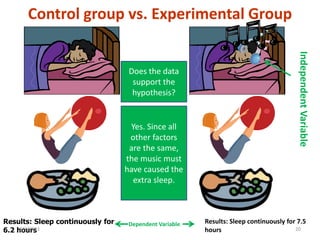
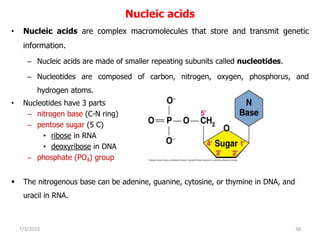
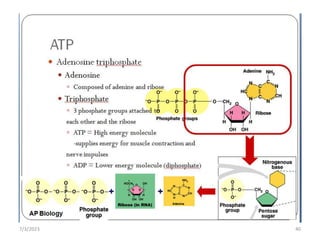
1. Biological molecules are essential components of living organisms and perform many important functions in cells. The four major classes are carbohydrates, lipids, proteins, and nucleic acids.
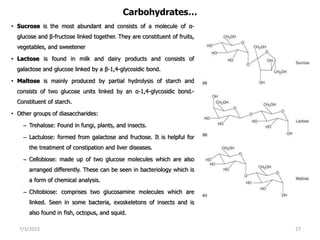
2. Carbohydrates are composed of carbon, hydrogen, and oxygen and serve as important energy sources. They include sugars, starches, and cellulose.
3. Carbohydrates can be classified as monosaccharides, disaccharides, or polysaccharides. Common monosaccharides include glucose and fructose, which differ in whether they have an aldehyde or ketone functional group.