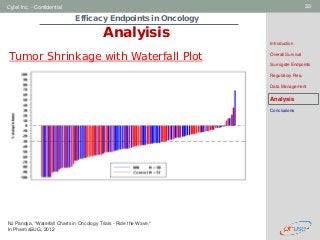
This document discusses efficacy endpoints in oncology drug development. It begins with an introduction to endpoints used in early and late phase trials. Overall survival is discussed as the gold standard, but surrogate endpoints are also examined, including objective response rate, progression-free survival, and time to progression. Considerations for various surrogate endpoints like bias, validation, and data management are provided. The document reviews regulatory requirements and considerations for different cancer types and endpoints.
















![Geneva Branch
Cytel Inc. - Confidential 22
Efficacy Endpoints in Oncology
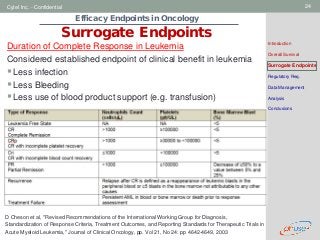
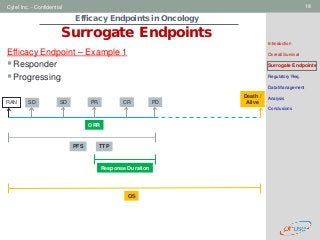
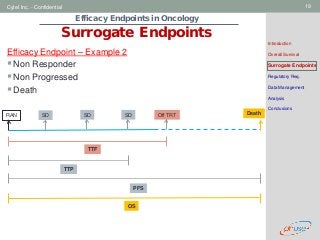
Surrogate Endpoints
Introduction
Overall Survival
Surrogate Endpoints
Regulatory Req.
Data Management
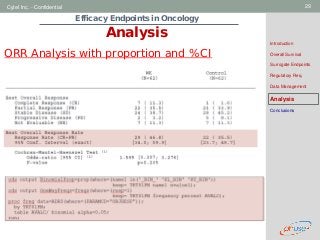
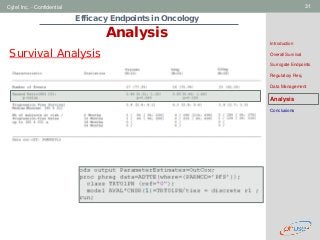
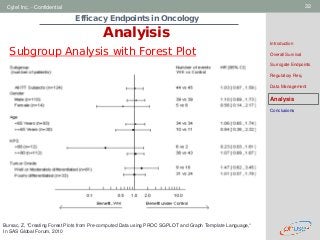
Analysis
Conclusions
Other endpoints: Time to symptom progression (TTSP)
e.g. TTSP in Lung Cancer Trials as per the Lung Cancer
Symptom Scale (LCSS)
Symptomatic progression defined as an increase
(worsening) of the average symptomatic burden index
(ASBI, i.e., the mean of the six major lung cancer
specific symptom scores [fatigue, pain, dyspnoea,
cough, anorexia and hemoptysis])
The worsening is defined as an at least 10% increase
of the scale breadth (i.e., at least 10 mm increase on
the 100 mm scale) from the baseline score.
Hollen PJ, Gralla RJ, Kris MG, et al. Quality of life assessment in individuals
with lung cancer: Testing the lung cancer symptom scale (LCSS). Eur J Cancer.
1993;29A(1):51-8..](https://image.slidesharecdn.com/d5-efficacyendpointsinoncology-150220132854-conversion-gate01/85/D5-efficacy-endpoints-in-oncology-22-320.jpg?cb=1675841753)