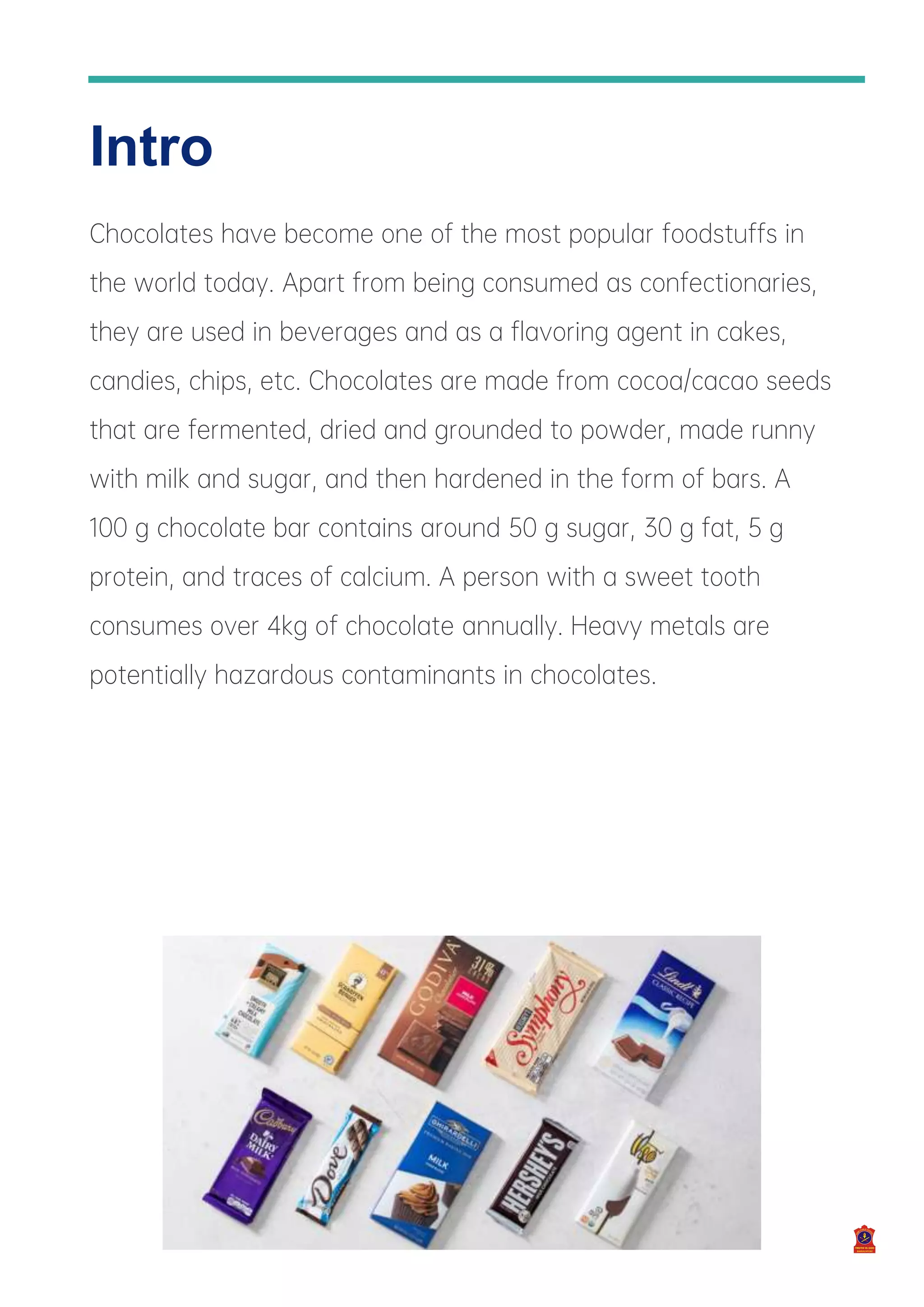
The investigatory project by Shubhrajit Sadhukhan analyzes the contents of different chocolates available in the market, focusing on the presence of sugars, fats, proteins, calcium, and heavy metals. The project findings indicate that tested chocolate samples contained sugar, fat, protein, and calcium, while no iron, magnesium, or nickel was detected. The work includes an introduction, methodology, experimental results, a conclusion, and acknowledgments.