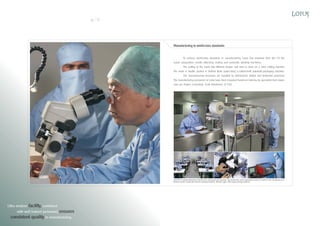
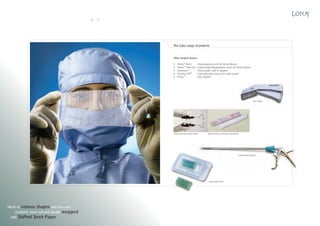
Lotus Surgicals Pvt. Ltd. is a modern medical device manufacturing facility located in Dehradun, India, specializing in surgical sutures and other medical devices. The facility features class 10,000 clean rooms and employs advanced sterilization and packaging technologies to ensure high product quality and compliance with international standards. Led by experienced professionals, Lotus aims to establish a prominent position in the global medical device market while maintaining a commitment to environmental sustainability.