Electron shell
•Download as DOCX, PDF•
0 likes•83 views
The number of electrons in an atom's outermost shell determines its reactivity. Atoms are most stable when their outer shell is full. The periodic table arranges elements based on their electron configuration and properties. Elements in the same column generally have the same number of valence electrons and similar reactivity, with reactivity increasing as you move left to right within a period. Noble gases have full outer shells and are highly stable as single atoms, while group 1 elements have one valence electron and tend to lose or share it to become stable.
Report
Share
Report
Share
Recommended
Chapter 4

This document provides an overview of how elements are organized in the periodic table. It discusses early classification systems developed by Newlands and Mendeleev and how Moseley later determined that atomic number, not atomic mass, is the basis for organization. Key periodic properties like valence electrons and how they determine chemical properties are explained. Finally, it gives a brief tour of different groups of elements and trends seen in the periodic table.
The periodic table

The periodic table arranges elements from left to right based on increasing atomic number. Elements in the same column or group have similar properties, while properties gradually change across each period or row from left to right as atoms get smaller but more massive. The periodic table was created by Russian chemist Dmitri Mendeleev and represents each element with a chemical symbol and atomic number to classify its identity and properties.
Periodic table of elements

This presentation includes familiarization of periodic table of elements, familiarization of an element, and electron configuration of an element and of the noble gas.
Periodic table-shells-valence

Each row of the periodic table is called a period. Elements in the same period have the same number of electron shells. Each column is called a group. Elements in the same group have the same number of valence electrons, except for helium which has two electrons. Valence electrons are the outermost electrons of an atom and are involved in bonding. Reactive elements bond easily to gain or lose valence electrons to achieve a full outer electron shell of eight electrons.
Periodic Table of Elements

The document summarizes key information about the periodic table of elements, including its organization of elements according to atomic number and properties. Elements are grouped into families with similar properties, and the periodic table can be used to predict chemical reactions and properties of elements. Different areas of the periodic table are described, including alkali metals, transition metals, noble gases, and more.
Concept Map 

The document identifies several examples as either ionic compounds or covalent compounds. Potassium chloride and calcium oxide are identified as ionic compounds, while sulfur tetrafluoride and hydrochloric acid are identified as covalent compounds. It then provides information about the basic structure of atoms including protons, neutrons, electrons, energy levels, and isotopes and ions. Finally, it discusses the periodic table and classifications of elements as metals, metalloids, and nonmetals, and defines ionic and covalent compounds.
Shells/ Valence Electrons

The document discusses the periodic table and properties of elements. It explains that each row of the periodic table is called a period, with elements in the same period having the same number of electron shells. It also explains that each column is called a group, with elements in the same group having the same number of valence electrons in their outer shell. The document provides examples of determining the number of shells and valence electrons for various elements based on their period and group.
Periodic table (1)

The document provides information about the periodic table of elements. It explains that the periodic table organizes the 118 known elements according to properties like atomic number and mass. Elements are grouped into families that have similar chemical behaviors, and periods that show how properties change across rows. Key aspects like reactivity, metallic and non-metallic properties, and the discoveries of scientists like Mendeleev who developed the periodic table are summarized.
Recommended
Chapter 4

This document provides an overview of how elements are organized in the periodic table. It discusses early classification systems developed by Newlands and Mendeleev and how Moseley later determined that atomic number, not atomic mass, is the basis for organization. Key periodic properties like valence electrons and how they determine chemical properties are explained. Finally, it gives a brief tour of different groups of elements and trends seen in the periodic table.
The periodic table

The periodic table arranges elements from left to right based on increasing atomic number. Elements in the same column or group have similar properties, while properties gradually change across each period or row from left to right as atoms get smaller but more massive. The periodic table was created by Russian chemist Dmitri Mendeleev and represents each element with a chemical symbol and atomic number to classify its identity and properties.
Periodic table of elements

This presentation includes familiarization of periodic table of elements, familiarization of an element, and electron configuration of an element and of the noble gas.
Periodic table-shells-valence

Each row of the periodic table is called a period. Elements in the same period have the same number of electron shells. Each column is called a group. Elements in the same group have the same number of valence electrons, except for helium which has two electrons. Valence electrons are the outermost electrons of an atom and are involved in bonding. Reactive elements bond easily to gain or lose valence electrons to achieve a full outer electron shell of eight electrons.
Periodic Table of Elements

The document summarizes key information about the periodic table of elements, including its organization of elements according to atomic number and properties. Elements are grouped into families with similar properties, and the periodic table can be used to predict chemical reactions and properties of elements. Different areas of the periodic table are described, including alkali metals, transition metals, noble gases, and more.
Concept Map 

The document identifies several examples as either ionic compounds or covalent compounds. Potassium chloride and calcium oxide are identified as ionic compounds, while sulfur tetrafluoride and hydrochloric acid are identified as covalent compounds. It then provides information about the basic structure of atoms including protons, neutrons, electrons, energy levels, and isotopes and ions. Finally, it discusses the periodic table and classifications of elements as metals, metalloids, and nonmetals, and defines ionic and covalent compounds.
Shells/ Valence Electrons

The document discusses the periodic table and properties of elements. It explains that each row of the periodic table is called a period, with elements in the same period having the same number of electron shells. It also explains that each column is called a group, with elements in the same group having the same number of valence electrons in their outer shell. The document provides examples of determining the number of shells and valence electrons for various elements based on their period and group.
Periodic table (1)

The document provides information about the periodic table of elements. It explains that the periodic table organizes the 118 known elements according to properties like atomic number and mass. Elements are grouped into families that have similar chemical behaviors, and periods that show how properties change across rows. Key aspects like reactivity, metallic and non-metallic properties, and the discoveries of scientists like Mendeleev who developed the periodic table are summarized.
HOW ARE ELEMENTS ORGANIZED?

The document summarizes key concepts from Chapter 4 of the textbook, including:
1) Elements are organized in the periodic table based on their atomic number, with elements in the same group sharing similar chemical properties due to having the same number of valence electrons.
2) Early scientists like Newlands and Mendeleev noticed repeating patterns in element properties and developed the first periodic tables, with Mendeleev arranging elements by atomic mass and predicting missing elements.
3) Moseley later determined that atomic number, not atomic mass, is the fundamental basis for the periodic table's organization.
4) The periodic table is divided into blocks like s-block main group elements and d-block transition metals that have
The Periodic Table

organizing the elements, periodic table, Mendeleev, periodic law, metals, nonmetals, metalloinds, periodic trends
Periodic Table

The document discusses the history and development of the periodic table. It explains that Mendeleev was the first to publish a periodic table in 1869, organizing elements based on atomic mass and recognizing that elements with similar properties fell into vertical columns. However, Mendeleev's table had some anomalies, like incorrect placement of some elements. Moseley later proposed using atomic number instead of atomic mass, solving these anomalies. The modern periodic table is organized into periods and groups based on atomic number, with blocks for different orbital types. It provides information about elemental properties and reactions.
Periodic table

The document summarizes key concepts about the periodic table of elements, including how it organizes the 118 known elements according to their atomic structure and properties. Elements are classified into families based on their valence electrons and include metals, non-metals, metalloids and noble gases. The periodic table provides a way to predict an element's properties and reactivity.
G9 chemistry vocabulary

A nearly-comprehensive list of vocabulary terms needed for introductory chemistry in grade 9 science, including a variety of source websites for reference.
Periodic table

The document provides information about the periodic table of elements, including how it organizes the 118 known elements according to their atomic structure and properties. Elements are arranged based on their atomic number and can be grouped into families that show similar traits. The periodic table provides a way to predict chemical behaviors and allows scientists to identify unknown elements based on trends in atomic structure.
Periodic table

The document summarizes key concepts about the periodic table of elements, including:
1) Elements are organized into groups based on their valence electrons, with elements in the same group having similar properties.
2) Periods refer to horizontal rows, with the first element being very reactive and the last being an inactive gas.
3) Non-metals are generally poor conductors while metals are good conductors.
4) Metalloids have properties between metals and non-metals.
5) Reactive elements easily bond to complete their outer electron levels.
Ix chemistry full notes chapter 3

1. The document discusses key concepts relating to atoms, molecules, ions, and chemical formulas. It covers Dalton's atomic theory, isotopes, relative atomic mass, ion formation, and rules for writing chemical formulas.
2. The main topics covered include the basic structure of atoms and molecules, including that atoms combine in whole number ratios to form compounds. It also addresses limitations of Dalton's theory regarding isotopes and isobars.
3. The document defines important terms like molecules, ions, valency, and molar mass and explains the relationship between mass and moles of a substance.
Periodic Classification of Elements and Periodicity

The document discusses the history and development of the periodic table. It explains that early scientists like Dobereiner and Newlands began classifying elements based on their properties, but that Dmitri Mendeleev created the first recognizable periodic table in 1871. His table arranged elements in order of atomic mass and predicted properties of undiscovered elements. Later, Moseley's discovery of atomic number allowed the modern periodic table to arrange elements by atomic number instead of mass. The document also discusses trends in properties within the periodic table.
Ch 5.1,5.2 organizing elements & the periodic table

The document summarizes key aspects of the periodic table, including how Mendeleev organized the elements and used the periodic table to predict undiscovered elements. It describes the modern periodic table as arranging elements by atomic number in rows called periods and columns called groups, with elements in the same group having similar properties due to their electron configurations. The document also discusses atomic structure including atomic number and mass, and classifies elements as metals, nonmetals, and metalloids based on their physical and chemical properties.
01 chemical bonding-theory-final-e

Chemical bonding occurs when atoms combine to form molecules or ionic compounds to achieve stable electronic configurations. There are several types of bonds including ionic bonds, covalent bonds, and coordinate bonds. Ionic bonds form when electrons are transferred from electropositive atoms to electronegative atoms, resulting in oppositely charged ions that are attracted to each other. Covalent bonds form through the sharing of electron pairs between atoms. Coordinate bonds form through the interaction of a Lewis acid and base where one species provides a pair of electrons. Chemical bonds provide stability and determine many properties of substances.
Blb12 ch02 lecture

- The document discusses atomic theory and the development of models of the atom over time, from Dalton's atomic theory to Thomson's "plum pudding" model to Rutherford's nuclear model.
- It also covers the discovery of subatomic particles like electrons, protons, and neutrons as well as radioactive decay.
- Additional topics include atomic structure, isotopes, ions, chemical formulas and nomenclature of compounds and acids.
Chemical Reactions JQ

The periodic table organizes all known elements and contains information about atoms and molecules. It arranges metals, non-metals, and metalloids using elemental symbols. Elements are composed of atoms with a specific number of protons. Compounds consist of two or more elements in a fixed ratio that are bonded together. Chemical reactions transform one set of substances into another, either spontaneously or through external energy sources, while physical changes only alter the state of matter.
Interactive textbook ch. 13 chemical bonding

Ionic bonds form when valence electrons are transferred from metal atoms to nonmetal atoms, resulting in positively charged metal ions and negatively charged nonmetal ions that are attracted to each other. Metal atoms easily lose electrons to achieve stable full outer energy levels, while nonmetal atoms gain electrons for the same reason. The ions associate in repeating three-dimensional crystal lattices to form solid ionic compounds that are brittle with high melting points and often dissolve in water.
AP Chem Chapter 2 Outline

This document provides an overview of atomic theory and basic concepts in chemistry including:
1) Dalton's atomic theory which states that matter is made of extremely small indivisible particles called atoms.
2) Atoms of different elements have different properties and atoms of the same element are identical.
3) Chemical reactions involve changes in how atoms are combined but atoms themselves are neither created nor destroyed.
4) The discovery of subatomic particles like electrons, protons and neutrons led to Rutherford's nuclear model of the atom.
5) Isotopes are atoms of the same element with different numbers of neutrons. The periodic table organizes elements by atomic number.
Sca.2.4.5 CHEMISTRY and Periodic Table

This document provides information about groups, periods, and electrons on the periodic table. It discusses how elements in the same group have the same number of valence electrons and will therefore react similarly. It gives examples of sodium and potassium both having one valence electron. It also discusses how atoms gain or lose electrons to form ions, and how the periodic table is organized by increasing atomic number.
Elements

Elements are pure substances that cannot be broken down further through chemical or physical means. There are currently 118 known elements, with new ones occasionally being discovered through artificial nuclear reactions. Elements are identified by their atomic number, which represents the number of protons in the nucleus. Elements are the basic building blocks of all matter and are used to create thousands of materials, though some like plutonium are also used in nuclear weapons due to their radioactive properties.
Biochemistry--6.1 Basic Chemistry

The document discusses basic chemistry concepts including the composition of matter at the atomic level, elements, compounds, and bonding. It explains that atoms are made up of protons, neutrons, and electrons. Elements are pure substances made of one type of atom, with 92 naturally occurring elements and 26 found in living things. The four main elements that make up nearly all of an organism's weight are hydrogen, oxygen, nitrogen, and carbon. Atoms can bond through ionic bonds by gaining or losing electrons, or covalent bonds by sharing electrons, in order to achieve stable full energy levels.
Chemistry notes the periodic table powerpoint

This document provides an overview of the periodic table, including its history and organization. It was invented by Dmitri Mendeleev in 1869 to show recurring trends in the properties of the chemical elements. The periodic table arranges the elements horizontally by increasing atomic number and vertically into groups of elements with similar chemical properties. Key points covered include atomic symbols, families of elements including metals/nonmetals/metalloids and their properties, valence electrons, and periodic trends in properties across and down the table.
Atoms

This document defines key concepts related to atoms, molecules, and ions:
1) Atoms are the fundamental units that all matter is made of, consisting of protons, neutrons, and electrons.
2) Molecules are formed when two or more atoms bond together and can be monoatomic, diatomic, or polyatomic.
3) Ions are formed when atoms gain or lose electrons to have a positive or negative charge, and can be monatomic or polyatomic.
THE PERIODIC TABLE OF ELEMENTS 2021 (1).pptx

The document discusses the periodic table of elements and valency. It explains that the periodic table classifies elements according to properties like valence number. Elements are arranged in groups and periods. Each group has a characteristic valence charge. Valence refers to an element's combining power and is determined by its outer electron configuration and ability to gain or lose electrons to achieve an octet. Examples of calculating valence for sodium and chlorine are provided based on their electronic configurations. Valence is important for writing chemical formulas and determining combinations of elements.
Chem chapt 6

The document summarizes the development and key features of the periodic table. It traces how early scientists like Newlands and Mendeleev organized the elements and developed periodic trends. The modern periodic table is arranged by atomic number and contains metals, nonmetals, and metalloids organized into blocks and groups. Elements in the same group have similar properties due to their electron configurations, and properties vary periodically according to trends like atomic radius and ionization energy.
More Related Content
What's hot
HOW ARE ELEMENTS ORGANIZED?

The document summarizes key concepts from Chapter 4 of the textbook, including:
1) Elements are organized in the periodic table based on their atomic number, with elements in the same group sharing similar chemical properties due to having the same number of valence electrons.
2) Early scientists like Newlands and Mendeleev noticed repeating patterns in element properties and developed the first periodic tables, with Mendeleev arranging elements by atomic mass and predicting missing elements.
3) Moseley later determined that atomic number, not atomic mass, is the fundamental basis for the periodic table's organization.
4) The periodic table is divided into blocks like s-block main group elements and d-block transition metals that have
The Periodic Table

organizing the elements, periodic table, Mendeleev, periodic law, metals, nonmetals, metalloinds, periodic trends
Periodic Table

The document discusses the history and development of the periodic table. It explains that Mendeleev was the first to publish a periodic table in 1869, organizing elements based on atomic mass and recognizing that elements with similar properties fell into vertical columns. However, Mendeleev's table had some anomalies, like incorrect placement of some elements. Moseley later proposed using atomic number instead of atomic mass, solving these anomalies. The modern periodic table is organized into periods and groups based on atomic number, with blocks for different orbital types. It provides information about elemental properties and reactions.
Periodic table

The document summarizes key concepts about the periodic table of elements, including how it organizes the 118 known elements according to their atomic structure and properties. Elements are classified into families based on their valence electrons and include metals, non-metals, metalloids and noble gases. The periodic table provides a way to predict an element's properties and reactivity.
G9 chemistry vocabulary

A nearly-comprehensive list of vocabulary terms needed for introductory chemistry in grade 9 science, including a variety of source websites for reference.
Periodic table

The document provides information about the periodic table of elements, including how it organizes the 118 known elements according to their atomic structure and properties. Elements are arranged based on their atomic number and can be grouped into families that show similar traits. The periodic table provides a way to predict chemical behaviors and allows scientists to identify unknown elements based on trends in atomic structure.
Periodic table

The document summarizes key concepts about the periodic table of elements, including:
1) Elements are organized into groups based on their valence electrons, with elements in the same group having similar properties.
2) Periods refer to horizontal rows, with the first element being very reactive and the last being an inactive gas.
3) Non-metals are generally poor conductors while metals are good conductors.
4) Metalloids have properties between metals and non-metals.
5) Reactive elements easily bond to complete their outer electron levels.
Ix chemistry full notes chapter 3

1. The document discusses key concepts relating to atoms, molecules, ions, and chemical formulas. It covers Dalton's atomic theory, isotopes, relative atomic mass, ion formation, and rules for writing chemical formulas.
2. The main topics covered include the basic structure of atoms and molecules, including that atoms combine in whole number ratios to form compounds. It also addresses limitations of Dalton's theory regarding isotopes and isobars.
3. The document defines important terms like molecules, ions, valency, and molar mass and explains the relationship between mass and moles of a substance.
Periodic Classification of Elements and Periodicity

The document discusses the history and development of the periodic table. It explains that early scientists like Dobereiner and Newlands began classifying elements based on their properties, but that Dmitri Mendeleev created the first recognizable periodic table in 1871. His table arranged elements in order of atomic mass and predicted properties of undiscovered elements. Later, Moseley's discovery of atomic number allowed the modern periodic table to arrange elements by atomic number instead of mass. The document also discusses trends in properties within the periodic table.
Ch 5.1,5.2 organizing elements & the periodic table

The document summarizes key aspects of the periodic table, including how Mendeleev organized the elements and used the periodic table to predict undiscovered elements. It describes the modern periodic table as arranging elements by atomic number in rows called periods and columns called groups, with elements in the same group having similar properties due to their electron configurations. The document also discusses atomic structure including atomic number and mass, and classifies elements as metals, nonmetals, and metalloids based on their physical and chemical properties.
01 chemical bonding-theory-final-e

Chemical bonding occurs when atoms combine to form molecules or ionic compounds to achieve stable electronic configurations. There are several types of bonds including ionic bonds, covalent bonds, and coordinate bonds. Ionic bonds form when electrons are transferred from electropositive atoms to electronegative atoms, resulting in oppositely charged ions that are attracted to each other. Covalent bonds form through the sharing of electron pairs between atoms. Coordinate bonds form through the interaction of a Lewis acid and base where one species provides a pair of electrons. Chemical bonds provide stability and determine many properties of substances.
Blb12 ch02 lecture

- The document discusses atomic theory and the development of models of the atom over time, from Dalton's atomic theory to Thomson's "plum pudding" model to Rutherford's nuclear model.
- It also covers the discovery of subatomic particles like electrons, protons, and neutrons as well as radioactive decay.
- Additional topics include atomic structure, isotopes, ions, chemical formulas and nomenclature of compounds and acids.
Chemical Reactions JQ

The periodic table organizes all known elements and contains information about atoms and molecules. It arranges metals, non-metals, and metalloids using elemental symbols. Elements are composed of atoms with a specific number of protons. Compounds consist of two or more elements in a fixed ratio that are bonded together. Chemical reactions transform one set of substances into another, either spontaneously or through external energy sources, while physical changes only alter the state of matter.
Interactive textbook ch. 13 chemical bonding

Ionic bonds form when valence electrons are transferred from metal atoms to nonmetal atoms, resulting in positively charged metal ions and negatively charged nonmetal ions that are attracted to each other. Metal atoms easily lose electrons to achieve stable full outer energy levels, while nonmetal atoms gain electrons for the same reason. The ions associate in repeating three-dimensional crystal lattices to form solid ionic compounds that are brittle with high melting points and often dissolve in water.
AP Chem Chapter 2 Outline

This document provides an overview of atomic theory and basic concepts in chemistry including:
1) Dalton's atomic theory which states that matter is made of extremely small indivisible particles called atoms.
2) Atoms of different elements have different properties and atoms of the same element are identical.
3) Chemical reactions involve changes in how atoms are combined but atoms themselves are neither created nor destroyed.
4) The discovery of subatomic particles like electrons, protons and neutrons led to Rutherford's nuclear model of the atom.
5) Isotopes are atoms of the same element with different numbers of neutrons. The periodic table organizes elements by atomic number.
Sca.2.4.5 CHEMISTRY and Periodic Table

This document provides information about groups, periods, and electrons on the periodic table. It discusses how elements in the same group have the same number of valence electrons and will therefore react similarly. It gives examples of sodium and potassium both having one valence electron. It also discusses how atoms gain or lose electrons to form ions, and how the periodic table is organized by increasing atomic number.
Elements

Elements are pure substances that cannot be broken down further through chemical or physical means. There are currently 118 known elements, with new ones occasionally being discovered through artificial nuclear reactions. Elements are identified by their atomic number, which represents the number of protons in the nucleus. Elements are the basic building blocks of all matter and are used to create thousands of materials, though some like plutonium are also used in nuclear weapons due to their radioactive properties.
Biochemistry--6.1 Basic Chemistry

The document discusses basic chemistry concepts including the composition of matter at the atomic level, elements, compounds, and bonding. It explains that atoms are made up of protons, neutrons, and electrons. Elements are pure substances made of one type of atom, with 92 naturally occurring elements and 26 found in living things. The four main elements that make up nearly all of an organism's weight are hydrogen, oxygen, nitrogen, and carbon. Atoms can bond through ionic bonds by gaining or losing electrons, or covalent bonds by sharing electrons, in order to achieve stable full energy levels.
Chemistry notes the periodic table powerpoint

This document provides an overview of the periodic table, including its history and organization. It was invented by Dmitri Mendeleev in 1869 to show recurring trends in the properties of the chemical elements. The periodic table arranges the elements horizontally by increasing atomic number and vertically into groups of elements with similar chemical properties. Key points covered include atomic symbols, families of elements including metals/nonmetals/metalloids and their properties, valence electrons, and periodic trends in properties across and down the table.
Atoms

This document defines key concepts related to atoms, molecules, and ions:
1) Atoms are the fundamental units that all matter is made of, consisting of protons, neutrons, and electrons.
2) Molecules are formed when two or more atoms bond together and can be monoatomic, diatomic, or polyatomic.
3) Ions are formed when atoms gain or lose electrons to have a positive or negative charge, and can be monatomic or polyatomic.
What's hot (20)
Periodic Classification of Elements and Periodicity

Periodic Classification of Elements and Periodicity
Ch 5.1,5.2 organizing elements & the periodic table

Ch 5.1,5.2 organizing elements & the periodic table
Similar to Electron shell
THE PERIODIC TABLE OF ELEMENTS 2021 (1).pptx

The document discusses the periodic table of elements and valency. It explains that the periodic table classifies elements according to properties like valence number. Elements are arranged in groups and periods. Each group has a characteristic valence charge. Valence refers to an element's combining power and is determined by its outer electron configuration and ability to gain or lose electrons to achieve an octet. Examples of calculating valence for sodium and chlorine are provided based on their electronic configurations. Valence is important for writing chemical formulas and determining combinations of elements.
Chem chapt 6

The document summarizes the development and key features of the periodic table. It traces how early scientists like Newlands and Mendeleev organized the elements and developed periodic trends. The modern periodic table is arranged by atomic number and contains metals, nonmetals, and metalloids organized into blocks and groups. Elements in the same group have similar properties due to their electron configurations, and properties vary periodically according to trends like atomic radius and ionization energy.
CHEMISTRY-TOPIC-3-Trends.docx trends in the periodic table

The document discusses trends in the periodic table. It explains that periods are rows in the periodic table where elements have the same number of atomic orbitals. Groups are columns where elements have the same number of valence electrons. It describes how atomic size decreases left to right in a period but increases top to bottom in a group. Electronegativity increases left to right across a period but decreases top to bottom in a group. Ionization energy increases left to right in a period due to valence shell stability but decreases top to bottom in a group due to electron shielding. Electron affinity increases left to right in a period due to decreasing atomic radius but decreases top to bottom in a group due to increasing atomic radius.
Karanjot

The document summarizes the history and development of the periodic table. It describes early classifications by Döbereiner, Newlands and Mendeleev, and refinements made by Moseley. Mendeleev arranged elements in order of atomic weight and predicted undiscovered elements. Moseley reordered elements by atomic number using X-ray wavelengths, correcting some placements and predicting gaps for radioactive elements. The periodic table organizes elements and allows prediction of properties based on position.
Periodic table Development and Trends

The periodic table is arranged based on atomic number and properties repeat periodically. Early forms grouped elements based on atomic mass but Moseley found atomic number was key. The table is divided into periods and groups with trends in properties like atomic size decreasing left to right across periods and ionization energy increasing. Metallic character increases down groups and nonmetallic character increases right across periods. The periodic table organizes the elements and allows prediction of properties.
Periodic trends

The document discusses periodic trends in elemental properties, including atomic radius, ionization energy, electron affinity, electronegativity, and reactivity. It explains that Dmitri Mendeleev was the first to organize elements into a periodic table based on their properties and predicted undiscovered elements. The trends are due to changes in atomic structure and the effective nuclear charge as protons and electrons are added. Atomic radius generally decreases left to right and increases top to bottom. Ionization energy and electronegativity increase as you move up and to the left on the periodic table.
Structure of atom (igcse)

1. Atoms consist of a positively charged nucleus surrounded by electrons that orbit in defined shells or energy levels.
2. The number of protons in the nucleus defines the atomic number of an element, while the total number of protons and neutrons gives the mass number.
3. Chemical properties are determined by valence electrons in the outer shell. Elements tend to gain or lose electrons to achieve a stable outer shell of 8 electrons.
Inorganic

INORGANIC CHEMISTRY
Is the chemistry of all the elements and their compounds with the exception of most carbon compounds out of which only the oxides, cyanides and carbonates are considered as inorganic compound. Inorganic compound can also be defined as the study of the elements in the periodic table.
PERIODIC TABLE
Periodic table is the table of all the known elements arranged in order of increasing atomic numbers. The arrangement reflects the electronics configuration of the elements.
THE PERIODIC TABLE CLASSIFICATION OF THE ELEMENTS (MENDELEEV'S AND LOTHAR MEYER 1869)
Acids and Bases 2014 GBL 

This document outlines the key concepts to be covered in a Year 11 100 Science course on aspects of acids and bases, including atomic structure, properties of acids and bases, rates of reaction and particle theory, uses of acids and bases, and restrictions on the acids and bases included in the course. Students will study electron configuration, ionic bonding, naming ionic compounds, properties of acids and bases such as releasing hydrogen ions in water and reacting to form salts, and the rates of reactions and particle theory explanations. Assessment will include selected aspects of acids and bases such as atomic structure, properties, uses, and rates of reaction.
Structure Of Atoms Part 3

The document discusses atomic structure and ions. It defines isotopes as atoms of the same element with different numbers of neutrons. The outermost electrons of an atom are involved in chemical reactions. The periodic table arranges elements by their number of outermost electrons. Noble gases have full outermost shells with 2 or 8 electrons. Atoms form ions by gaining or losing electrons to achieve stable configurations. Positively charged ions are formed when electrons are lost, and negatively charged ions are formed when electrons are gained.
The Periodic Law Notes.ppt

The document summarizes key concepts about the periodic table, including its history and development by Mendeleev and Moseley. It discusses trends in various periodic properties, including atomic radius, ionization energy, electron affinity, and electronegativity. These properties follow predictable trends as one moves across or up/down the periodic table, which is explained by electron configurations and the organization of elements. The periodic table provides an organized framework for understanding similarities and differences among elements.
Periodic table.pdfx

The document provides biographical information about Dmitri Mendeleyev, the discoverer of the periodic table. It notes that he was a Russian chemist born in 1834 who served as a professor of chemistry. In 1869, he announced the principle of periodicity of properties in chemical elements and created the first periodic table. His table arranged elements in order of atomic weight and grouped them by similar properties, allowing him to predict properties of undiscovered elements. The periodic table organized the known chemical elements and reflected recurring trends in their properties based on atomic structure.
General principles of organic chemistry

This document discusses general principles of atomic structure and organic chemistry. It covers topics such as the atomic structure of atoms, atomic orbitals and quantum numbers, electron configuration, and the octet rule. The key points are that atoms are composed of protons, neutrons and electrons; electrons occupy distinct regions called orbitals that can be described by quantum numbers; and elements seek to attain stable electron configurations, often an octet in the outer shell like noble gases.
Periodic Table Chapter 14

The document summarizes key periodic trends in the properties of elements as they relate to their position in the periodic table, including:
1) Atomic radius generally increases down a group and decreases across a period as more protons are added to the same principal energy level.
2) Ionization energy decreases down a group as atoms gain principal energy levels but increases across a period as more protons are added.
3) Positive ion size decreases and negative ion size increases relative to their parent atoms. Ion sizes also increase down a group.
4) Electronegativity decreases down a group but increases across a period toward the nonmetals.
4-1 Ions & Ionic Bonding

1) Atoms form ions by gaining or losing electrons in order to achieve a stable electron configuration like that of a noble gas.
2) Metals form positive ions (cations) by losing electrons, while nonmetals form negative ions (anions) by gaining electrons.
3) Ionic bonds form when oppositely charged ions attract each other, such as metal cations and nonmetal anions. The transfer of electrons allows the atoms to achieve stable electron configurations.
Chemchapt6 101015132910-phpapp02

The document discusses the development and key features of the periodic table. It describes how early scientists like Newlands, Meyer, and Mendeleev organized the elements based on properties like atomic mass and predicted new elements. Moseley later showed atomic number was fundamental. The modern periodic table is organized into rows (periods) and columns (groups) based on electron configuration. Elements in the same group have similar properties due to their valence electrons. The document outlines trends in properties like atomic radius, ionization energy, and electronegativity across periods and down groups.
4-1: Ions & Ionic Bonding

1) Atoms form ions when they lose or gain electrons to achieve a stable electron configuration like the noble gases.
2) Metals form positive ions (cations) by losing electrons, while nonmetals form negative ions (anions) by gaining electrons.
3) Ionic bonds form when oppositely charged ions are attracted to each other, such as sodium (Na+) and chlorine (Cl-), resulting in an ionic compound.
The electrons which are involved in bond formatio.pdf

The electrons which are involved in bond formation between atoms are found in the
outermost shell (sometimes in the next to the outer-most shell) of the neutral atom; these are
called VALENCE ELECTRONS. The atoms of elements which have only one or two electrons
in their outermost shells (active shells) may lose electrons when they combine with atoms of
other elements. An atom which has lost one or more valence electrons possesses a positive
charge, and is called a POSITIVE ION. The sodium atom loses its one valence electron and
acquires a +1 charge when it enters into chemical combination with an atom of an element such
as chlorine. The magnesium atom may lose its two valence electrons and assume a +2 charge.
Na Na+ + e- The Na symbol to the left of the arrow represents a stable sodium atom while the
Na+ symbol to the right of the arrow represents an unstable sodium ion which has had a single
electron removed. Mg Mg++ + 2e- The Mg symbol to the left of the arrow represents a stable
magnesium atom while the Mg++ symbol to the right of the arrow represents an unstable
magnesium ion which has had two electrons removed. The smaller the number of valence
electrons in the atom, the greater the tendency of the element to lose electrons and thus form
positive ions during chemical combination with atoms of other elements. The energy required to
remove an electron from a neutral atom to form a positive ion is called the IONIZATION
POTENTIAL of the atom. Some metals have small ionization potentials and readily form
positive ions. The nonmetals, which have more electrons in their outer shells than the metals,
have large ionization potentials and show little tendency toward the formation of positive ions.
Atoms which lack one or two electrons of having an outermost shell of eight electrons readily
gain sufficient electrons from certain other atoms, such as sodium and magnesium, to make a full
compliment of eight electrons in the outside shell. Neutral atoms become NEGATIVE IONS by
gaining electrons. The nonmetals, such as Fluorine (F), Chlorine (Cl), Bromine (Br), Iodine (I),
Oxygen (O), Nitrogen (N) and Sulfur (S), readily form negative ions. Cl + e- Cl- Chlorine,
when in its stable form, possesses seven valence electrons and therefore has the ability to gain
one electron (as represented to the left of the arrow) giving it a negative charge of one when in
its unstable ionic form (as represented to the right of the arrow above). S + 2e- S-2 Sulfur, when
in its stable form, possesses six valence electrons and therefore has the ability to gain two
electrons giving it a negative charge of two when in its unstable ionic form. The attraction of a
neutral atom for electrons is known as its ELECTRON AFFINITY. The nonmetals have high
electron affinities and the metals have very low electron affinities. Thus, mainly the nonmetals
tend to form negative ions during chemical combination. When a positive ion and a negative ion
are brought close together, strong electr.
Chemunit5presentation 111106095110-phpapp02

The document discusses the development and organization of the periodic table. It describes how scientists like Newlands, Meyer, and Mendeleev contributed to organizing the elements. Key features of the modern periodic table are that elements are arranged by atomic number in periods and groups. Elements have characteristic properties based on their position and electron configuration.
3b. The periodic table and trends.pptx 11

1. This document provides an overview of trends in the periodic table, including atomic size, ionization energy, electronegativity, and metallic/non-metallic character. It explains that these properties trend both across periods, from left to right as atomic number increases, and down groups as additional shells are filled.
2. Specifically, it notes that atomic size decreases across periods as nuclear charge increases, while increasing down groups as additional shells are added. Ionization energy and electronegativity both increase from left to right as nuclear charge increases, while decreasing down groups as electrons are farther from the nucleus. Metallic character increases down groups and decreases across periods.
3. The document aims to explain these
Similar to Electron shell (20)
CHEMISTRY-TOPIC-3-Trends.docx trends in the periodic table

CHEMISTRY-TOPIC-3-Trends.docx trends in the periodic table
The electrons which are involved in bond formatio.pdf

The electrons which are involved in bond formatio.pdf
Recently uploaded
Lifting the Corporate Veil. Power Point Presentation

"Lifting the Corporate Veil" is a legal concept that refers to the judicial act of disregarding the separate legal personality of a corporation or limited liability company (LLC). Normally, a corporation is considered a legal entity separate from its shareholders or members, meaning that the personal assets of shareholders or members are protected from the liabilities of the corporation. However, there are certain situations where courts may decide to "pierce" or "lift" the corporate veil, holding shareholders or members personally liable for the debts or actions of the corporation.
Here are some common scenarios in which courts might lift the corporate veil:
Fraud or Illegality: If shareholders or members use the corporate structure to perpetrate fraud, evade legal obligations, or engage in illegal activities, courts may disregard the corporate entity and hold those individuals personally liable.
Undercapitalization: If a corporation is formed with insufficient capital to conduct its intended business and meet its foreseeable liabilities, and this lack of capitalization results in harm to creditors or other parties, courts may lift the corporate veil to hold shareholders or members liable.
Failure to Observe Corporate Formalities: Corporations and LLCs are required to observe certain formalities, such as holding regular meetings, maintaining separate financial records, and avoiding commingling of personal and corporate assets. If these formalities are not observed and the corporate structure is used as a mere façade, courts may disregard the corporate entity.
Alter Ego: If there is such a unity of interest and ownership between the corporation and its shareholders or members that the separate personalities of the corporation and the individuals no longer exist, courts may treat the corporation as the alter ego of its owners and hold them personally liable.
Group Enterprises: In some cases, where multiple corporations are closely related or form part of a single economic unit, courts may pierce the corporate veil to achieve equity, particularly if one corporation's actions harm creditors or other stakeholders and the corporate structure is being used to shield culpable parties from liability.
快速办理(SCU毕业证书)澳洲南十字星大学毕业证文凭证书一模一样

学校原件一模一样【微信:741003700 】《(SCU毕业证书)澳洲南十字星大学毕业证文凭证书》【微信:741003700 】学位证,留信认证(真实可查,永久存档)原件一模一样纸张工艺/offer、雅思、外壳等材料/诚信可靠,可直接看成品样本,帮您解决无法毕业带来的各种难题!外壳,原版制作,诚信可靠,可直接看成品样本。行业标杆!精益求精,诚心合作,真诚制作!多年品质 ,按需精细制作,24小时接单,全套进口原装设备。十五年致力于帮助留学生解决难题,包您满意。
本公司拥有海外各大学样板无数,能完美还原。
1:1完美还原海外各大学毕业材料上的工艺:水印,阴影底纹,钢印LOGO烫金烫银,LOGO烫金烫银复合重叠。文字图案浮雕、激光镭射、紫外荧光、温感、复印防伪等防伪工艺。材料咨询办理、认证咨询办理请加学历顾问Q/微741003700
【主营项目】
一.毕业证【q微741003700】成绩单、使馆认证、教育部认证、雅思托福成绩单、学生卡等!
二.真实使馆公证(即留学回国人员证明,不成功不收费)
三.真实教育部学历学位认证(教育部存档!教育部留服网站永久可查)
四.办理各国各大学文凭(一对一专业服务,可全程监控跟踪进度)
如果您处于以下几种情况:
◇在校期间,因各种原因未能顺利毕业……拿不到官方毕业证【q/微741003700】
◇面对父母的压力,希望尽快拿到;
◇不清楚认证流程以及材料该如何准备;
◇回国时间很长,忘记办理;
◇回国马上就要找工作,办给用人单位看;
◇企事业单位必须要求办理的
◇需要报考公务员、购买免税车、落转户口
◇申请留学生创业基金
留信网认证的作用:
1:该专业认证可证明留学生真实身份
2:同时对留学生所学专业登记给予评定
3:国家专业人才认证中心颁发入库证书
4:这个认证书并且可以归档倒地方
5:凡事获得留信网入网的信息将会逐步更新到个人身份内,将在公安局网内查询个人身份证信息后,同步读取人才网入库信息
6:个人职称评审加20分
7:个人信誉贷款加10分
8:在国家人才网主办的国家网络招聘大会中纳入资料,供国家高端企业选择人才
Patenting_Innovations_in_3D_Printing_Prosthetics.pptx

slide deck : Patenting innovations in 3D printing
Presentation (1).pptx Human rights of LGBTQ people in India, constitutional a...

Human rights of LGBTQ people in India, constitutional and judicial approach
一比一原版(Lincoln毕业证)新西兰林肯大学毕业证如何办理

Lincoln硕士毕业证成绩单【微信95270640】办理新西兰林肯大学毕业证原版一模一样、Lincoln毕业证制作【Q微信95270640】《新西兰林肯大学毕业证购买流程》《Lincoln成绩单制作》新西兰林肯大学毕业证书Lincoln毕业证文凭新西兰林肯大学
本科毕业证书,学历学位认证如何办理【留学国外学位学历认证、毕业证、成绩单、大学Offer、雅思托福代考、语言证书、学生卡、高仿教育部认证等一切高仿或者真实可查认证服务】代办国外(海外)英国、加拿大、美国、新西兰、澳大利亚、新西兰等国外各大学毕业证、文凭学历证书、成绩单、学历学位认证真实可查。
[留学文凭学历认证(留信认证使馆认证)新西兰林肯大学毕业证成绩单毕业证证书大学Offer请假条成绩单语言证书国际回国人员证明高仿教育部认证申请学校等一切高仿或者真实可查认证服务。
多年留学服务公司,拥有海外样板无数能完美1:1还原海外各国大学degreeDiplomaTranscripts等毕业材料。海外大学毕业材料都有哪些工艺呢?工艺难度主要由:烫金.钢印.底纹.水印.防伪光标.热敏防伪等等组成。而且我们每天都在更新海外文凭的样板以求所有同学都能享受到完美的品质服务。
国外毕业证学位证成绩单办理方法:
1客户提供办理新西兰林肯大学新西兰林肯大学毕业证学位证信息:姓名生日专业学位毕业时间等(如信息不确定可以咨询顾问:我们有专业老师帮你查询);
2开始安排制作毕业证成绩单电子图;
3毕业证成绩单电子版做好以后发送给您确认;
4毕业证成绩单电子版您确认信息无误之后安排制作成品;
5成品做好拍照或者视频给您确认;
6快递给客户(国内顺丰国外DHLUPS等快读邮寄)
— — — — 我们是挂科和未毕业同学们的福音我们是实体公司精益求精的工艺! — — — -
一真实留信认证的作用(私企外企荣誉的见证):
1:该专业认证可证明留学生真实留学身份同时对留学生所学专业等级给予评定。
2:国家专业人才认证中心颁发入库证书这个入网证书并且可以归档到地方。
3:凡是获得留信网入网的信息将会逐步更新到个人身份内将在公安部网内查询个人身份证信息后同步读取人才网入库信息。
4:个人职称评审加20分个人信誉贷款加10分。
5:在国家人才网主办的全国网络招聘大会中纳入资料供国家500强等高端企业选择人才。问山娃想买什么想吃什么山娃知道父亲赚钱很辛苦除了书籍和文具山娃啥也不要能牵着父亲的手满城闲逛他已心满意足了父亲连挑了三套童装叫山娃试穿山娃有点不想父亲说城里不比乡下要穿得漂漂亮亮爸怎么不穿得漂亮望着父亲山娃反问道父亲听了并不回答只是吃吃地笑山娃很精神越逛越起劲父亲却越逛越疲倦望着父亲呵欠连天的样子山娃也说困了累了回家吧小屋闷罐一般头顶上的三叶扇彻夜呜呜作响搅得满屋热气腾腾也搅得山娃心烦意乱父亲一的
在线办理(UNE毕业证书)新英格兰大学毕业证成绩单一模一样

学校原件一模一样【微信:741003700 】《(UNE毕业证书)新英格兰大学毕业证成绩单》【微信:741003700 】学位证,留信认证(真实可查,永久存档)原件一模一样纸张工艺/offer、雅思、外壳等材料/诚信可靠,可直接看成品样本,帮您解决无法毕业带来的各种难题!外壳,原版制作,诚信可靠,可直接看成品样本。行业标杆!精益求精,诚心合作,真诚制作!多年品质 ,按需精细制作,24小时接单,全套进口原装设备。十五年致力于帮助留学生解决难题,包您满意。
本公司拥有海外各大学样板无数,能完美还原。
1:1完美还原海外各大学毕业材料上的工艺:水印,阴影底纹,钢印LOGO烫金烫银,LOGO烫金烫银复合重叠。文字图案浮雕、激光镭射、紫外荧光、温感、复印防伪等防伪工艺。材料咨询办理、认证咨询办理请加学历顾问Q/微741003700
【主营项目】
一.毕业证【q微741003700】成绩单、使馆认证、教育部认证、雅思托福成绩单、学生卡等!
二.真实使馆公证(即留学回国人员证明,不成功不收费)
三.真实教育部学历学位认证(教育部存档!教育部留服网站永久可查)
四.办理各国各大学文凭(一对一专业服务,可全程监控跟踪进度)
如果您处于以下几种情况:
◇在校期间,因各种原因未能顺利毕业……拿不到官方毕业证【q/微741003700】
◇面对父母的压力,希望尽快拿到;
◇不清楚认证流程以及材料该如何准备;
◇回国时间很长,忘记办理;
◇回国马上就要找工作,办给用人单位看;
◇企事业单位必须要求办理的
◇需要报考公务员、购买免税车、落转户口
◇申请留学生创业基金
留信网认证的作用:
1:该专业认证可证明留学生真实身份
2:同时对留学生所学专业登记给予评定
3:国家专业人才认证中心颁发入库证书
4:这个认证书并且可以归档倒地方
5:凡事获得留信网入网的信息将会逐步更新到个人身份内,将在公安局网内查询个人身份证信息后,同步读取人才网入库信息
6:个人职称评审加20分
7:个人信誉贷款加10分
8:在国家人才网主办的国家网络招聘大会中纳入资料,供国家高端企业选择人才
Corporate Governance : Scope and Legal Framework

CORPORATE GOVERNANCE
MEANING
Corporate Governance refers to the way in which companies are governed and to what purpose. It identifies who has power and accountability, and who makes decisions. It is, in essence, a toolkit that enables management and the board to deal more effectively with the challenges of running a company.
San Remo Manual on International Law Applicable to Armed Conflict at Sea

Presentation by Justin Ordoyo
University of the Philippines College of Law
原版制作(PSU毕业证书)宾州州立大学公园分校毕业证学历证书一模一样

学校原件一模一样【微信:741003700 】《(PSU毕业证书)宾州州立大学公园分校毕业证学历证书》【微信:741003700 】学位证,留信认证(真实可查,永久存档)原件一模一样纸张工艺/offer、雅思、外壳等材料/诚信可靠,可直接看成品样本,帮您解决无法毕业带来的各种难题!外壳,原版制作,诚信可靠,可直接看成品样本。行业标杆!精益求精,诚心合作,真诚制作!多年品质 ,按需精细制作,24小时接单,全套进口原装设备。十五年致力于帮助留学生解决难题,包您满意。
本公司拥有海外各大学样板无数,能完美还原。
1:1完美还原海外各大学毕业材料上的工艺:水印,阴影底纹,钢印LOGO烫金烫银,LOGO烫金烫银复合重叠。文字图案浮雕、激光镭射、紫外荧光、温感、复印防伪等防伪工艺。材料咨询办理、认证咨询办理请加学历顾问Q/微741003700
【主营项目】
一.毕业证【q微741003700】成绩单、使馆认证、教育部认证、雅思托福成绩单、学生卡等!
二.真实使馆公证(即留学回国人员证明,不成功不收费)
三.真实教育部学历学位认证(教育部存档!教育部留服网站永久可查)
四.办理各国各大学文凭(一对一专业服务,可全程监控跟踪进度)
如果您处于以下几种情况:
◇在校期间,因各种原因未能顺利毕业……拿不到官方毕业证【q/微741003700】
◇面对父母的压力,希望尽快拿到;
◇不清楚认证流程以及材料该如何准备;
◇回国时间很长,忘记办理;
◇回国马上就要找工作,办给用人单位看;
◇企事业单位必须要求办理的
◇需要报考公务员、购买免税车、落转户口
◇申请留学生创业基金
留信网认证的作用:
1:该专业认证可证明留学生真实身份
2:同时对留学生所学专业登记给予评定
3:国家专业人才认证中心颁发入库证书
4:这个认证书并且可以归档倒地方
5:凡事获得留信网入网的信息将会逐步更新到个人身份内,将在公安局网内查询个人身份证信息后,同步读取人才网入库信息
6:个人职称评审加20分
7:个人信誉贷款加10分
8:在国家人才网主办的国家网络招聘大会中纳入资料,供国家高端企业选择人才
The Future of Criminal Defense Lawyer in India.pdf

https://veteranlegal.in/defense-lawyer-in-india/ | Criminal defense Lawyer in India has always been a vital aspect of the country's legal system. As defenders of justice, criminal Defense Lawyer play a critical role in ensuring that individuals accused of crimes receive a fair trial and that their constitutional rights are protected. As India evolves socially, economically, and technologically, the role and future of criminal Defense Lawyer are also undergoing significant changes. This comprehensive blog explores the current landscape, challenges, technological advancements, and prospects for criminal Defense Lawyer in India.
It's the Law: Recent Court and Administrative Decisions of Interest

2024 Idaho Water Users Association, Sun Valley, Idaho - June 11, 2024. Presented by Payton Hampton and Garrett Kitamura
V.-SENTHIL-BALAJI-SLP-C-8939-8940-2023-SC-Judgment-07-August-2023.pdf

सुप्रीम कोर्ट ने यह भी माना था कि मजिस्ट्रेट का यह कर्तव्य है कि वह सुनिश्चित करे कि अधिकारी पीएमएलए के तहत निर्धारित प्रक्रिया के साथ-साथ संवैधानिक सुरक्षा उपायों का भी उचित रूप से पालन करें।
Business Laws Sunita saha

Business law for the students of undergraduate level. The presentation contains the summary of all the chapters under the syllabus of State University, Contract Act, Sale of Goods Act, Negotiable Instrument Act, Partnership Act, Limited Liability Act, Consumer Protection Act.
Sangyun Lee, 'Why Korea's Merger Control Occasionally Fails: A Public Choice ...

Presentation slides for a session held on June 4, 2024, at Kyoto University. This presentation is based on the presenter’s recent paper, coauthored with Hwang Lee, Professor, Korea University, with the same title, published in the Journal of Business Administration & Law, Volume 34, No. 2 (April 2024). The paper, written in Korean, is available at <https://shorturl.at/GCWcI>.
在线办理(SU毕业证书)美国雪城大学毕业证成绩单一模一样

学校原件一模一样【微信:741003700 】《(SU毕业证书)美国雪城大学毕业证成绩单》【微信:741003700 】学位证,留信认证(真实可查,永久存档)原件一模一样纸张工艺/offer、雅思、外壳等材料/诚信可靠,可直接看成品样本,帮您解决无法毕业带来的各种难题!外壳,原版制作,诚信可靠,可直接看成品样本。行业标杆!精益求精,诚心合作,真诚制作!多年品质 ,按需精细制作,24小时接单,全套进口原装设备。十五年致力于帮助留学生解决难题,包您满意。
本公司拥有海外各大学样板无数,能完美还原。
1:1完美还原海外各大学毕业材料上的工艺:水印,阴影底纹,钢印LOGO烫金烫银,LOGO烫金烫银复合重叠。文字图案浮雕、激光镭射、紫外荧光、温感、复印防伪等防伪工艺。材料咨询办理、认证咨询办理请加学历顾问Q/微741003700
【主营项目】
一.毕业证【q微741003700】成绩单、使馆认证、教育部认证、雅思托福成绩单、学生卡等!
二.真实使馆公证(即留学回国人员证明,不成功不收费)
三.真实教育部学历学位认证(教育部存档!教育部留服网站永久可查)
四.办理各国各大学文凭(一对一专业服务,可全程监控跟踪进度)
如果您处于以下几种情况:
◇在校期间,因各种原因未能顺利毕业……拿不到官方毕业证【q/微741003700】
◇面对父母的压力,希望尽快拿到;
◇不清楚认证流程以及材料该如何准备;
◇回国时间很长,忘记办理;
◇回国马上就要找工作,办给用人单位看;
◇企事业单位必须要求办理的
◇需要报考公务员、购买免税车、落转户口
◇申请留学生创业基金
留信网认证的作用:
1:该专业认证可证明留学生真实身份
2:同时对留学生所学专业登记给予评定
3:国家专业人才认证中心颁发入库证书
4:这个认证书并且可以归档倒地方
5:凡事获得留信网入网的信息将会逐步更新到个人身份内,将在公安局网内查询个人身份证信息后,同步读取人才网入库信息
6:个人职称评审加20分
7:个人信誉贷款加10分
8:在国家人才网主办的国家网络招聘大会中纳入资料,供国家高端企业选择人才
Guide on the use of Artificial Intelligence-based tools by lawyers and law fi...

This guide aims to provide information on how lawyers will be able to use the opportunities provided by AI tools and how such tools could help the business processes of small firms. Its objective is to provide lawyers with some background to understand what they can and cannot realistically expect from these products. This guide aims to give a reference point for small law practices in the EU
against which they can evaluate those classes of AI applications that are probably the most relevant for them.
What are the common challenges faced by women lawyers working in the legal pr...

The legal profession, which has historically been male-dominated, has experienced a significant increase in the number of women entering the field over the past few decades. Despite this progress, women lawyers continue to encounter various challenges as they strive for top positions.
From Promise to Practice. Implementing AI in Legal Environments

Pim Betist's presentation about generative AI in the legal sector at Lexpo.com '24
Search Warrants for NH Law Enforcement Officers

Training aid for law enforcement officers related to search warrants, the requirements needed, drafting, and execution of the search warrant.
Recently uploaded (20)
Lifting the Corporate Veil. Power Point Presentation

Lifting the Corporate Veil. Power Point Presentation
Patenting_Innovations_in_3D_Printing_Prosthetics.pptx

Patenting_Innovations_in_3D_Printing_Prosthetics.pptx
Presentation (1).pptx Human rights of LGBTQ people in India, constitutional a...

Presentation (1).pptx Human rights of LGBTQ people in India, constitutional a...
San Remo Manual on International Law Applicable to Armed Conflict at Sea

San Remo Manual on International Law Applicable to Armed Conflict at Sea
The Future of Criminal Defense Lawyer in India.pdf

The Future of Criminal Defense Lawyer in India.pdf
fnaf lore.pptx ...................................

fnaf lore.pptx ...................................
It's the Law: Recent Court and Administrative Decisions of Interest

It's the Law: Recent Court and Administrative Decisions of Interest
V.-SENTHIL-BALAJI-SLP-C-8939-8940-2023-SC-Judgment-07-August-2023.pdf

V.-SENTHIL-BALAJI-SLP-C-8939-8940-2023-SC-Judgment-07-August-2023.pdf
Sangyun Lee, 'Why Korea's Merger Control Occasionally Fails: A Public Choice ...

Sangyun Lee, 'Why Korea's Merger Control Occasionally Fails: A Public Choice ...
Guide on the use of Artificial Intelligence-based tools by lawyers and law fi...

Guide on the use of Artificial Intelligence-based tools by lawyers and law fi...
What are the common challenges faced by women lawyers working in the legal pr...

What are the common challenges faced by women lawyers working in the legal pr...
From Promise to Practice. Implementing AI in Legal Environments

From Promise to Practice. Implementing AI in Legal Environments
Electron shell
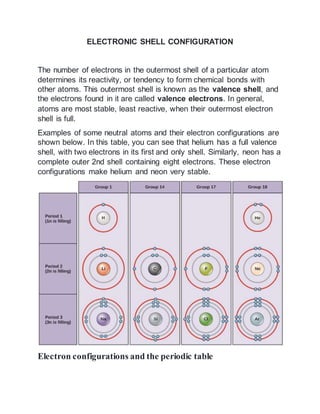
- 1. ELECTRONIC SHELL CONFIGURATION The number of electrons in the outermost shell of a particular atom determines its reactivity, or tendency to form chemical bonds with other atoms. This outermost shell is known as the valence shell, and the electrons found in it are called valence electrons. In general, atoms are most stable, least reactive, when their outermost electron shell is full. Examples of some neutral atoms and their electron configurations are shown below. In this table, you can see that helium has a full valence shell, with two electrons in its first and only shell. Similarly, neon has a complete outer 2nd shell containing eight electrons. These electron configurations make helium and neon very stable. Electron configurations and the periodic table
- 2. Elements are placed in order on the periodic table based on their atomic number, how many protons they have. In a neutral atom, the number of electrons will equal the number of protons, so we can easily determine electron number from atomic number. In addition, the position of an element in the periodic table—its column, or group, and row, or period—provides useful information about how those electrons are arranged. An element’s column number gives information about its number of valence electrons and reactivity. In general, the number of valence electrons is the same within a column and increases from left to right within a row. Group 1 elements have just one valence electron and group 8 elements have eight, except for helium, which has only two electrons total. Thus, group number is a good predictor of how reactive each element will be: Helium (He), neon (Ne), and argon (Ar) as group 8 elements, have outer electron shells that are full. This makes them highly stable as single atoms. Because of their non-reactivity, they are called the inert gases or noble gases. Hydrogen (H), lithium (Li) and sodium (Na) as group 1 elements, have just one electron in their outermost shells. They are unstable as single atoms, but can become stable by losing or sharing their one valence electron Fluorine (F) and chlorine (Cl), as group 7 elements, have seven electrons in their outermost shells. Carbon (C ) as a group 4 element, has four electrons in its outer shell. Thus, the columns of the periodic table reflect the number of electrons found in each element’s valence shell, which in turn determines how the element will react.