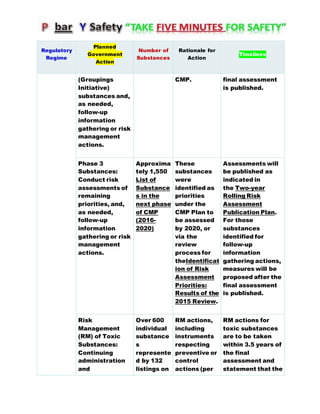
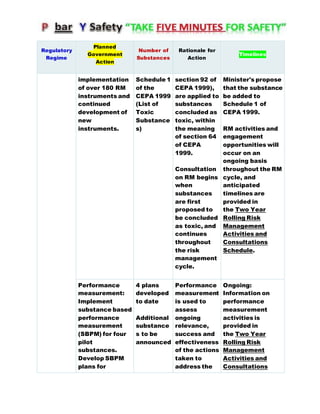
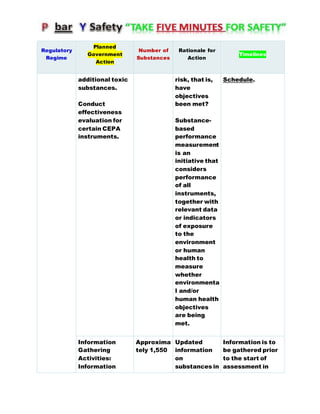
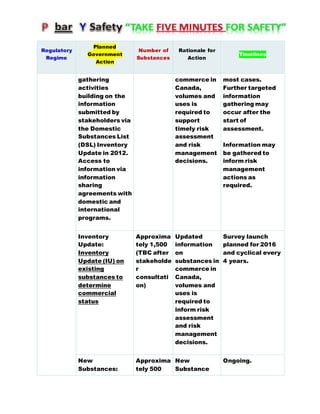
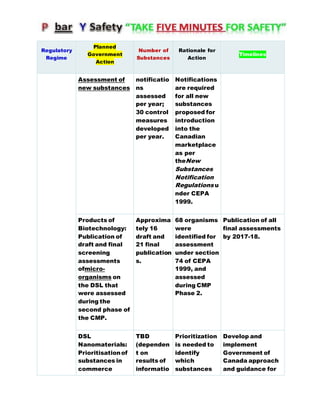
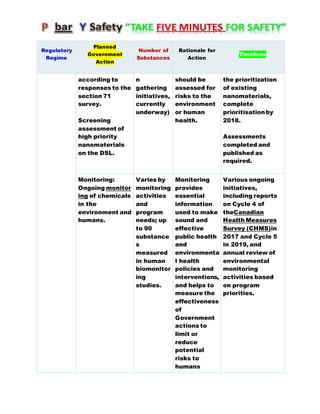
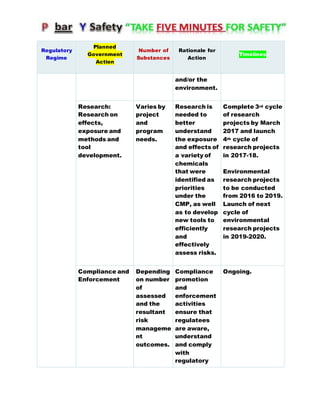
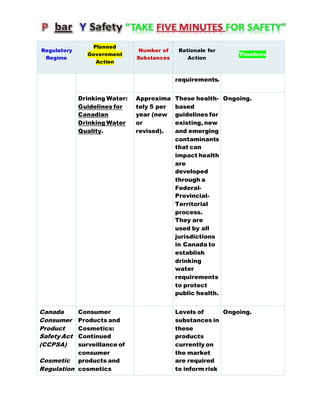
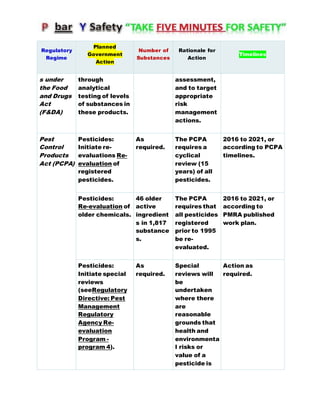
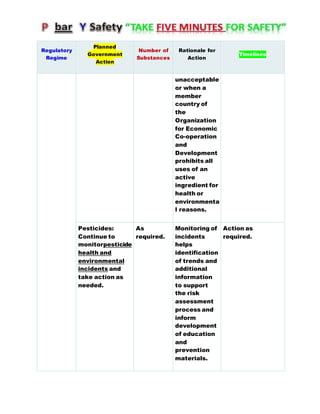
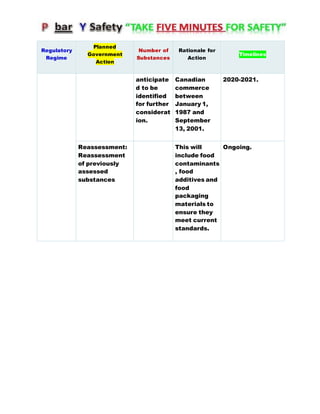
Canada's Chemicals Management Plan is set to span into 2020, reviewing the Canadian Environmental Protection Act (CEPA) of 1999 to ensure safety against chemical risks. The federal government, in conjunction with various stakeholders, aims to manage chemicals of concern by enhancing research, monitoring, and industry stewardship, alongside implementing risk management actions. The initiative also emphasizes transparency and stakeholder engagement throughout the chemical assessment process and ongoing monitoring of chemical impacts on health and the environment.