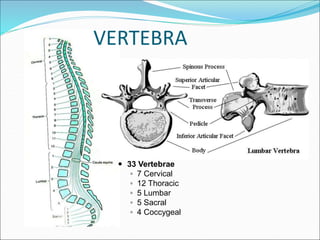
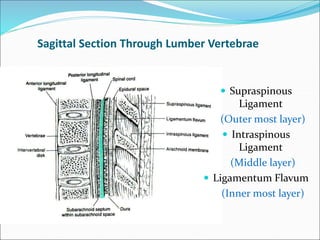
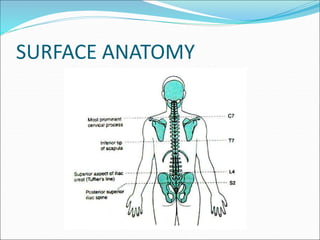
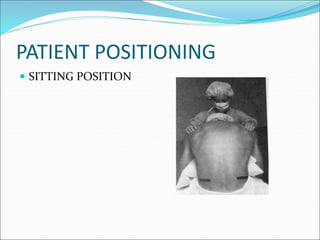
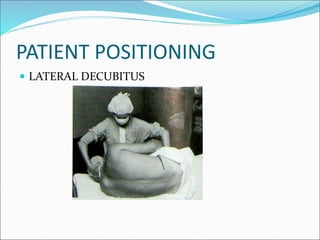
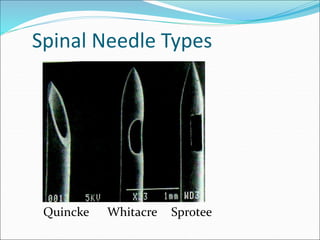
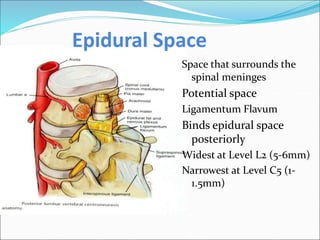
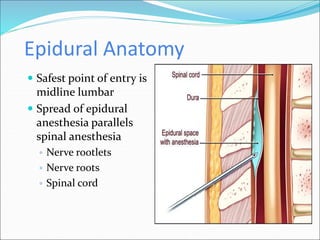
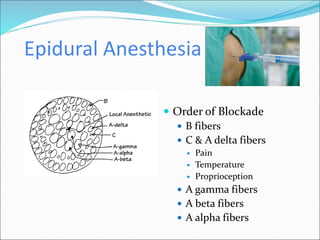
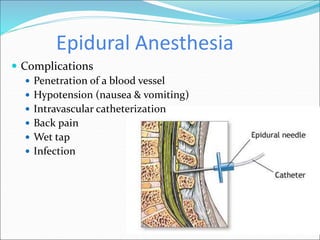
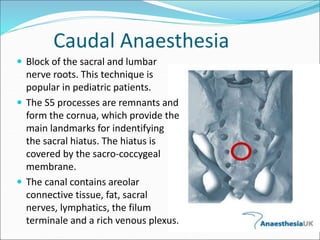
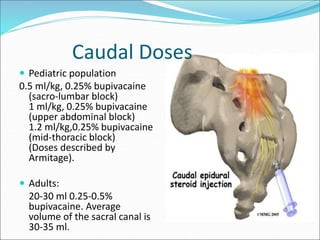
Neuraxial anesthesia involves injecting anesthetic medication into the epidural space surrounding the spinal nerve roots or into the cerebrospinal fluid surrounding the spinal cord. This numbs the patient from the abdomen to the toes and often eliminates the need for general anesthesia. There are several types of neuraxial anesthesia including spinal, epidural and caudal blocks. Potential complications include hypotension, neurological issues, infection and post-dural puncture headache. Proper patient positioning, drug selection and dosage are important factors for achieving the desired level and density of the block with the fewest complications.