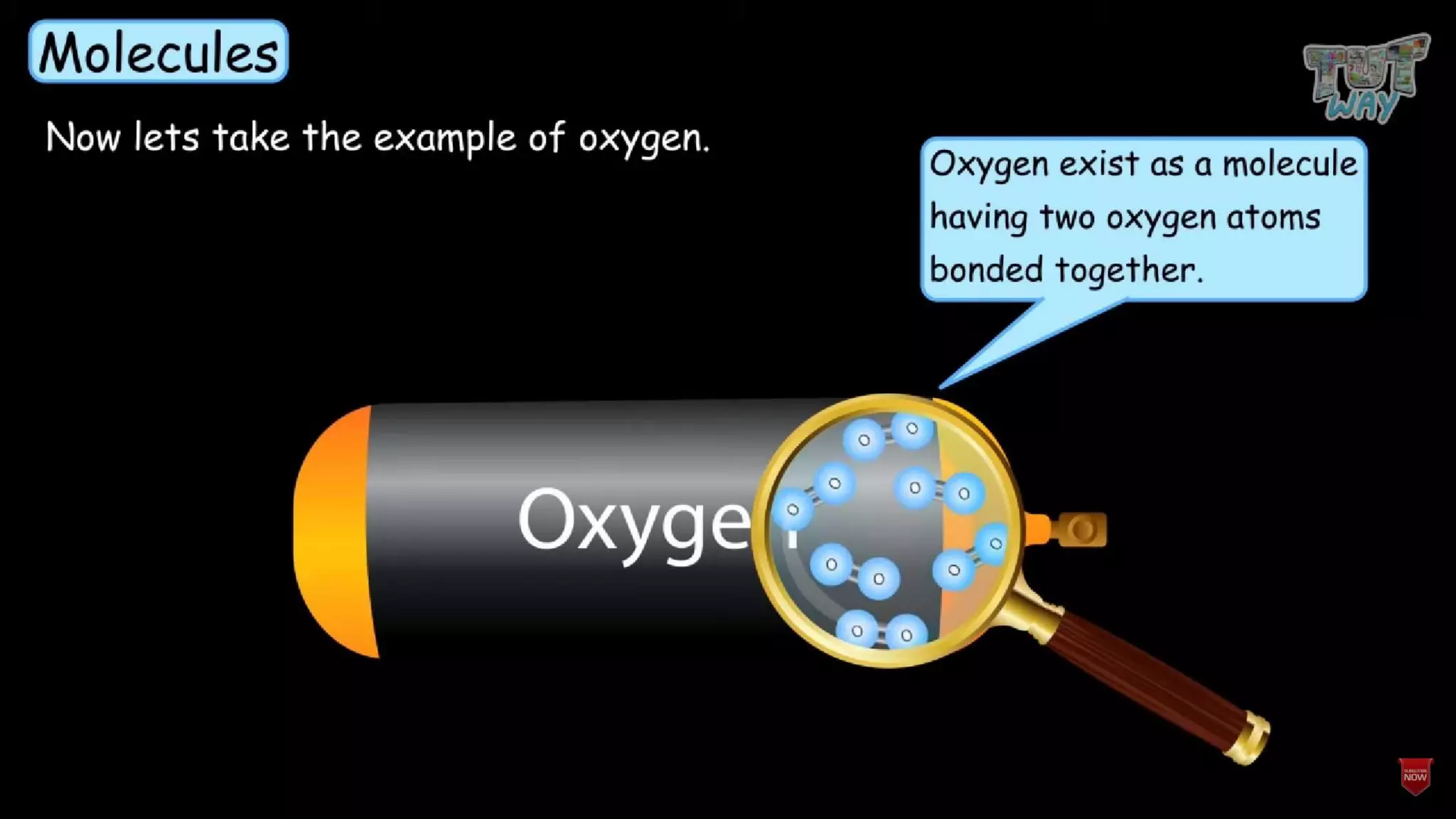

The document provides information about elements and their symbols, atomic structure, and subatomic particles. It lists the symbols for common elements in Column A and their names in Column B. It also explains that elements are the simplest form of matter made up of 118 types of substances that cannot be broken down further. The number of protons, electrons, and neutrons in an atom can be calculated using the atomic number and mass number.














