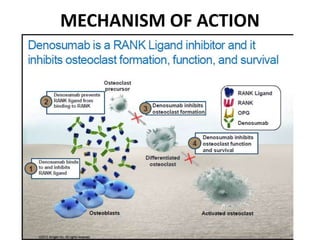
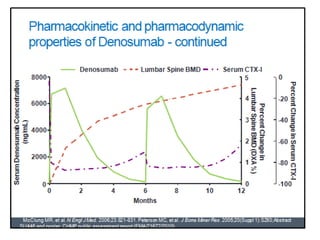
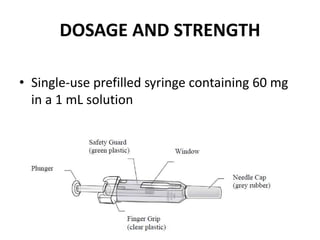
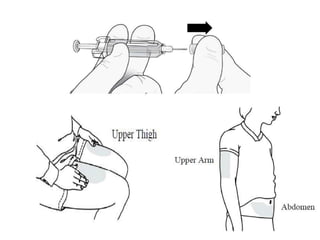
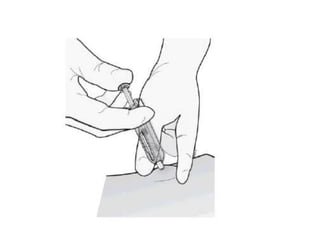
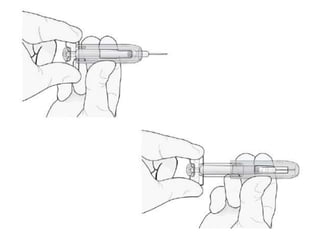
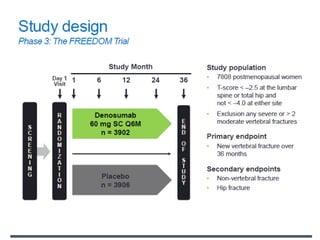
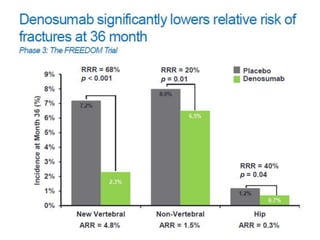
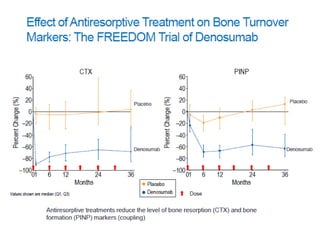
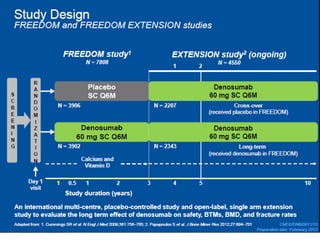
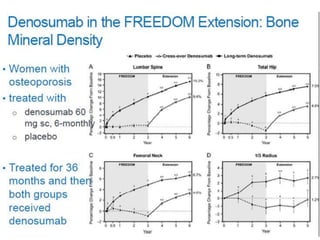
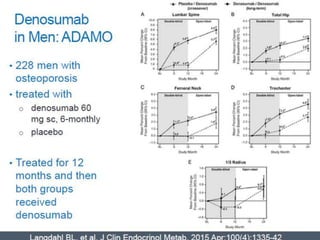
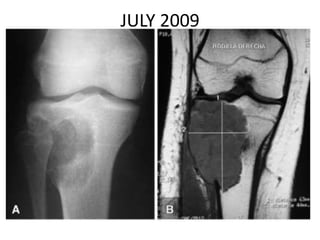
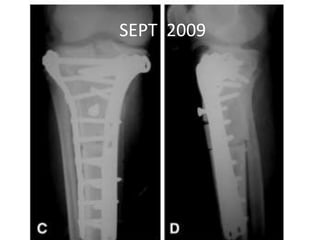
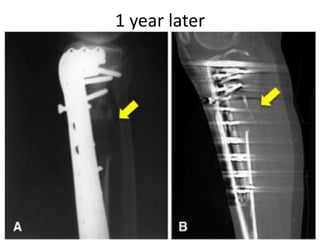
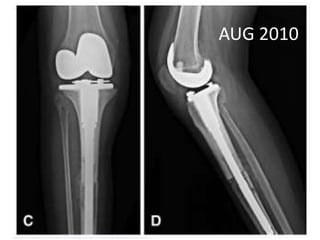
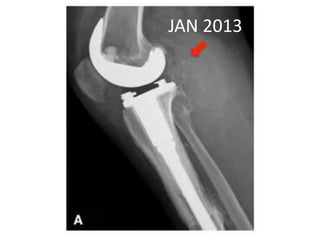
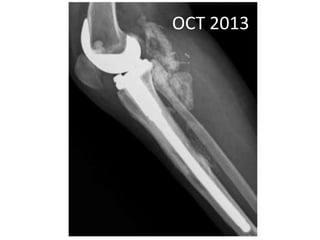
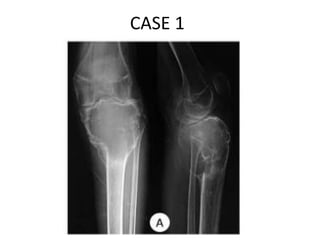
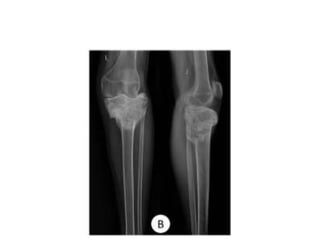
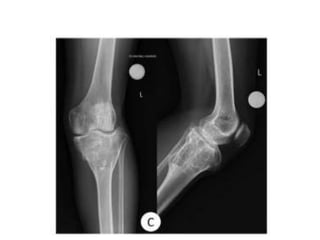
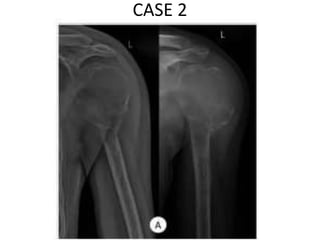
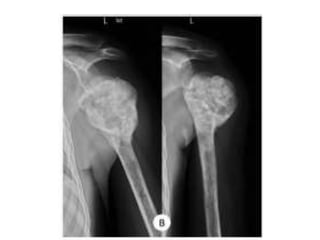
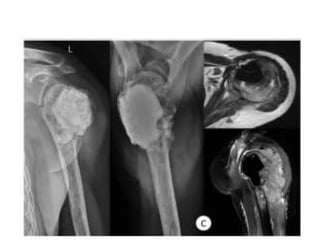
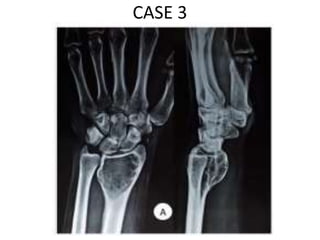
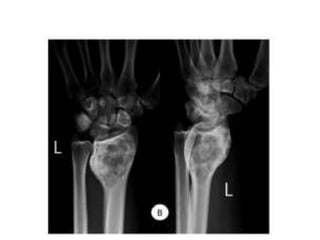
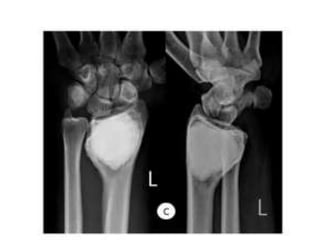
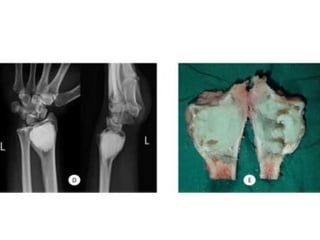
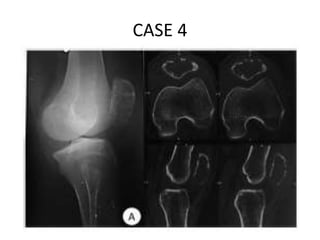
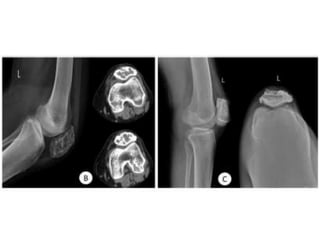
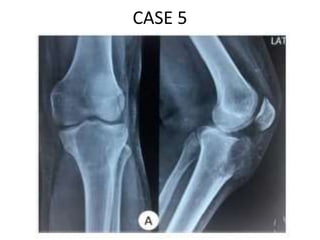
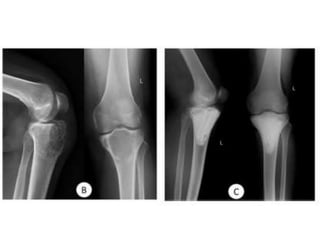
Donesumab is a monoclonal antibody that inhibits RANKL, a protein involved in bone resorption. It is approved to treat osteoporosis and increase bone mass in high risk patients, as well as to prevent skeletal complications in patients with bone metastases or giant cell tumor of bone. Common side effects include back pain, joint pain, and hypocalcemia. While early studies found Denosumab reduced tumor giant cells and risk of recurrence in giant cell tumor patients, one case report found malignant transformation in a patient after long term use. Further research is still needed on its use for giant cell tumor.