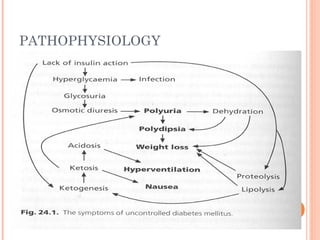
1. Diabetes mellitus is a chronic metabolic disease characterized by hyperglycemia due to either lack of insulin production or insulin resistance. The two main types are type 1 DM caused by autoimmune destruction of beta cells and type 2 DM caused by insulin resistance with relative insulin deficiency.
2. Type 1 DM results from autoimmune destruction of pancreatic beta cells leading to absolute insulin deficiency. Its natural history involves four stages from preclinical autoimmunity to established diabetes with complications. Type 2 DM ranges from predominant insulin resistance to predominant insulin secretory defect.
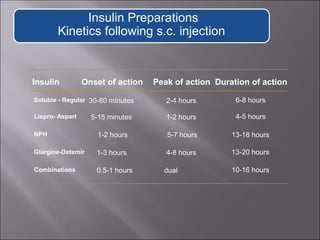
3. The goals of treatment in pediatric patients are to achieve glycemic control while avoiding hypoglycemia and allowing for normal growth and development through insulin therapy, education,