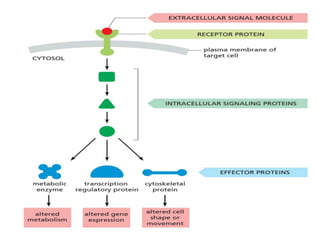
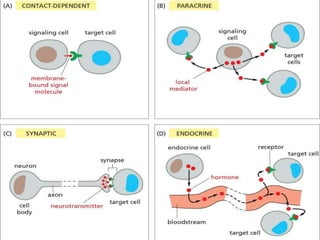
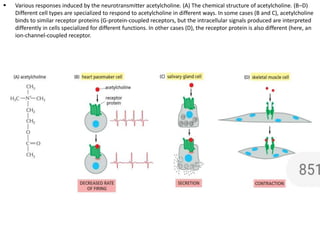
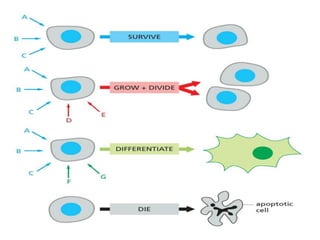
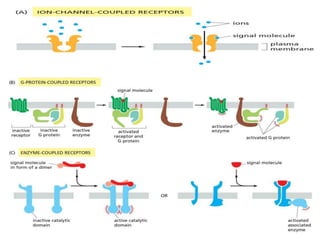
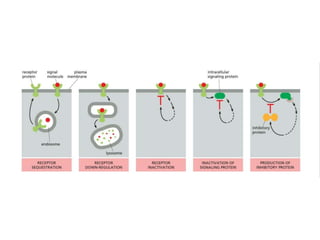
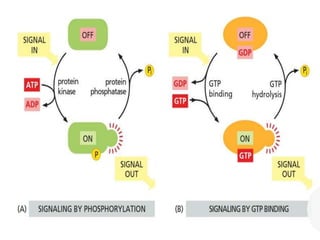
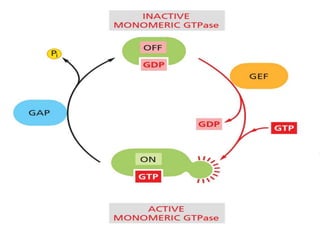
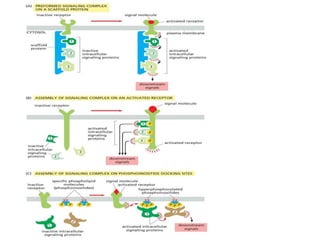
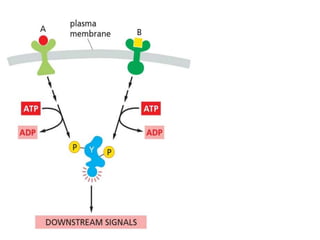
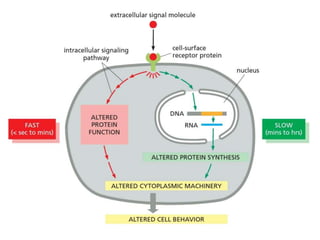
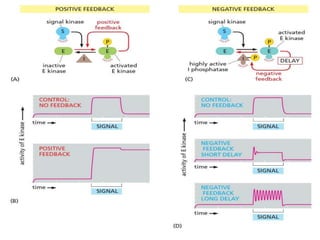
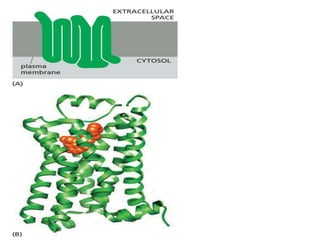
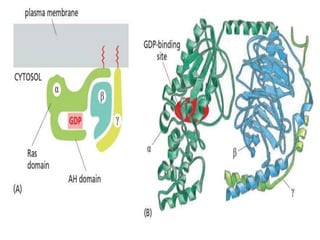
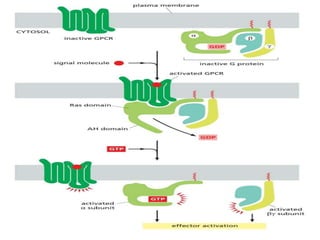
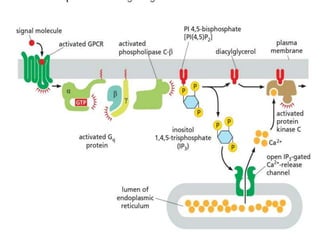
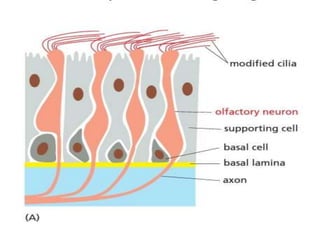
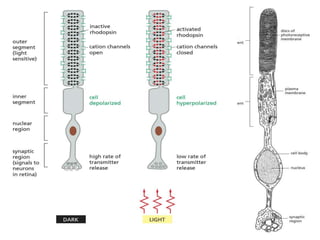
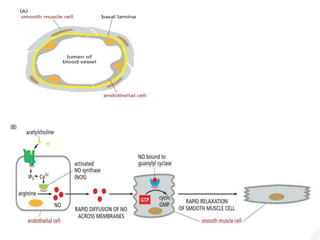
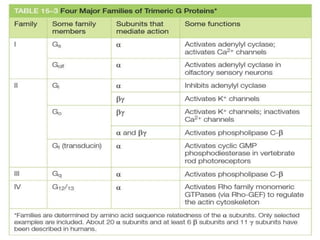
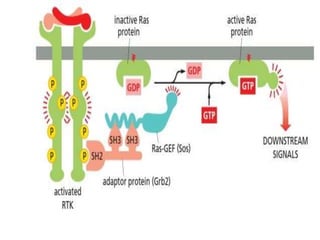
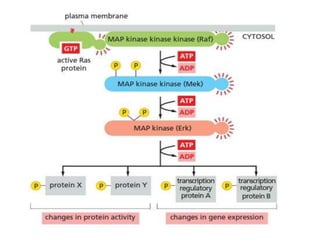
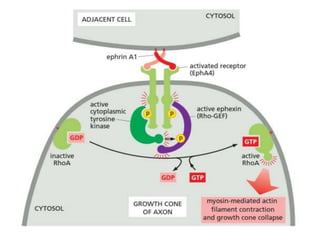
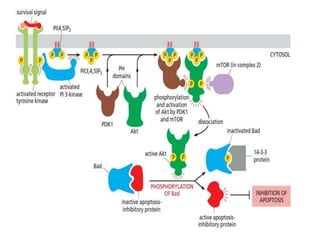
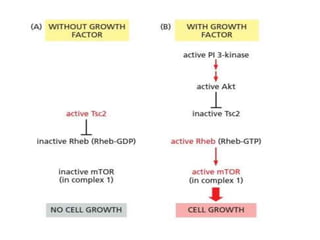
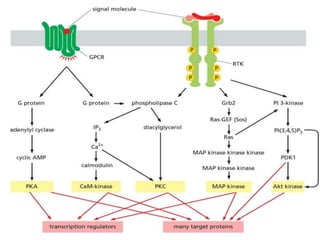
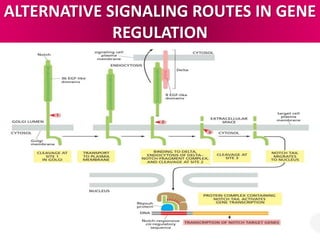
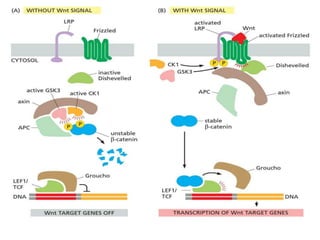
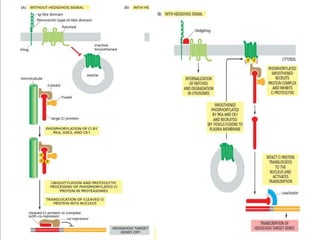
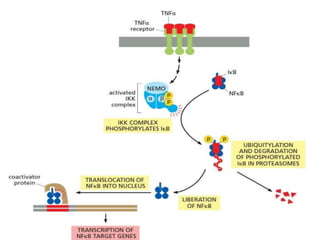
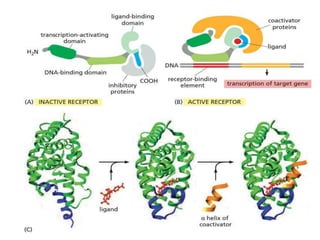
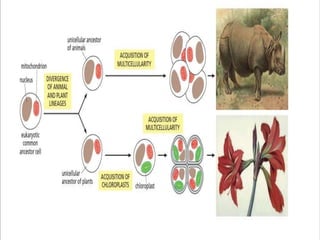
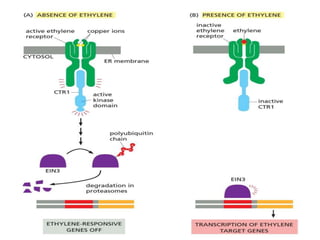
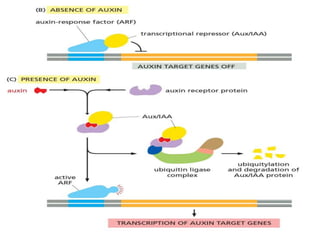
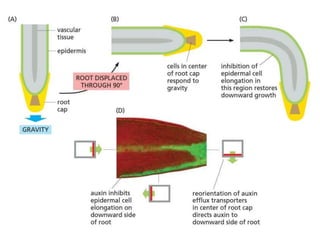
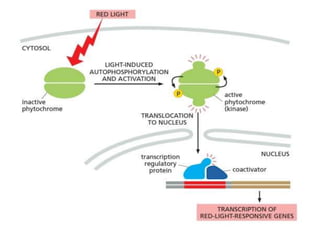
The document discusses cell signaling in multicellular organisms, detailing how cells detect and respond to external and internal signals through receptor proteins and regulatory pathways. It describes various signaling mechanisms such as paracrine and autocrine signaling, the role of neurotransmitters, and the complex intercellular communication required for coordinated cell behavior. Additionally, it highlights the importance of G-protein-coupled receptors and enzyme-coupled receptors in mediating responses to a diverse array of signal molecules, emphasizing their roles in processes like growth and differentiation.