Cpm 14 06 2009 Paper 2 12th (Abcd) Code B
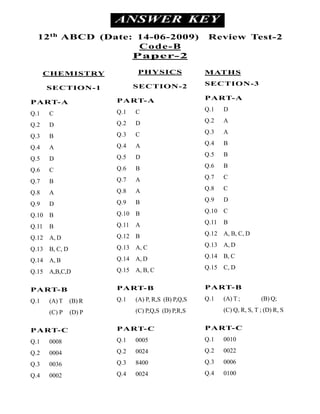
- 1. ANSWER KEY 12 th ABCD (Date: 14-06-2009) Review Test-2 Code-B Paper-2 CHEMISTRY PHYSICS MATHS SECTION-2 SECTION-3 SECTION-1 PART-A PART-A PART-A Q.13 C Q.1 D Q.1 C Q.24 D Q.2 A Q.2 D Q.35 C Q.3 A Q.3 B Q.41 A Q.4 B Q.4 A Q.52 D Q.5 B Q.5 D Q.66 B Q.6 B Q.6 C Q.77 A Q.7 C Q.7 B Q.88 A Q.8 C Q.8 A Q.99 B Q.9 D Q.9 D Q.1010 B Q.10 C Q.10 B Q.1111 A Q.11 B Q.11 B Q.1215 B Q.12 A, B, C, D Q.12 A, D Q.1312 A, C Q.13 A, D Q.13 B, C, D Q.1413 A, D Q.14 B, C Q.14 A, B Q.1514 A, B, C Q.15 C, D Q.15 A,B,C,D PART-B PART-B PART-B Q.1 (A) T (B) R Q.1 (A) P, R,S (B) P,Q,S Q.1 (A) T ; (B) Q; (C) P (D) P (C) P,Q,S (D) P,R,S (C) Q, R, S, T ; (D) R, S PART-C PART-C PART-C Q.1 0008 Q.1 0005 Q.1 0010 Q.2 0004 Q.2 0024 Q.2 0022 Q.3 0036 Q.3 8400 Q.3 0006 Q.4 0002 Q.4 0024 Q.4 0100
- 2. PHYSICS PART-A Q.1 [Sol. n11 = n22 1 4000Å n11 = n22 n2 = n1 = 60 6000Å = 40 ] 2 Q.2 (I) O [Sol. D = 25cm v = –25 cm f = +10 cm 1 1 1 25.10 50 – = 25 u 10 25 10 u= = ] 7 Q.3 Com [Sol. x Greater the value of , lesser the value of x. hence for the sector of angle position of COM will be farther than the semicircle (sector of angle ) ] Q.4 [Sol. Fringe width does not depend on distance between the source and plane of slits. ] Q.5 [Sol. Angular spacing between two consecutive maxima = = =2× ] D d 180 Q.6 [Sol. Luminous flux per unit area × area of hole d d 2 2 = ds = × = ] 4r 2 4r 2 2 16r 2 Q.7 T.I.R. [Sol. S sin i = µ sin r 1 sin = n sin sin = ] 2 n Code-B Page # 1
- 3. PHYSICS Q.8 [Sol. For TIR >c 90 – > c cos > sin c n 2 1 1 > n> 2 ] n n Q.9 [Sol. Chemical energy of explosive is converted to heat which is connected to K.E. of the cannon-ball ] Q.10 [Sol. Force on cannon ball = PA – Patm A ( P Patm ) A acceleration m displacement inside ballerl = length of barrel = v2 = 2as (P Patm )A 2 (51 1) 105 8 10 3 2.5 2 = v= = 100 m/s ] m 20 Q.11 [Sol. PV = nRT dn dV = R' & = A.V. . dt dt dV dn P = RT T dt dt PAv = R' RT v R' ] Q.12 [Sol. We know VIBGYOR Since in option V & Y are not given so possible are BG YOR R > B R < B By cauchy’s relation 1 sin C = and by T.I.R. so (C)B < (C)R so there is probability of reflection of Blue colour so D must be blue and C nearest to blue which is green R > y so R < y i.e. A is red B is yellow. Q.13 R r Fv FR [Sol. 10cm fR Code-B Page # 2
- 4. PHYSICS From similar triangles, r fR fv = f R R 1 f R 10 100 = f fR = 10 R 9 1 2 f R = (µR – 1) R 2 9 (µR – 1) = µR = 1.27 6 100 fR fv fR fv 100 / 9 10 1 = fy fR = = ~ 0.1 ] 100 / 9 10 Q.14 [Sol. We know O2 = 5 7 He = , 3 5 PV = const. L 5/3 PHe (LA) = 5/3 A 2 PHe PHe = PHe (2)5/3 ... (1) PO 2 (LA)7/5 = PO 2 (LA)7/5 L 7/5 L PO 2 = PO 2 ... (2) Now PO 2 PHe PO 2 PHe L PO 2 PHe 7/5 1 = = L PO 2 PHe 5/3 2 7/5 L7 / 5 (L) = 25 / 3 L L L = 5/3 5/ 7 = 2 x 2 25 / 21 LA so volume of O2 = LA = ] 2 25 / 21 Code-B Page # 3
- 5. PHYSICS Q.15 [Sol. (A) For convex mirror object is not at focus (focus is on other side). The mirror will cause the diverging incident beam to diverge even more. (B) The rays emerging towards left from the lens must be parallel to P.A. (C) Object is between optic center and focus. Image will be diverging (virtual) (D) Initial object is at O whose image is formed at pole of mirror by the lens. Hence find image will also be at O. ] PART-B Q.1 [Sol. (1) y1 = n11 = n22 = LCM of 1 and 2 2y1 = 2n11 = 2n22 Hence at this point both maxima again coincide. 1 1 y2 = n1 1 = n 2 2 2 2 n2 1 1 2 2 = n1 1 2 1 2n 2 1 1 odd 2 = 2n 1 which will have a solution. Iff expressed as a proper fraction will be of form . 1 2 odd 1 odd Option B & C : is of form . Hence no solution i.e. the two minima will never coincide. 2 even 1 odd For option A& D : is of form . Hence at some finite y2 the two minima will coincide. 2 odd At 2y2 the two maxima (and not minima) will coincide. y = 3y2 is the next nearest point where minima coincide. ] PART-C Q.1 [Sol. New volume of flask = 2(1 + 3Glass T) litres (1dm3 = Litre) New volume of water = 1(1 + Ywater T) litres New volume of iron = 1(1 + 3iron T) dm3 Volume of water overflow = Vwater + Viron – Vflask = (Ywater + 3iron – 6Glass)T = [(180 + 3 × 12 – 6 × 8) × 10–6] × 50 = 168 × 50 × 10–6 = 8400 × 10–6 litres = 8400 mm3 ] Q.2 [Sol. Heat released by water = Heat absorbed by ice (15 × 103(gm))(cal/gm) × (80 – 20)°C = ms (20°C – 0°C) + mL = (100 cal/g) m(gram) Code-B Page # 4
- 6. PHYSICS 15 60 m= × 103 gm = 9kg 100 total water = 15 + 9 = 24 kg minimum volume of vessel = 24 litres. ] Q.3 dv Adx [Sol. Temperature is constant E = 0, dW = nRT = nRT v AL / 2 Q = E + W dW nRT dx = dt L / 2 dt 1 T 2nRT dx dQ dW L dt k = dt dt 900 L dx k 27 415.5 27 2 1 900 0.5 8.31 300 200 = = = m/s = 5 mm/s ] dt 900.nRT Q.4 [Sol. Heat supplied (1 2 & 2 3) = nCv9T0 + nCp90T0 Heat rejected (3 4 & 4 1) = nCv90T0 + nCp9T0 10C v C p 10 = 1 C 10C = 1 – 11.4 3.6 12 =1– p 1 10 = = V 15 15 50 %efficiency = 24% ] Code-B Page # 5
- 7. CHEMISTRY PART-A Q.1 [Sol. H = V + (PV) V = W = – 4 × – 10 = 40 l-bar (PV) = P2V2 – P1V1 = 4 × 30 – 2 × 40 = 40 l – bar ] Q.2 [Sol. (A) nBeH2(g) Cool. (BeH2)n(s) H H H –– Be –– H Be Be Be Hyb. of Be : sp H H (gas) 3 .. Hyb. of Be : sp .. (solid) .. ..O O. . (B) .. [NO2]+ [NO3]+ N–O–N .. Hyb. of N in NO2+ : sp .. Hyb. of N in NO3¯ : sp2 . . O. . O: .. 2 (N2O5 in solid state) Hyb. of each N:sp (gaseous state) (C) XeF6 [XeF5]+ F¯ Gaseous state Solid state Hyb. sp3d3 Hyb. sp3d2 (D) PF5 PF5 Gaseous state Solid state Hyb. : sp3d Hyb.: sp3d ] Q.3 [Sol. q = – E I law E = – + 2E = 2CVdT = – PdV RT 2CVdT = – dV V 2C V dT dV – = ] R T V Q.4 a P 2 (V – b) = RT V [Sol. T RT a – 2 Vb V P= PV V a – V b VRT Z= = RT Code-B Page # 1
- 8. CHEMISTRY a T V b – Vb V Rb = b2 V ( V b) =1+ ] Q.5 [Sol. Tertiary alkyl halides can take part in the reaction but prefers disproportionation so the yield of dimerized product will be less but it can show Wurtz reaction. ] Paragraph for question nos. 6 to 8 Me CH2 CH3 Me Me [Sol. + + Me Me CH3 CH3 CH3 CH3 Me (X) (R) 3° Free radical (P) 1° Free radical (Q) 2° Free radical ] CH2–Cl CH3 Me Me Cl H Cl CH3 CH3 CH3 CH3 Me Dia + Dia + CH3 Me Me H Cl Me CH3 CH3 Cl 9 9×1 =9 1°Product = × 100 = 19.86% 45.3 22.8 6 × 3.8 = 22.8 2° Product = × 100 = 50.33% 45.3 13.5 3 × 4.5 = 13.5 3° Product = × 100 = 29.80% 45.3 45.3 Code-B Page # 2
- 9. CHEMISTRY Q.9 F N [Sol. (A) F F F Hybridisation of N : sp3 FNF = 109° As all substituents are same Cl C (B) Cl Cl Cl Hybridisation of c : sp3 l CC = 109° As all substituents are same. O¯ O O O (C) Cl Cl Cl Cl O O O¯ O O O O O¯ O O O¯ O Hybridisation of Cl : sp3 OClO : 109° , as well Cl–O bonds are of equal length. ] Q.10 F F Br —F < 90° [Sol. (A) B F F F Hybridisation of B : sp2 Hybridisation of Br : sp3d Shape : Trigonal planar Shape : Bent 'T' shape. Cl I 180° Cl — Be — Cl (B) Cl Hybridisation : sp3d Hybridisation : sp Shape : linear Shape : linear Code-B Page # 3
- 10. CHEMISTRY Cl (C) B P Cl Cl Cl Cl Cl Hybridisation of N : sp3 Hybridisation of P : sp3 Shape : Trigonal planar Shape : Triangular pyramidal N P (D) Cl Cl Cl Cl Cl Cl Hybridisation of N : sp3 Hybridisation of P : sp3 Shape : Triangular pyramidal Shape : Triangular pyramidal. ] Q.11 H C [Sol. (A) C H (B) H F H F Triangular pryamidal (Planar) (Nonplanar) • C N (C) F F (D) O O Planar (singlet carbene) (Planar) Q.12 [ B]t k1 [Sol.(A) [C] = 2k = 1 t 2 k1 (B) ln E a for same temp. change k 0.693 (C) t1/2 = k k = 2.772 sec. ] 1 2 Code-B Page # 4
- 11. CHEMISTRY Q.13 [Sol. NCl3 undergoes hydrolysis and H2O attacks at Cl-atoms. H 2SO3 4HF SF4 + 3H2O R .T. Pr oton donor acids + Cl OH2 B B¯ H 2O Cl Cl Cl Cl 2 Cl Hyb. sp 3 Hyb. sp (Transition state) Q.15 COOH COOH NO2 [Sol. (A) Ethanol can solvate anion of as compared to anion of O2N NO2 (B) H H N N N N No flipping 2°amine 3°amine l.p.is present in more EN orbital (C) L.G. tendency does not depend on solvent nature. (D) Polar Aprotic solvent does not solvate anion. ] PART-B Q.1 Potential Energy [Sol. (A) As after rearrangement two product are formed. Reaction progress (B) No rearrangement (C) Two products formed. (D) Two products formed. ] Code-B Page # 5
- 12. CHEMISTRY PART-C Q.1 [Sol. WAB = WCD = 0 D C WBC = – nRT = – 1 × R × 300 = 300 R 40 WDA = – nRT = – 1 × R × – 200 = 200 R V (L) Wtotal = – 100 R 20 lit. A B |W| = 8 atm. lit. ] 200 300 400 600 T (K) Q.2 [Sol. AsCl3 + 3H2O H 3AsO3 3HCl ] Basicity 3 Basicity 1 Total basicity of products 4 Q.3 [Sol. A(g) B(g) + 2C(g) P0 – – P0 – x x 2x – P0 2P0 if N is pressure of inert gas P0 + N = 200 3P0 + N = 500 P0 = 150 ; N = 50 at t = 70 sec. Pt = 350 = P0 + 2x + N x = 75 t = t1/2 = 70 sec. 0.693 k (hr–1) = t sec × 3600 ] 1/ 2 Q.4 [Sol. Cl –– Be –– Cl No. of 2C – 2e– bonds = 2 Total no. of 2C – 2e– & No. of 3C – 4e– bonds = 0 3C – 2e– bonds = 2 ] Code-B Page # 6
- 13. MATHEMATICS PART-A Q.1 x2 t2 x2 [Sol. 2 f ' (2x) = x2 f ' (2x) = ; put 2x = t f ' (t) = f ' (x) = Ans.] 2 8 8 Q.2 dx Let cos–1x = t – 1 x2 [Sol. = dt (1 cos t ) sin t 1 sin t I = – e t dt = – e t 1 cos t (1 cos t ) 2 dt (1 cos t ) 2 1 et e cos x =C– =C– 1 cos t Ans. ] 1 x Q.3 [Sol. Equation of line : y – 1 = m(x – 7) mx – y + 1 – 7m = 0 r | 7m 1 | r 1 m2 Perpendicular distancer from (0, 0)= = =5 2 2 (7m – 1) 2 = 25(1 + m2) 49m2 – 14m + 1 = 25 + 25m2 24m2 – 14m – 24 = 0 m1m2 = – 1 Ans. ] Q.4 2 x (5 x 1) (5 x 1) ( 2 x 1) (5 x 1) Lim ( 2 1) (5 1) x x l = Lim = Lim x · x ln (sec x )1 x [Sol. = x 0 2 x 0 ln (sec x ) x 0 ln (sec x ) x 2 ln 2 · ln 5 (ln 2 · ln 5) x 2 = Lim x 0 ln (1 (sec x 1)) = Lim = 2 (ln 2 · ln 5) = (ln 4) (ln 5) Ans.] x 0 (1 cos x ) Q.5 a a [Sol. 1 r1 = r1 and 1 r2 = r2 a r2 – r + a = 0 1 r hence r1 and r2 are the roots of =r r1 + r2 = 1 Ans.] Code-B Page # 1
- 14. MATHEMATICS Paragraph for question nos. 6 to 8 [Sol. y = f (x) = x 2 – 4ax + a2 + 5a + 2 4a vertex = = 2a 2 (i) f (2a) = 4a2 – 8a2 + a2 + 5a + 2 = – 3a2 + 5a + 2 2 5a 2 5 25 24 49 2 5 2 = – 3 a = – 3 a = 3 a 3 3 6 36 35 12 6 5 f (2a) is maximum if a = Ans.(i) 6 (ii) f (x) 0 x R x2 – 4ax + a2 + 5a + 2 0 D0 16a2 – 4(a2 + 5a + 2) 0 4a2 – a2 – 5a – 2 0 3a2 – 6a + a – 2 0 (3a + 1)(a – 2) 0 – 1/3 a 2 amax = 2 Ans.(ii) (iii) Given minimum value of f (x) is 2, for x 0 Case-1: when vertex is 0 i.e. 2a 0 a0 minimum f (x) occurs at x = 2a f (2a) = 4a2 – 8a2 + a2 + 5a + 2 = – 3a2 + 5a + 2 f (2a) = 2 a(5 – 3a) = 0 a = 0 or a = 5/3 Case-2: when vertex is < 0 i.e. a<0 in this case, minimum occurs when x = 0 f (0) = a2 + 5a + 2 = a(a + 5) = 0 a = 0 or a = – 5 hence a {0, – 5, 5/3} 3 values of 'a' Ans.(iii) ] Paragraph for question nos. 9 to 11 Sol. We have r sin x x (r 1) n 6 6 r f1(x) = 2 r 1 cos x (r 1) · cos x 6 6 2 3 2 = 2 · tan x tan x tan x tan x tan x tan x 6 6 6 6 6 n ................ tan x tan x (n 1) 6 6 n f1(x) = 2 · tan x tan x 6 for n = 3, f1(x) = 2 tan x tan x = 2 cot x tan x = – 2 1 2 sin x cos x Code-B Page # 2
- 15. MATHEMATICS n 2 f2(x) = f1(x) – 2 · tan x 6 Now n n = 2 tan x – 2 tan x – 2 tan x 6 6 f2(x) = – tan x f3(x) = – f2(x), so f3(x) = tan x e( e x 1) 1 2·(e x 1) e ; x 0 Now f 4 ( x ) k1 ; x0 1 | tan x | tan x ; x 0 k2 1 f (0¯) = e 2 e lne Clearly f (0+) = e k 2 & f (0) k1 (i) As f4(x) is continuous at x = 0, so by definition of continuity, we get f (0¯ ) = f (0+) = f (0) e = e k 2 = k1 1 k1 e & k2 = (D) 2 (ii) As y = f3(x) = tan x clearly f3(x) is continuous as well as derivable everywhere in 0, 2 . (C) (iii) for n = 3, f1(x) = – 4 sin x cos x – 2(tan x + cot x) = – 4 cos x sin x = 2 1 = 2 sin x · cos x 0, 4 2 x= (B)] Q.12 [Sol. (A) f (x) = x – cos x ; f (0) < 0, f (/2) > 0 (B) f (x) = x + sin x – 1 1 f (0) = – 1 < 0; f (/6) = 1 0 6 2 (C) f (x) = a(x – 3) + b(x – 1) in [1, 3] f (1) = – 2a < 0; f (3) = 2b > 0 f (x) = 0 in (1, 3) (D) h (x) = f (x) – g (x) h (a) = f (a) – g (a) > 0 h (b) = f (b) – g (b) < 0 hence using IVT all the four have at least one root in indicated interval. ] Code-B Page # 3
- 16. MATHEMATICS Q.13 [Sol. g ' (x) = aeax + f ' (x) g ' (0) = a + f ' (0) = a – 5 ....(1) g '' (x) = a2eax + f '' (x) g '' (0) = a2 + f '' (0) = a2 + 3 ....(2) g ' (0) + g '' (0) = 0 a – 5 + a2 + 3 = 0 a2 + a – 2 = 0 (a + 2)(a – 1) = 0 a = 1, – 2 Ans.] Q.14 = 1 cos 1 cos 1 cos 1 cos 4k 2 4k 2 4k 4k [Sol. P(k) = 1 cos 1 sin 1 sin 1 cos 4k 4k 4k 4k 4 sin 2 · cos 2 = 1 cos 1 sin = 2 2 4k 4k 4k 4k 4 1 2 sin · = 1 1 1 2k P(k) = P(3) = ; 4 4 4 16 2 1 2 1 sin sin 1 cos = 2 2 (B) 8 8 4 P(4) = 4 2k 4 16 1 2 1 2 1 5 1 3 5 = 2 sin = 1 cos 36 = 1 1 sin 4 = 32 (C) 8 10 8 P(5) = 8 4 10 1 2 1 2 1 1 3 2 3 = 2 sin = 1 cos = 1 2 = 16 sin 8 12 8 6 8 P(6) = ] 4 12 Q.15 [Sol. for continuity at x = 0 1 f (0) = 0 ; f (0–) = 0; f (0+) = Lim h sin n =0 n>0 h0 h for derivability at x = 0 1 h n sin f ' (0–) = 0; f ' (0+) = Lim h for this limit not to exist n 1 h0 h hence 0 < n 1 n can not be 3/2 or 2 C, D ] Code-B Page # 4
- 17. MATHEMATICS PART-B Q.1 [Sol. e ln x ! x! = 20 = 20 x(x – 1) = 20 x2 – x – 20 = 0 (x – 5)(x + 4) = 0 ( x 2)! ( x 2)! (A) x = 5 Ans. (T) (B) 4{x} = x + [x] ....(1) 2x 2x 5 = x + x – {x} 5{x} = 2x {x} = 0 <1 0x< 5 5 2 hence [x] = 0, 1, 2 Again from (1) 4x – 4[x] = x + [x] 3x = 5[x] Case-I: If x [0, 1) [x] = 0 3x = 0 x0 Case-II: If x [1, 2) [x] = 1 x 5 3x = 5 3 Case-III: If x (2, 5/2] [x] = 2 10 3x = 10 x= (reject) 3 number of solutions = 2 Ans. Alternatively : Let x = I + f 2I 2 4 4f = I + f + I f= I = 0, 1 x=0&x=1+ = 3 3 3 (C) Given equation is x2 + 2x(y + g) + y2 + 2fy + 4 = 0 2 x = – 2 (y + g) ± 4( y g) 2 4( y 2 2fy 4) x = – (y + g) ± (g 2 4) 2 y(g f ) ....(1) (1) will represent a pair of lines if its discriminant is zero + 4(g – f) = 0 g=f x = – (y + g) ± g 2 4 . For two real lines g2 4 g 2 or g – 2 Ans. Q, R, S, T (D) If d = 6; A = (0, 0); B(6, 0) Consider a circle with centre A are radius 2(r1) and a circle with centre B and radius 3(r2). The circles will be separated. There will 4 common tangents at a distance of 2 from A and 3 from B 4 lines |||ly if d = 5; r1 + r2 = 5 circles touches externally 3 common tangents 3 lines R, S NOTE: |||ly for two intersecting circles r2 – r1 < d < r1 + r2 i.e. 1 < d < 5 2 common tangents 2 lines if d = r2 – r1 i.e. if d = 1 circles touches internally 1 lines ] Code-B Page # 5
- 18. MATHEMATICS PART-C Q.1 [Sol. AAAAA | B B B B B B M Middle digit must be A (think !) so that even number of A's and B's are available Take AABBB on one side of M (6th place) and then their image about M in a unique way 5! Number of ways = 2!· 3! = 10 Ans.] Q.2 [Sol. Using L'Hospital's Rule using f g h ' + g h f ' + h f g' 5 cos 4 x (sin x ) cos 3 2x cos 3 3x 3 cos 2 2 x 2 sin 2x cos 5 x cos 3 3x 3 cos 2 3x 3 sin 3x cos 5 x cos 3 2 x Lim x 0 2x 6 3 = 22 Ans. 5 9 = 2 2 Alternatively: Using expansion x2 (2x ) 2 9x 2 5 3 3 5x · cos32x · cos33x = 1 .... · 1 .... · 1 .... cos 2! 2! 2! 5x 2 1 = .... 1 6 x 2 .... 27 x 2 1 .... 2 2 5 2 27 x 2 1 6 x .... 1 = .... 2 2 27 x 2 5x 2 = 1 6x 2 ......... = 1 – 22x2 + ..... 2 2 1 cos5 x · cos3 2 x ·cos3 3x 1 (1 22x 2 ....) Lim = Lim = 22 Ans. ] x 0 x2 x 0 x2 Q.3 [Sol. Let the roots are a – 3d, a – d, a + d, a + 3d sum of roots = 4a = 0 a=0 hence roots are – 3d, – d, d, 3d m product of roots = 9d4 = m2 d2 = ....(1) 3 again x 1x 2 = 3d2 – 3d2 – 9d2 – d2 – 3d2 + 3d2 = – 10d2 = – (3m + 2) = 10d2 = 3m + 2 10m = = 3m + 2 3 = 10m = 9m + 6 Code-B m = 6 Ans. ] Page # 6
- 19. MATHEMATICS Q.4 [Sol. Let P(x) = k(x – 1)(x – 3)(x – 5)(x – r) where r 1, 3, 5 ln P(x) = ln k + ln (x – 1) + ln (x – 3) + ln (x – 5) + ln (x – r) differentiate w.r.t. x P' ( x ) 1 1 1 1 x 1 x 3 x 5 x r = P( x ) put x = 7, as P'(7) = 0 P(7) 0 23 6 1 1 1 1 1 1 11 =0 =0 6 4 2 7r 12 7r r 7 12 12 89 p r–7= r= = p + q = 100 Ans.] 11 11 q Code-B Page # 7
