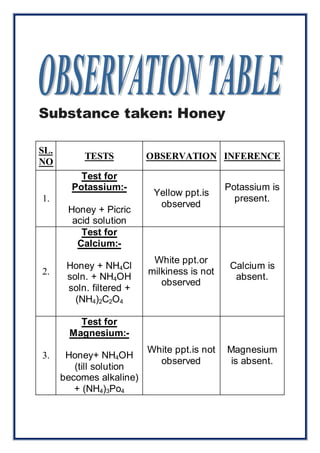
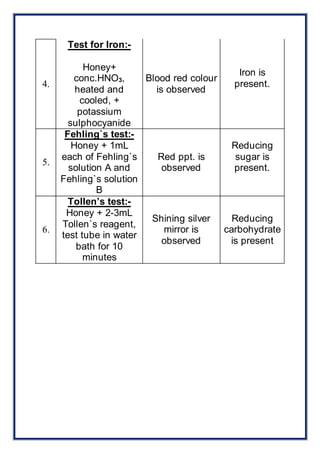
The project titled 'Analysis of Honey' by Akshara Gupta for the 2014-2015 practical examination investigates the presence of various minerals and carbohydrates in honey. The study employs specific tests to identify the presence of potassium, calcium, magnesium, and iron, as well as reducing sugars. The findings indicate potassium and iron are present, while calcium and magnesium are absent, and honey contains reducing sugars.








![Honey has a fuel value of about 3307
cal/kg [1520 cal/ lbs]. It readily picks up
moisture from the air and is
consequently used as a moistioning
agent for Tobacco and in baking.
Glucose crystallizes out of honey on
standing at room temperature, leaving
on uncrystallized layer of dissolved
fructose. Honey to be MARKETED is
usually heated by a special process to
about 66oC [150.01 F] to dissolve the
crystals and is sealed to prevent
crystallization. The fructose in
crystallized honey ferments readily at
about 160C.](https://image.slidesharecdn.com/honeyisbunny2-141214055856-conversion-gate01/85/chemistry-project-for-class-12-on-analysis-of-honey-9-320.jpg)