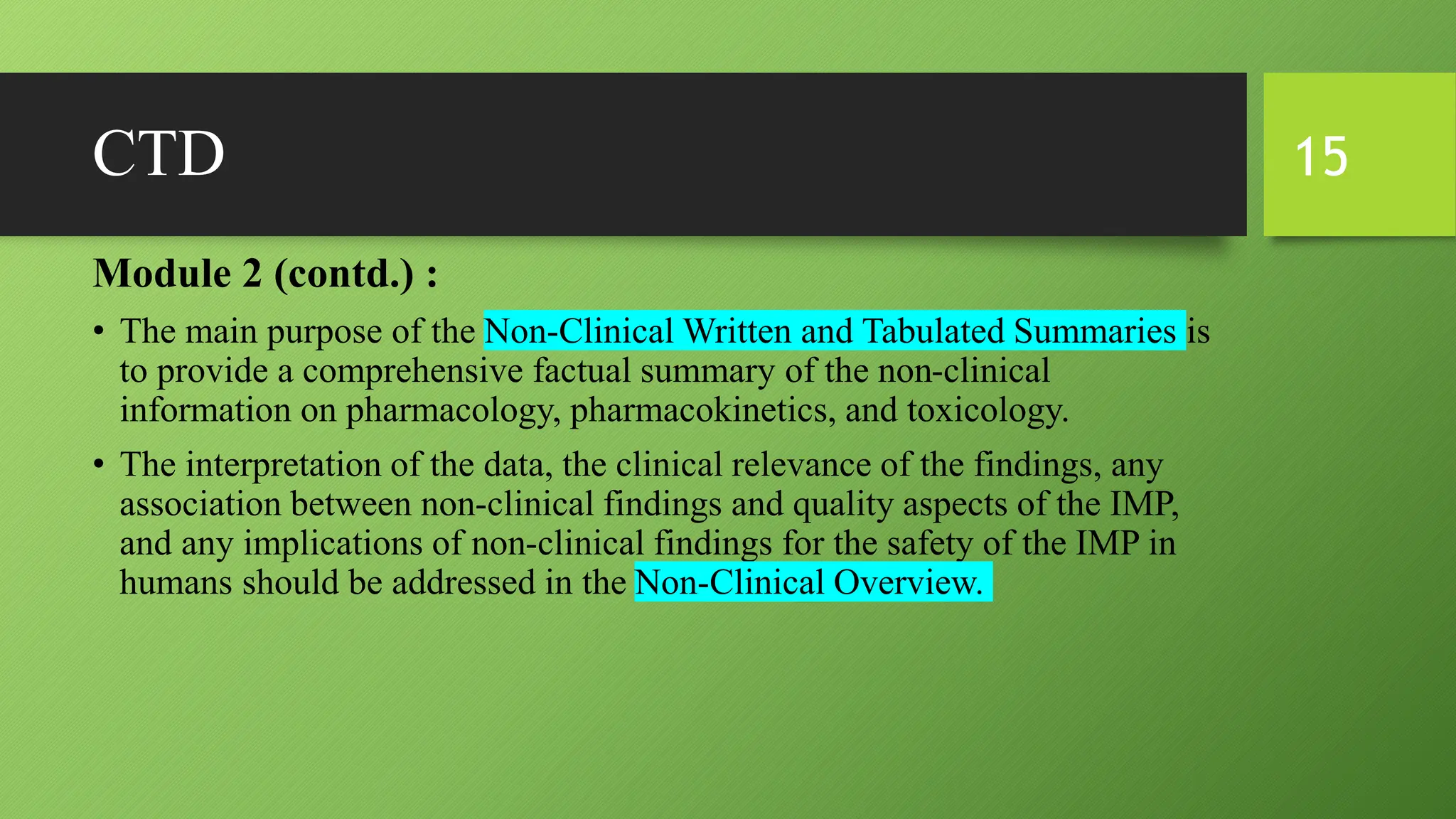
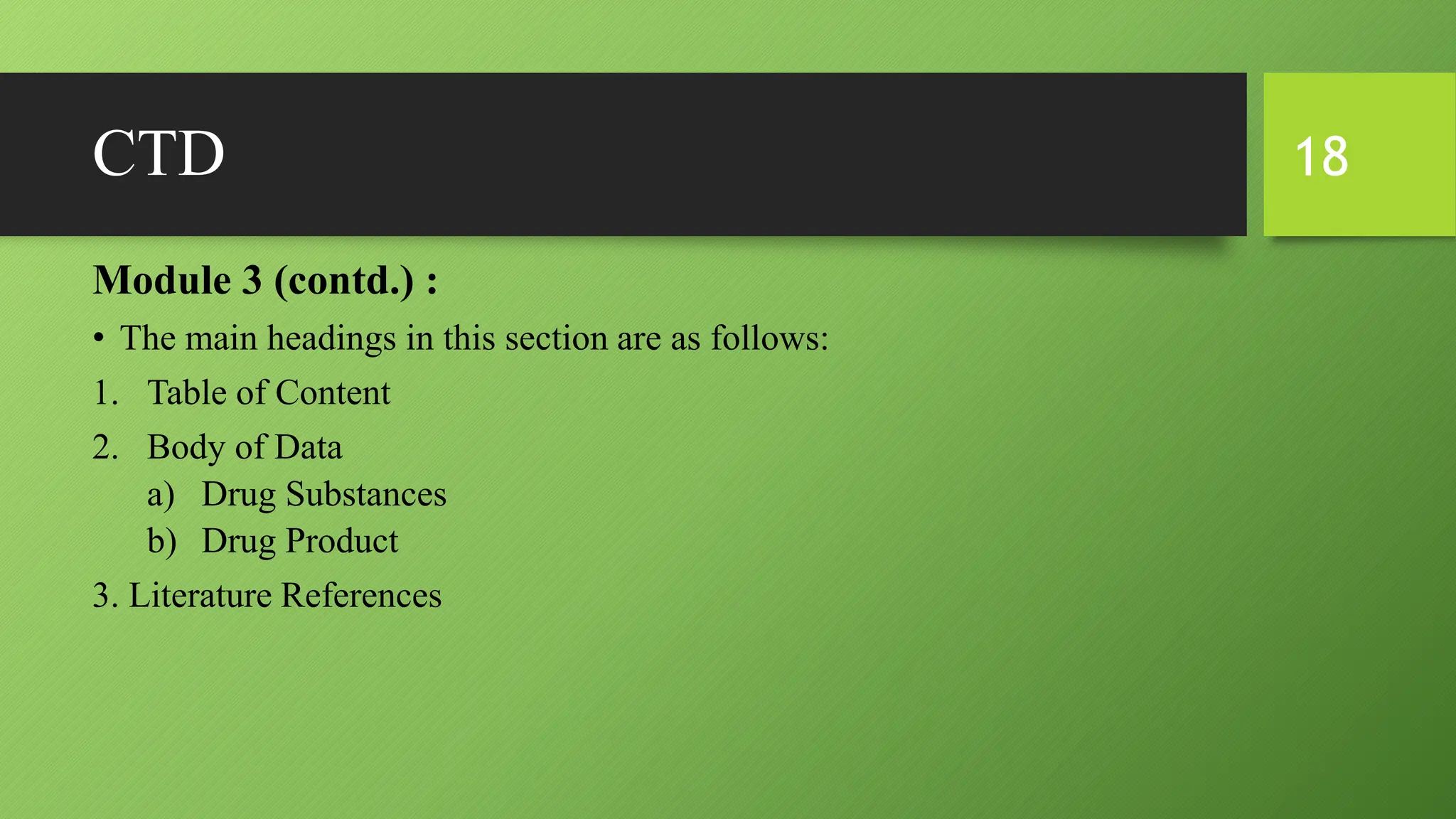
The document discusses CTD (Common Technical Document) and eCTD (electronic Common Technical Document). CTD is an internationally agreed format for organizing technical documents for new drug applications. It was created to harmonize application formats between regions like the US, EU, and Japan. eCTD builds upon CTD by allowing electronic submission and review of applications using an XML structure for easy transfer and organization of information. Both CTD and eCTD aim to streamline the drug approval process globally by creating standardized common formats for application dossiers.